At a Glance
| Property | Value |
|---|---|
| Evidence Level | Strong (mechanistic and animal data); Moderate (human interventions) |
| Primary Use | Cellular quality control, neurodegeneration prevention, longevity |
| Key Mechanism | Lysosomal degradation of damaged organelles, misfolded proteins, and intracellular debris |
| How to Activate | Fasting (18+ hours), exercise, sleep, caloric restriction |
| Pharmacological Activators | Rapamycin, spermidine (both under investigation) |
Why Your Body Slows Down With Age — and What You Can Do About It
If you are over 40 and noticing that you recover more slowly from illness, that your energy is not what it used to be, or that your body seems to accumulate damage faster than it repairs it, you are experiencing something measurable: the decline of your body’s cellular maintenance system. This system — called autophagy — is one of the most fundamental determinants of how well you age.
Autophagy is your body’s built-in recycling program. Every cell generates waste. Proteins misfold. Mitochondria become dysfunctional. Intracellular debris accumulates. Autophagy identifies this damaged material, breaks it down, and recycles the building blocks into new, functional structures. The word comes from the Greek “auto” (self) and “phagein” (to eat). Self-eating. Your cells literally consume their own damaged components.
Yoshinori Ohsumi won the 2016 Nobel Prize in Physiology or Medicine for elucidating the molecular mechanisms of autophagy — a recognition of just how fundamental this process is to cellular life [1]. And yet, in clinical medicine, autophagy remains underappreciated and poorly understood by most practitioners.
Here is what the evidence shows, what you can actually do to support autophagy, and where the hype exceeds the science.
How Autophagy Works
Autophagy is not a single event. It is a multi-step process involving dozens of proteins working in coordinated sequence [2].
Initiation: When the cell senses nutrient deprivation, energy stress, or accumulation of damaged components, autophagy is initiated. The key regulators are mTOR (which inhibits autophagy when nutrients are abundant) and AMPK (which activates autophagy during energy stress). When mTOR is suppressed and AMPK is activated — as happens during fasting or exercise — the autophagy machinery is switched on.
Phagophore formation: A double-membraned structure called a phagophore begins to form around the material targeted for degradation. This is like a trash bag forming around cellular waste.
Cargo selection: Autophagy is not random demolition. Selective autophagy uses receptor proteins (p62/SQSTM1, NBR1, OPTN, and others) to identify and tag specific cargo for degradation. Damaged mitochondria are targeted through a process called mitophagy — a pathway that can be pharmacologically supported with urolithin A, which has demonstrated PINK1/Parkin-mediated mitophagy activation in phase-II human trials. Misfolded protein aggregates are targeted through aggrephagy. Intracellular pathogens can be targeted through xenophagy. This selectivity is crucial — you want the system to remove damaged components while preserving functional ones.
Autophagosome formation: The phagophore seals around the cargo, forming a complete double-membraned vesicle called an autophagosome.
Lysosomal fusion and degradation: The autophagosome fuses with a lysosome — an acidic compartment containing digestive enzymes. The cargo is broken down into amino acids, fatty acids, and nucleotides, which are released back into the cytoplasm for reuse.
Recycling: The recovered building blocks are used to synthesize new proteins, membranes, and organelles. This is cellular recycling at its most elegant — destruction in service of renewal.
Why Autophagy Declines With Age
Autophagy becomes less efficient with age, and this decline has consequences across virtually every organ system [3].
Several factors contribute to the age-related decline:
Lysosomal dysfunction: Lysosomes accumulate lipofuscin — an indigestible, pigmented waste product — over a lifetime. As lipofuscin burden increases, lysosomal degradative capacity decreases. The recycling center itself becomes clogged.
Reduced AMPK signaling: The sensitivity of AMPK to energy stress diminishes with age, reducing the upstream signal that activates autophagy.
Chronic mTOR activation: Modern diets — high in protein, sugar, and total calories — chronically activate mTOR, which constitutively suppresses autophagy. Most people in developed societies rarely experience the nutrient deprivation that triggers robust autophagic activity.
Transcriptional changes: Expression of key autophagy genes (ATG5, ATG7, BECN1) declines with age in multiple tissues [4].
The consequences of impaired autophagy are directly relevant to the diseases of aging:
- Neurodegeneration: Alzheimer’s, Parkinson’s, and Huntington’s diseases all involve accumulation of misfolded protein aggregates (amyloid-beta, alpha-synuclein, huntingtin) that healthy autophagy would normally clear. Impaired autophagy in neurons is not merely associated with these diseases — in animal models, genetic disruption of autophagy in the brain produces neurodegeneration spontaneously [5].
- Cancer: Autophagy plays a complex role in cancer. In healthy cells, it functions as a tumor suppressor by removing damaged DNA, dysfunctional mitochondria, and oncogenic proteins. Mice with reduced autophagy (Beclin 1+/-) develop spontaneous tumors at higher rates [6]. However, established tumors can hijack autophagy to survive metabolic stress — making the relationship context-dependent.
- Cardiovascular disease: Autophagy maintains cardiac myocyte function and vascular integrity. Impaired cardiac autophagy contributes to age-related heart failure and atherosclerosis.
- Metabolic dysfunction: Reduced autophagy in the liver and pancreas contributes to insulin resistance, fatty liver disease, and impaired glucose homeostasis.
How to Activate Autophagy
This is the question every patient asks. Here is what the evidence actually supports — and where I see exaggeration in the wellness space.
Fasting — The Most Potent Activator
Nutrient deprivation is the strongest physiological stimulus for autophagy. When you stop eating, insulin falls, mTOR is suppressed, AMPK is activated, and the entire autophagic machinery is upregulated.
The critical question is: how long must you fast to meaningfully activate autophagy?
This is where honest communication matters. We cannot easily measure autophagy in living humans. Unlike blood glucose or ketones, there is no simple test for autophagic flux. What we know comes from animal studies, tissue biopsies in research settings, and indirect biomarkers.
In animal models, significant autophagy upregulation begins after approximately 16-24 hours of fasting and becomes more robust with longer fasts [7]. The transition from fed to fasting is not a binary switch — it is a gradient. Some autophagic activity occurs during normal overnight fasting. It increases progressively with fasting duration.
Based on the available evidence and my clinical experience, I recommend the following framework:
- 12-16 hours: Baseline autophagy activation. Sufficient for general metabolic health but likely suboptimal for deep cellular cleanup.
- 18-24 hours: Significant autophagy upregulation based on animal data. This is the range I recommend for patients pursuing autophagy as a longevity strategy — achievable through intermittent fasting protocols without excessive difficulty.
- 24-48 hours: Deeper autophagy. In animal models, this is where measurable clearance of damaged organelles becomes most pronounced. Practical for occasional use under medical supervision.
- Beyond 48 hours: Additional benefits are possible but must be weighed against the risks of prolonged fasting (muscle catabolism, electrolyte imbalances, refeeding syndrome).
Anyone selling you a supplement that “activates autophagy as effectively as fasting” is misleading you. The nutrient-deprivation signal is the most powerful driver of autophagy, and no supplement replicates it.
Exercise
Exercise activates autophagy through multiple mechanisms: AMPK activation, increased cellular stress, and metabolic demand. Both aerobic and resistance exercise stimulate autophagic flux in skeletal muscle, and emerging evidence suggests exercise-induced autophagy occurs in the brain, liver, and other organs as well [8].
The specific relationship between exercise intensity, duration, and autophagy activation is still being characterized. What we know is that regular exercise — particularly when combined with some degree of caloric or time restriction — creates a synergistic autophagic stimulus.
Zone 2 training may be particularly relevant because it stresses the oxidative muscle fibers and mitochondria most dependent on autophagic quality control. High-intensity exercise also triggers autophagy, though the mechanisms may differ (greater AMPK activation during intense efforts).
Sleep
Autophagy in the brain is particularly active during sleep. The glymphatic system — which clears metabolic waste from the central nervous system — works primarily during sleep, and autophagic processes in neurons are upregulated during rest [9]. Chronic sleep deprivation impairs both glymphatic clearance and autophagy, contributing to the accumulation of neurotoxic proteins.
This is one of many reasons I consider sleep optimization a non-negotiable component of any longevity protocol. You cannot fast and exercise your way out of poor sleep — the brain requires sleep-dependent clearance mechanisms that cannot be replicated while awake.
Pharmacological Activators
Rapamycin: The mTOR inhibitor rapamycin is the most potent pharmacological activator of autophagy. It has extended lifespan in every organism tested, including mice when started late in life [10]. In my practice, I use low-dose rapamycin in selected longevity patients, and autophagy activation is one of the key mechanisms underlying its benefit. However, rapamycin has real immunosuppressive effects and requires careful dosing and monitoring.
Spermidine: A naturally occurring polyamine found in wheat germ, aged cheese, and fermented soy products. Spermidine supplementation extends lifespan in animal models through autophagy-dependent mechanisms and directly inhibits EP300 acetyltransferase — the same epigenetic pathway suppressed by caloric restriction. The Bruneck Study — a prospective human cohort — found that higher dietary spermidine intake was associated with reduced all-cause mortality [11]. This is observational data, not causal proof, but the mechanistic coherence is compelling. A 12-month human RCT has since confirmed reduced inflammatory markers and improved memory performance. Typical supplementation doses are 0.5–1.2 mg daily of a standardized wheat-germ extract.
Metformin: Activates AMPK and has been shown to upregulate autophagy in multiple tissues. The TAME trial (Targeting Aging with Metformin) is investigating whether metformin can slow aging in humans — and autophagy activation is one of its proposed mechanisms.
Resveratrol and other polyphenols: Some evidence for autophagy activation at high doses, but human data is limited and bioavailability is poor. I do not consider polyphenol supplements reliable autophagy activators at currently achievable doses.
What I See in Practice
I cannot measure autophagy directly in my patients. No clinician can. What I observe instead are the downstream consequences of interventions that are known to activate autophagy.
In patients who adopt consistent intermittent fasting protocols (18:6 or periodic 24-hour fasts), combined with regular exercise and sleep optimization, I consistently observe:
- Improvements in inflammatory markers (hsCRP, IL-6) over 3-6 months
- Improved metabolic markers (fasting insulin, HOMA-IR, triglycerides)
- In patients on low-dose rapamycin, additional improvements in senescence markers and biological age scores
- Subjective improvements in cognitive clarity, joint comfort, and energy — though these are harder to attribute to autophagy specifically versus the broader lifestyle intervention
What I tell my patients: autophagy is not something you “biohack” with a single supplement or a weekend fast. It is a fundamental cellular process that responds to the sustained metabolic environment you create through daily habits. Fasting, exercise, sleep, and (in selected cases) pharmacological support create the conditions under which autophagy thrives. There is no shortcut.
The Relationship Between Autophagy and Senescent Cells
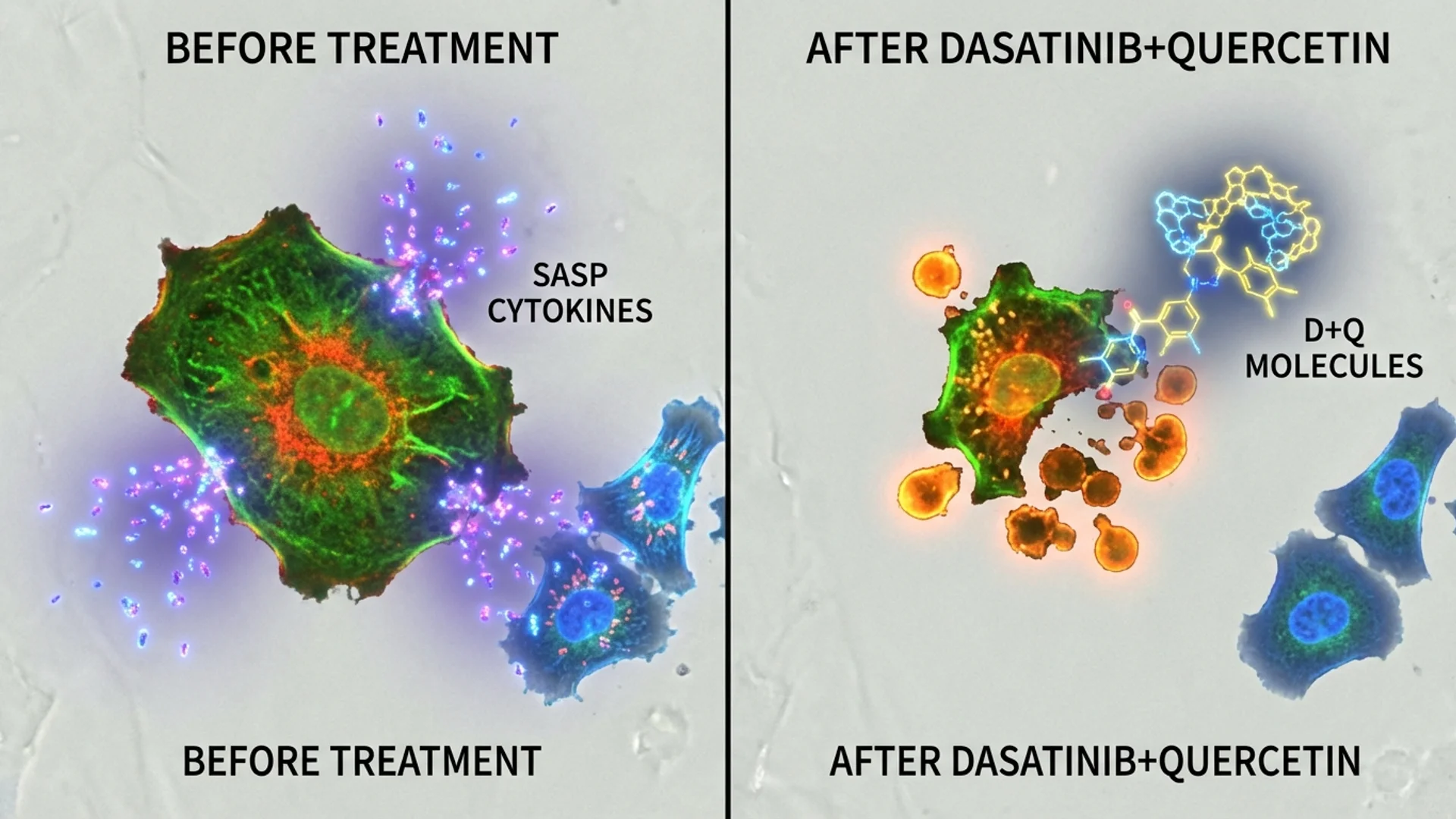
Autophagy and senolytic therapy are complementary but distinct longevity strategies. Autophagy prevents cellular damage from accumulating to the point where senescence is triggered. Senolytics clear senescent cells after they have already formed.
Think of autophagy as preventive maintenance and senolytics as remediation. A well-maintained cell is less likely to become senescent. But once a cell has entered senescence, autophagy alone cannot clear it — senolytic agents are needed to eliminate these “zombie cells.”
In my longevity protocols, I address both pathways: lifestyle interventions (fasting, exercise, sleep) to maximize autophagic activity and reduce senescent cell formation, and periodic senolytic protocols (dasatinib + quercetin, or fisetin) to clear cells that have already crossed the threshold.
Some emerging research suggests that autophagy activation may enhance senolytic efficacy — potentially by sensitizing pre-senescent cells to clearance or by improving the cellular environment after senescent cells are removed. This is an area of active investigation, and the data is early, but the mechanistic rationale is sound.
Safety and Considerations
Autophagy activation through fasting and exercise is generally safe for healthy adults. However:
- Cancer patients: The role of autophagy in established cancer is complex. In some cancers, autophagy provides a survival advantage to tumor cells under metabolic stress. Patients with active malignancy should discuss autophagy-focused interventions with their oncologist. In our practice, we evaluate this on a case-by-case basis.
- Immunocompromised patients: Pharmacological autophagy activators (particularly rapamycin) require careful consideration in patients with impaired immune function.
- Eating disorders: Promoting fasting as an “autophagy hack” can be harmful for patients with a history of disordered eating. In these patients, I focus on exercise and sleep as the primary autophagy activators.
- Pregnancy: Significant caloric restriction and extended fasting are contraindicated during pregnancy and breastfeeding.
Common Misconceptions
“I can measure my autophagy with a blood test.” You cannot. There is currently no validated, commercially available blood test for autophagic flux in humans. Claims to the contrary are misleading. Researchers use tissue biopsies, LC3-II/LC3-I ratios, and p62 levels in research settings, but these are not available or practical for clinical use.
“Coffee activates autophagy.” There is some preclinical evidence that polyphenols in coffee may have modest autophagy-stimulating effects. But drinking coffee is not a substitute for fasting. The autophagic stimulus from coffee is orders of magnitude smaller than from actual nutrient deprivation. If coffee had meaningful autophagy effects, nobody who drinks coffee regularly would develop neurodegeneration — and that is clearly not the case.
“Autophagy is always good, more is always better.” Excessive or dysregulated autophagy can be harmful. Autophagic cell death is a recognized phenomenon. The goal is regulated, healthy autophagic flux — not maximal autophagy at all times. The body’s natural cycling between fed (growth/building) and fasted (cleanup/repair) states is the ideal pattern.
The Bottom Line
Autophagy is one of the most fundamental cellular quality control mechanisms, and its decline with age directly contributes to neurodegeneration, cancer, metabolic disease, and accelerated aging. The most evidence-supported activators are fasting (18+ hours), regular exercise, adequate sleep, and — in selected cases — pharmacological agents like rapamycin and spermidine.
What I tell my patients: autophagy is not a hack. It is a biological process that your ancestors activated naturally through periodic food scarcity and physical labor. Modern life — with constant food availability, sedentary behavior, and poor sleep — chronically suppresses it. Restoring the conditions that support autophagy requires deliberate choices: time-restricted eating, regular movement, prioritized sleep. These are not exotic interventions. They are the foundations of healthy aging.
References
- Ohsumi Y. Historical landmarks of autophagy research. Cell Res. 2014;24(1):9-23. PMID: 24366340.
- Mizushima N, Komatsu M. Autophagy: Renovation of cells and tissues. Cell. 2011;147(4):728-741. PMID: 22078875.
- Aman Y, Schmauck-Medina T, Hansen M, et al. Autophagy in healthy aging and disease. Nat Aging. 2021;1(8):634-650. PMID: 34901876.
- Lipinski MM, Zheng B, Lu T, et al. Genome-wide analysis reveals mechanisms modulating autophagy in normal brain aging and in Alzheimer’s disease. Proc Natl Acad Sci USA. 2010;107(32):14164-14169. PMID: 20660724.
- Hara T, Nakamura K, Matsui M, et al. Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature. 2006;441(7095):885-889. PMID: 16625204.
- Qu X, Yu J, Bhagat G, et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J Clin Invest. 2003;112(12):1809-1820. PMID: 14638851.
- Alirezaei M, Kemball CC, Flynn CT, et al. Short-term fasting induces profound neuronal autophagy. Autophagy. 2010;6(6):702-710. PMID: 20534972.
- He C, Sumpter R Jr, Bhatt D, et al. Exercise induces autophagy in peripheral tissues and in the brain. Autophagy. 2012;8(10):1548-1551. PMID: 22892563.
- Xie L, Kang H, Xu Q, et al. Sleep drives metabolite clearance from the adult brain. Science. 2013;342(6156):373-377. PMID: 24136970.
- Harrison DE, Strong R, Sharp ZD, et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature. 2009;460(7253):392-395. PMID: 19587680.
- Kiechl S, Pechlaner R, Willeit P, et al. Higher spermidine intake is linked to lower mortality: A prospective population-based study. Am J Clin Nutr. 2018;108(2):371-380. PMID: 29955838.