At a Glance
| Property | Value |
|---|---|
| Topic | NAD+ IV Therapy |
| Evidence Level | Moderate (clinical studies available) |
| Mechanism | NAD+ is a coenzyme essential for over 500 enzymatic reactions including mitochondrial energy production, DNA repair via sirtuins, and… |
| Protocol | 250-500 mg IV NAD+ administered over two to four h |
| Key Takeaway | NAD+ is a coenzyme essential for over 500 enzymatic reactions including mitochondrial energy production, DNA repair via sirtuins, and immune signaling via CD38. |
Nicotinamide adenine dinucleotide — NAD+ — is not a supplement trend. It is a coenzyme found in every living cell, essential for over 500 enzymatic reactions. Without adequate NAD+, cells cannot produce energy efficiently, repair DNA, or regulate the epigenetic changes associated with aging.
Here is the problem: NAD+ levels decline with age. By the time a person reaches their fifties, intracellular NAD+ may be 50% lower than it was in their twenties [1]. This decline correlates with many of the hallmarks of aging I discussed in my longevity overview — mitochondrial dysfunction and cellular energy, genomic instability, and chronic inflammation pathways among them.
The question that matters clinically is whether restoring NAD+ levels can meaningfully influence aging processes. The honest answer is that the mechanistic data is strong, the animal data is compelling, and the human clinical data is growing but not yet definitive.
The Mechanistic Rationale
NAD+ serves as a critical substrate for three families of enzymes that are directly relevant to aging:

Sirtuins (SIRT1-7) are NAD+-dependent deacetylases that regulate gene expression, DNA repair, mitochondrial biogenesis, and inflammation. They cannot function without NAD+. When NAD+ levels drop, sirtuin activity drops with it. This is not theoretical — it is well-established biochemistry [2].
PARPs (poly ADP-ribose polymerases) consume NAD+ to repair DNA damage. As DNA damage accumulates with age, PARPs consume more NAD+, creating a competition with sirtuins for a shrinking NAD+ pool. This competition may be one mechanism through which aging becomes self-accelerating.
CD38, an NAD+-consuming enzyme, increases with age and is one of the primary drivers of age-related NAD+ decline. CD38 activity rises with chronic inflammation, creating another feedback loop.
IV vs. Oral: Why the Route Matters
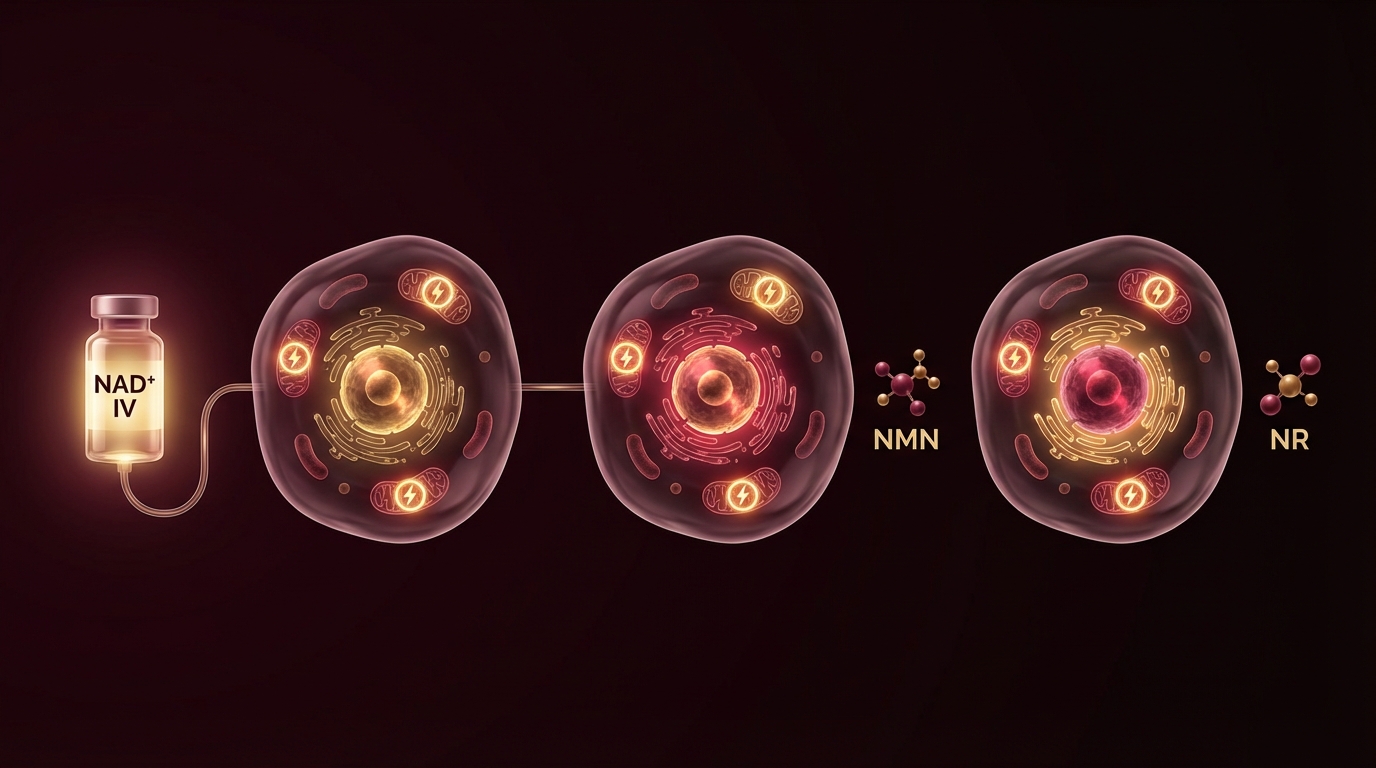
Patients frequently ask why I recommend IV administration rather than oral supplements like NMN (nicotinamide mononucleotide) or NR (nicotinamide riboside).
The honest answer is that both approaches have merit, and neither is definitively superior in all contexts.
IV NAD+ delivers the molecule directly into the bloodstream, bypassing gastrointestinal absorption and hepatic first-pass metabolism. In my clinical experience, the effects are more immediate and pronounced — patients typically report improved mental clarity, energy, and a sense of physiological “reset” during or shortly after infusion. The limitation is that IV therapy requires clinical administration, is more expensive, and the infusions can cause discomfort (flushing, nausea, chest tightness) that requires careful rate titration.
Oral precursors (NMN, NR) are more accessible and can be taken daily. Human trials have demonstrated that oral NMN increases blood NAD+ levels in a dose-dependent manner [3]. Whether this translates to equivalent intracellular NAD+ repletion compared to IV delivery has not been directly compared in controlled human trials. For a detailed breakdown of every oral NAD+ precursor — including NADH, NAD3, cofactor requirements, and stacking strategy — see the NAD Supplement Guide.
What I tell my patients: IV NAD+ is best suited for periodic intensive restoration — I typically recommend a series of infusions as an initial loading protocol, followed by maintenance with oral precursors. This is a clinical observation, not a randomized trial result, and I am transparent about that distinction.
What the Evidence Shows
Animal Studies
The preclinical data for NAD+ repletion is extensive and consistently positive. In mouse models, boosting NAD+ has demonstrated improvements in mitochondrial function, insulin sensitivity, exercise capacity, DNA repair, and lifespan extension in certain strains [4]. These results have been replicated across multiple laboratories using different NAD+ precursors.
It is important to note that mouse results do not automatically translate to humans. They provide mechanistic rationale and justify clinical investigation, but they are not clinical proof.
Human Studies
A growing number of human trials have been completed:
- A 2022 randomized controlled trial of oral NMN (250 mg daily) in healthy middle-aged adults showed increased blood NAD+ levels and improved muscle insulin sensitivity and remodeling after 10 weeks [5].
- Multiple safety studies have confirmed that both oral NMN and NR are well-tolerated in humans at doses up to 1,200 mg daily.
- A 2023 trial demonstrated that NMN supplementation improved aerobic capacity in middle-aged recreational runners [6].
The human evidence is promising but still relatively early-stage. We do not yet have large, long-term randomized trials demonstrating hard clinical endpoints like reduced disease incidence or extended lifespan.
Clinical Observation
At Klinik St. Georg, we have administered IV NAD+ to several hundred patients. What we have observed consistently:
- Improved subjective energy levels, typically lasting two to four weeks after a series of infusions
- Enhanced cognitive clarity, particularly in patients with baseline cognitive complaints
- Improved exercise recovery
- In patients undergoing biological age testing, a trend toward improved epigenetic age scores after sustained NAD+ protocols (combined with other interventions, so attribution is difficult)
I want to be transparent: these are clinical observations, not controlled trial results. Individual variation plays a significant role. Not every patient responds dramatically, and I have learned that patients with the most pronounced NAD+ depletion (often those with chronic illness, post-COVID fatigue, or significant metabolic dysfunction) tend to respond most noticeably.
Dosing and Protocol
In my practice, a typical IV NAD+ protocol looks like this:
Initial loading phase: 250-500 mg IV NAD+ administered over two to four hours, repeated three to five times over one to two weeks. The infusion rate is critical — too fast, and patients experience significant flushing, nausea, and chest tightness. I start conservatively and titrate based on tolerance.
Maintenance: After the loading phase, I transition patients to oral NMN (500-1,000 mg daily) with periodic IV “booster” infusions every four to eight weeks, adjusted based on clinical response and laboratory markers.
Supportive nutrients: NAD+ metabolism requires adequate levels of B vitamins (particularly B3, B6, B12), magnesium, and zinc. I ensure these are optimized before or alongside NAD+ therapy.
Safety Considerations
IV NAD+ is generally well-tolerated when administered properly. Common side effects include:
- Flushing and warmth during infusion
- Nausea (rate-dependent — slowing the infusion usually resolves this)
- Chest or abdominal tightness
- Headache
These effects are typically transient and manageable with rate adjustment. Serious adverse events are rare in published literature and in my clinical experience. For a detailed breakdown of each reaction — mechanisms, frequency data, and practical mitigation strategies — see NAD+ IV Side Effects: What to Expect and How to Manage Them.
One area of ongoing discussion is whether boosting NAD+ could theoretically support the survival of existing cancer cells, since cancer cells also rely on NAD+ for energy metabolism. The preclinical data on this question is mixed — some studies suggest NAD+ repletion has anti-tumor effects through enhanced immune surveillance, while others raise theoretical concerns [7]. In my practice, I approach NAD+ therapy in patients with active malignancy on a case-by-case basis, weighing the evidence carefully.
The Bottom Line
NAD+ decline is a real and measurable phenomenon of aging. The mechanistic rationale for NAD+ repletion is among the strongest in longevity medicine. The human clinical data is growing and generally supportive, though we await larger and longer trials for definitive answers.
In the context of a comprehensive longevity program, NAD+ optimization — whether through IV therapy, oral precursors, or both — is a reasonable and evidence-informed intervention. It is not a standalone solution to aging, and anyone who presents it as such is overpromising. Patients pursuing intensive longevity work may find that combining NAD+ IV with hyperbaric oxygen therapy amplifies the mitochondrial biogenesis signal — both pathways converge on PGC-1α and sirtuin activation, and staggering sessions (HBOT in the morning, NAD+ IV in the afternoon) is a practical stacking approach.

Clinical Perspective — Julian Douwes M.D.
When patients ask me whether to pursue IV NAD+ or simply take oral NMN, my answer depends on where they are starting from. For patients with significant fatigue, post-COVID syndrome, or measurable metabolic dysfunction, I recommend an initial IV loading series — 500 mg over three to four hours for four consecutive days — because the clinical response is more immediate and pronounced, and it gives us a clear signal of whether NAD+ depletion is a meaningful contributor to their symptoms. After the loading phase, I transition to oral NMN at 500 mg daily with a quarterly IV maintenance infusion. For generally healthy patients pursuing longevity optimization, I typically start with oral NMN alone at 500-1,000 mg daily and reserve IV therapy for those who do not see subjective improvement after eight weeks. The key insight from my experience is that the patients who respond most dramatically to IV NAD+ are those who were most depleted to begin with — and that response itself becomes diagnostic information.
This reflects my clinical experience and professional opinion. It is not a substitute for individual medical advice.
References
- Camacho-Pereira J, et al. CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction. Cell Metabolism. 2016;23(6):1127-1139.
- Imai S, Guarente L. NAD+ and sirtuins in aging and disease. Trends in Cell Biology. 2014;24(8):464-471.
- Yoshino M, et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021;372(6547):1224-1229.
- Mills KF, et al. Long-Term Administration of Nicotinamide Mononucleotide Mitigates Age-Associated Physiological Decline in Mice. Cell Metabolism. 2016;24(6):795-806.
- Yi L, et al. The efficacy and safety of NMN supplementation in clinical studies. Journal of Biomedical Science. 2022;29:69.
- Liao B, et al. Nicotinamide mononucleotide supplementation enhances aerobic capacity in amateur runners. Journal of the International Society of Sports Nutrition. 2021;18(1):54.
- Navas LE, Carnero A. NAD+ metabolism, stemness, the immune response, and cancer. Signal Transduction and Targeted Therapy. 2021;6(1):2.
This content is educational and does not constitute medical advice. NAD+ therapy should be administered under medical supervision by a qualified physician.