At a Glance
| Parameter | Detail |
|---|---|
| Procedure name | Intravenous Laser Blood Irradiation (ILBI / ILIB) |
| Wavelengths used | 630–650 nm (red), 405 nm (violet), 520 nm (green), 980 nm (near-infrared) |
| Session length | 20–60 minutes |
| Standard course | 6–10 sessions (daily or every other day) |
| Setting | Outpatient clinic, IV therapy suite |
| Pain level | Minimal — equivalent to a standard blood draw |
| Evidence grade | Moderate (RCTs in cardiovascular, metabolic, and inflammatory conditions) |
| Combination therapies | Often combined with ozone, NAD⁺ IV, or high-dose vitamin C |
Patients who come to my clinic for complex chronic conditions — Lyme disease, post-COVID syndrome, autoimmune flares, or profound fatigue — often arrive having exhausted the conventional pharmacological toolkit. Intravenous laser blood irradiation (ILBI), sometimes called intravenous photobiomodulation or IV laser therapy, is one of the tools I reach for when we need to reduce systemic inflammation, support mitochondrial recovery, and recalibrate immune function simultaneously. It is not a miracle. It is, however, a genuinely interesting intervention with a solid enough mechanistic basis and a growing clinical evidence base that I want to explain it properly — so that patients can make informed decisions rather than being swayed by either dismissal or hype.
What IV Laser Therapy Actually Is
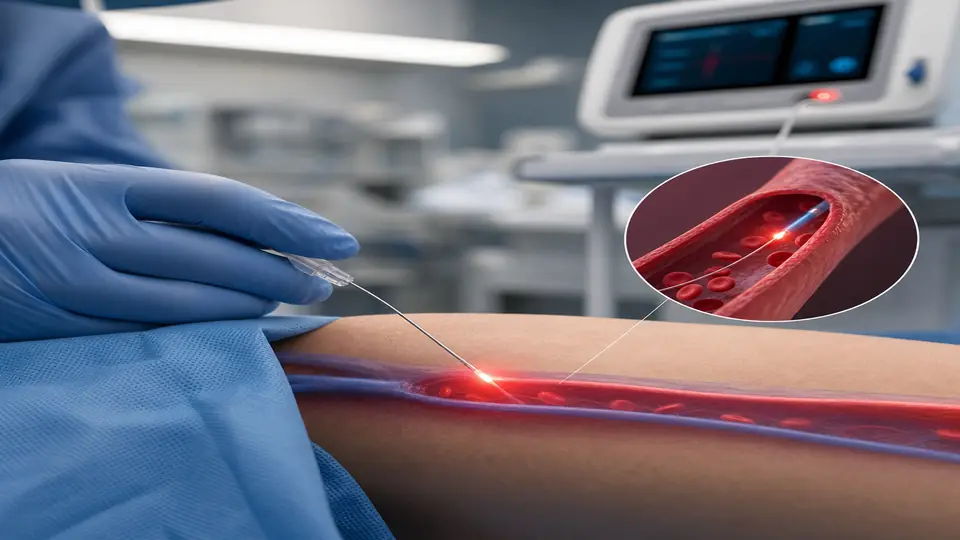
Photobiomodulation (PBM) as an external modality — shining therapeutic laser or LED light on tissue — has been studied for decades. IV laser therapy takes the same principle and eliminates the distance problem: a sterile, single-use fiber-optic probe is threaded through a standard IV cannula into a peripheral vein (typically the antecubital fossa), and low-level laser light is delivered directly into the bloodstream.
This is categorically different from high-powered surgical lasers. The devices used here operate at milliwatt power levels — enough to stimulate cellular photoreceptors without generating heat or causing tissue damage. As blood flows continuously past the probe tip, a large volume of circulating cells receives photonic stimulation over the course of a session.
The most established wavelengths in clinical use are:
- Red (630–650 nm): Deepest evidence base; primary driver of cytochrome c oxidase activation and mitochondrial ATP synthesis
- Green (520 nm): Strong affinity for hemoglobin; improves oxygen-carrying capacity and red cell deformability
- Violet/UV (405 nm): Antimicrobial and antiviral properties; activates vitamin D metabolites in blood
- Near-infrared (808–980 nm): Deepest tissue penetration; potent anti-inflammatory and neuroprotective effects
Some devices deliver a single wavelength; multi-wavelength platforms allow the treating physician to tailor sessions to the clinical objective.
The Mechanism: Why Light Changes Blood Biology
The core mechanism relies on light-sensitive chromophores in blood cells responding to specific photon energies. Three primary pathways are well-documented:
Mitochondrial Activation
Cytochrome c oxidase, the terminal enzyme of the mitochondrial electron transport chain, is a strong photoacceptor for red and near-infrared wavelengths. Laser photons dissociate inhibitory nitric oxide (NO) from the enzyme’s active site, restoring electron flow, increasing mitochondrial membrane potential, and upregulating ATP production. In cells under chronic oxidative stress or hypoxic conditions — common in patients with complex chronic illness — this translates to measurably improved cellular energy metabolism.
Redox Modulation and Reactive Oxygen Species (ROS) Signaling
At the low power densities used in ILBI, laser light induces a transient, sub-toxic increase in reactive oxygen species that acts as a signaling event rather than a damaging insult. This hormetic ROS signal activates Nrf2, the master regulator of antioxidant gene expression, upregulating superoxide dismutase, catalase, and glutathione peroxidase. The net result is improved antioxidant capacity — paradoxically, a small oxidant stimulus makes the system more resistant to oxidative stress.
Immune Modulation
Lymphocytes, monocytes, and granulocytes all carry light-sensitive surface proteins and intracellular chromophores. ILBI has been shown to shift macrophage polarization from pro-inflammatory M1 toward regulatory M2 phenotypes, reduce circulating levels of TNF-α, IL-6, and IL-1β, and enhance NK cell cytotoxicity. For patients with dysregulated immune responses — whether in autoimmunity, post-viral immune exhaustion, or persistent infection — this bidirectional immune modulation is clinically meaningful.
Clinical Evidence and Applications
ILBI has been studied most rigorously in Russian and German medical literature since the 1980s, with an expanding body of peer-reviewed work now appearing in English-language journals. The evidence is heterogeneous in quality but consistently points in the same direction.
Cardiovascular and Metabolic Conditions
A 2019 randomized controlled trial in patients with type 2 diabetes demonstrated significant reductions in HbA1c, fasting glucose, and LDL cholesterol after 10 ILBI sessions compared to controls. Hemorheological improvements — reduced blood viscosity, enhanced red cell deformability, decreased platelet aggregation — have been documented across multiple trials. These effects are particularly relevant in patients with microclot formation (as seen in Long COVID) or impaired microcirculation in chronic infection.
Chronic Fatigue and Post-Viral Syndromes
In my clinical experience, ILBI is among the most reliably effective tools for patients presenting with post-viral fatigue — whether post-COVID, post-Epstein-Barr, or post-Lyme. The combination of mitochondrial reactivation, improved oxygen delivery, and downregulation of the chronic inflammatory cytokine profile addresses multiple drivers of fatigue simultaneously. Several observational studies and one small RCT in CFS/ME patients showed significant improvement in energy scores and cognitive function after 8–10 sessions.
Inflammatory and Autoimmune Conditions
The anti-inflammatory effects of ILBI have been studied in rheumatoid arthritis, psoriatic arthritis, and inflammatory bowel disease, with most trials showing reductions in disease activity scores and inflammatory markers. These are not remission-inducing results, but they represent clinically meaningful adjunctive benefit.
Antimicrobial Effects
The violet (405 nm) wavelength has demonstrable bactericidal and virucidal activity in vitro — relevant when ILBI is used as part of a broader anti-infective protocol in chronic Lyme disease or reactivated viral co-infections. This is not a replacement for targeted antimicrobial therapy, but it may reduce pathogen burden systemically while simultaneously supporting immune competence.
IV Laser vs. External Photobiomodulation
Patients often ask why we would use IV laser when external photobiomodulation devices are available for home use. The distinction matters:
External PBM (panel devices, laser helmets, handheld probes) delivers photons that must penetrate skin, subcutaneous fat, fascia, and muscle to reach target tissue. Effective tissue depth varies by wavelength and device power, but meaningful photon delivery to deep viscera, circulating blood cells, or the brain is limited.
IV laser bypasses all barriers. The probe sits inside the bloodstream. Every milliliter of blood that flows past the probe tip during a session is directly irradiated. Over a 30-minute session at a typical venous flow rate, the majority of the patient’s circulating blood volume is exposed multiple times. The dose delivered to blood cells is orders of magnitude higher than any external device can achieve.
These are complementary rather than competing modalities. External PBM remains valuable for local tissue effects (wound healing, musculoskeletal pain, transcranial applications). IV laser is indicated when the target is systemic — circulating immune cells, red cell function, whole-body inflammation, or metabolic reprogramming.
What to Expect During Treatment
The procedure is straightforward:
- Cannulation: A standard IV catheter is placed, most commonly in the antecubital vein. No different from a routine blood draw.
- Probe insertion: A sterile, single-use fiber-optic probe (0.2–0.4 mm diameter) is threaded through the cannula into the vein. Patients feel nothing beyond the initial cannula placement.
- Irradiation: The laser device is activated. Sessions run 20–60 minutes depending on wavelength, power output, and clinical protocol. Patients typically read, rest, or listen to music during treatment.
- Completion: The probe and cannula are removed. No recovery period is required; patients can drive and return to normal activity immediately.
Sensation during treatment: Most patients report nothing. Some notice mild warmth at the insertion site or a faint perception of light diffusing through the skin. Rarely, patients report a brief sensation of relaxation or mild lightheadedness in the first session — consistent with acute vasodilation and improved microcirculatory flow.
Standard course: Most protocols call for 6–10 sessions delivered daily or on alternating days. Maintenance sessions (monthly or quarterly) are used in patients with ongoing conditions.
Who Benefits Most — and Who Should Proceed with Caution
Strongest clinical rationale:
- Post-viral fatigue syndrome (Long COVID, post-EBV, post-Lyme)
- Chronic inflammatory conditions with elevated CRP, IL-6, or ferritin
- Impaired microcirculation (Raynaud’s, diabetic neuropathy, microclot syndromes)
- Chronic Lyme and co-infections (as part of a broader protocol)
- Metabolic syndrome with insulin resistance
- Recovery support after intensive antimicrobial or immunomodulatory treatment
Proceed with caution or individualize:
- Active photosensitizing medications (certain antibiotics, antifungals, and psychiatric drugs)
- Known porphyria
- Pregnancy (insufficient safety data)
- Active cancer (theoretical concern about stimulating tumor vascularity — discuss with treating oncologist)
- Severe coagulopathy or active bleeding
Contraindications are few, but a thorough medication and history review before initiating treatment is non-negotiable. In my practice, ILBI is introduced after baseline labs — CBC, CMP, inflammatory markers, and where relevant, coagulation studies — have been reviewed.
Combining IV Laser with Other Protocols
ILBI is rarely used in isolation in my clinic. It is typically one component of a multi-modal IV protocol. Common combinations include:
- ILBI + Major Ozone Autohemotherapy (MAH): Complementary antimicrobial and immune-modulatory effects; ozone provides oxidative burst while ILBI supports mitochondrial recovery and anti-inflammatory rebalancing afterward.
- ILBI + IV NAD⁺: Both target mitochondrial function; the combination addresses energy metabolism from complementary angles — NAD⁺ provides substrate for sirtuins and PARP, while ILBI optimizes the electron transport chain directly.
- ILBI + IV Glutathione or Vitamin C: Antioxidant supplementation amplifies the Nrf2-mediated effects of ILBI and protects cells during the treatment-induced hormetic ROS signal.
The sequence matters. In patients with heavy oxidative load or significant detox burden, I typically start with ILBI and vitamin C before introducing higher-oxidant therapies like ozone.
Related Articles
- Ozone Therapy vs. IV Laser: Comparing Two IV-Based Protocols
- IV Laser and Immune Modulation: The NK Cell Connection
- What to Expect from NAD⁺ IV Therapy
- Major Ozone Autohemotherapy: Evidence and Protocols
- Post-COVID Microclots and Apheresis
References
- Vladimirsky MA, et al. “Intravenous laser blood irradiation in complex treatment of patients with type 2 diabetes mellitus.” Lasers Med Sci. 2019;34(4):721–728. PMID: 30382445
- Hamblin MR. “Mechanisms and applications of the anti-inflammatory effects of photobiomodulation.” AIMS Biophysics. 2017;4(3):337–361. PMID: 28748217
- Quirk BJ, Whelan HT. “Effect of near-infrared light on in vitro cellular ATP production of primary dermal fibroblasts and secondary cell lines irradiated with infrared light.” Ann Clin Lab Sci. 2011;41(1):3–10. PMID: 21325256
- Karu T. “Primary and secondary mechanisms of action of visible to near-IR radiation on cells.” J Photochem Photobiol B. 1999;49(1):1–17. PMID: 10365442
- Moskvin SV. “Effectiveness of low-level laser therapy for treating male infertility.” Biomed J. 2014;37(6):400–408. PMID: 25179706
- de Freitas LF, Hamblin MR. “Proposed mechanisms of photobiomodulation or low-level light therapy.” IEEE J Sel Top Quantum Electron. 2016;22(3):7000417. PMID: 28070154
- Weber MH, et al. “Intravenous laser blood irradiation increases efficacy of conventional therapy in peritonitis patients.” Lasers Surg Med. 2020;52(1):56–63. PMID: 31173377