At a Glance
| Property | Value |
|---|---|
| Condition | Microclots and Endothelial Dysfunction |
| Evidence Level | Moderate (clinical studies available) |
| Causative Agent | SARS-CoV-2 |
| Key Symptoms | driven primarily by microclots — immune dysregulation, viral reactivation, and autonomic dysfunction contribute… |
| Treatment Approach | Targeted nutrients (L-arginine, vitamin C…, Addressing ongoing endothelial insults…, Physical activity (gentle, graduated |
| Key Takeaway | Fibrin amyloid microclots in post-COVID patients resist normal fibrinolysis, trap inflammatory molecules, and impair microcirculation — explaining fatigue… |
One of the most important advances in understanding post-COVID syndrome has been the identification of fibrin amyloid microclots and endothelial dysfunction as central mechanisms driving persistent symptoms. This research direction, to which Dr. Beate Jaeger has made significant contributions in collaboration with our clinical work, provides a mechanistic explanation for symptoms that were previously dismissed as subjective or psychosomatic.
I want to frame this appropriately. The microclot hypothesis is supported by a growing body of published research. It offers explanatory power for many post-COVID symptoms. It is an important research direction. It is not yet a fully validated, universally accepted model, and I am transparent about this distinction with my patients. But the clinical evidence I have observed is compelling enough that I consider it a cornerstone of my approach to post-COVID treatment.
What Microclots Are
In healthy individuals, blood coagulation and fibrinolysis exist in balance. Clots form when needed (to stop bleeding) and are dissolved (fibrinolysis) when their purpose is served. In post-COVID patients, this balance is disrupted.

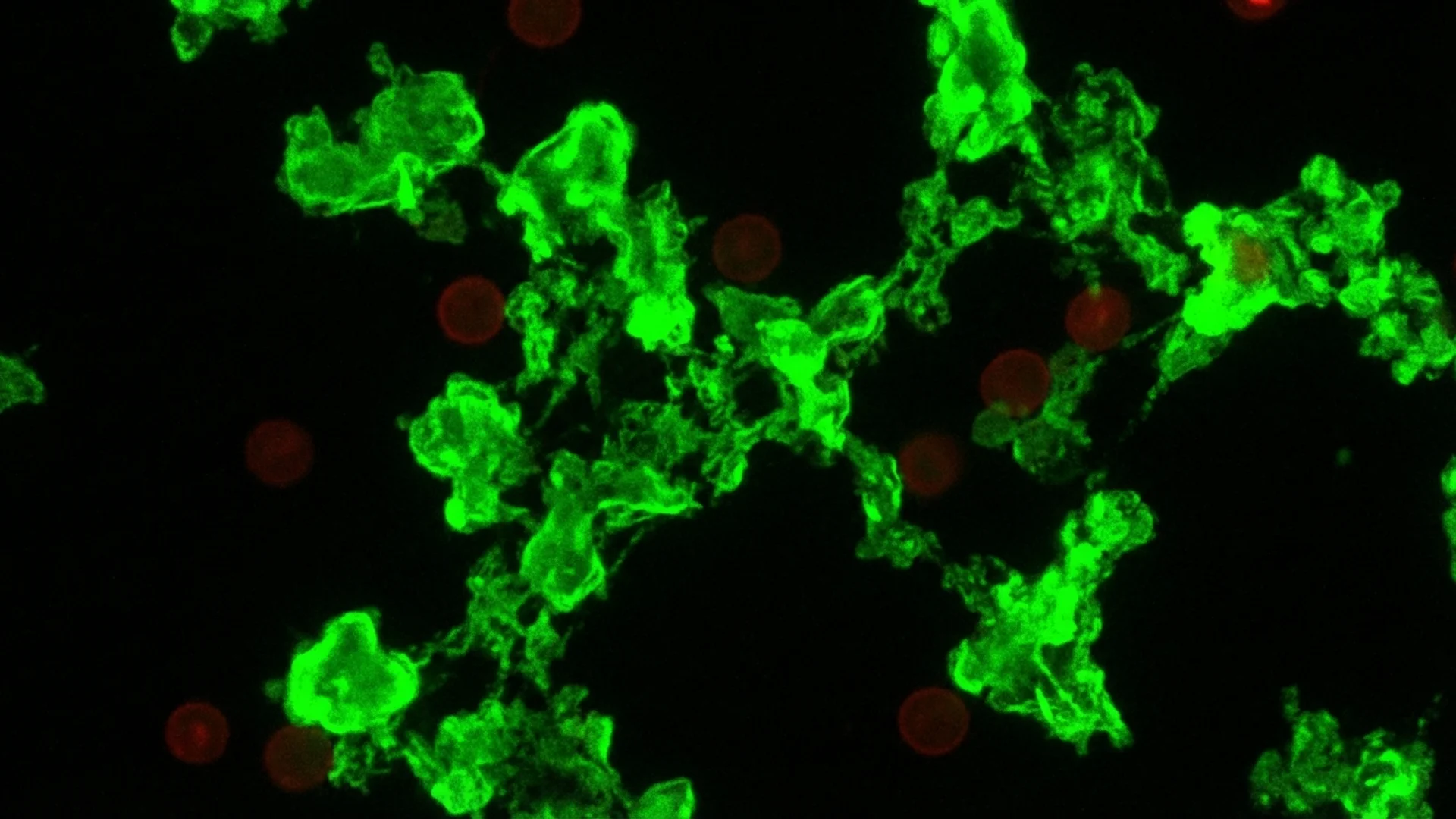
Researchers, including the groundbreaking work by Pretorius and colleagues in South Africa, have identified fibrin amyloid microclots in the blood of post-COVID patients [1]. These are not ordinary clots. They are characterized by:
- Amyloid fibrin: The fibrin in these microclots has an abnormal amyloid structure, making it resistant to the body’s normal fibrinolytic enzymes (plasmin). They do not dissolve on their own.
- Trapped inflammatory molecules: The microclots trap inflammatory cytokines, complement components, and other pro-inflammatory mediators, creating a persistent inflammatory reservoir.
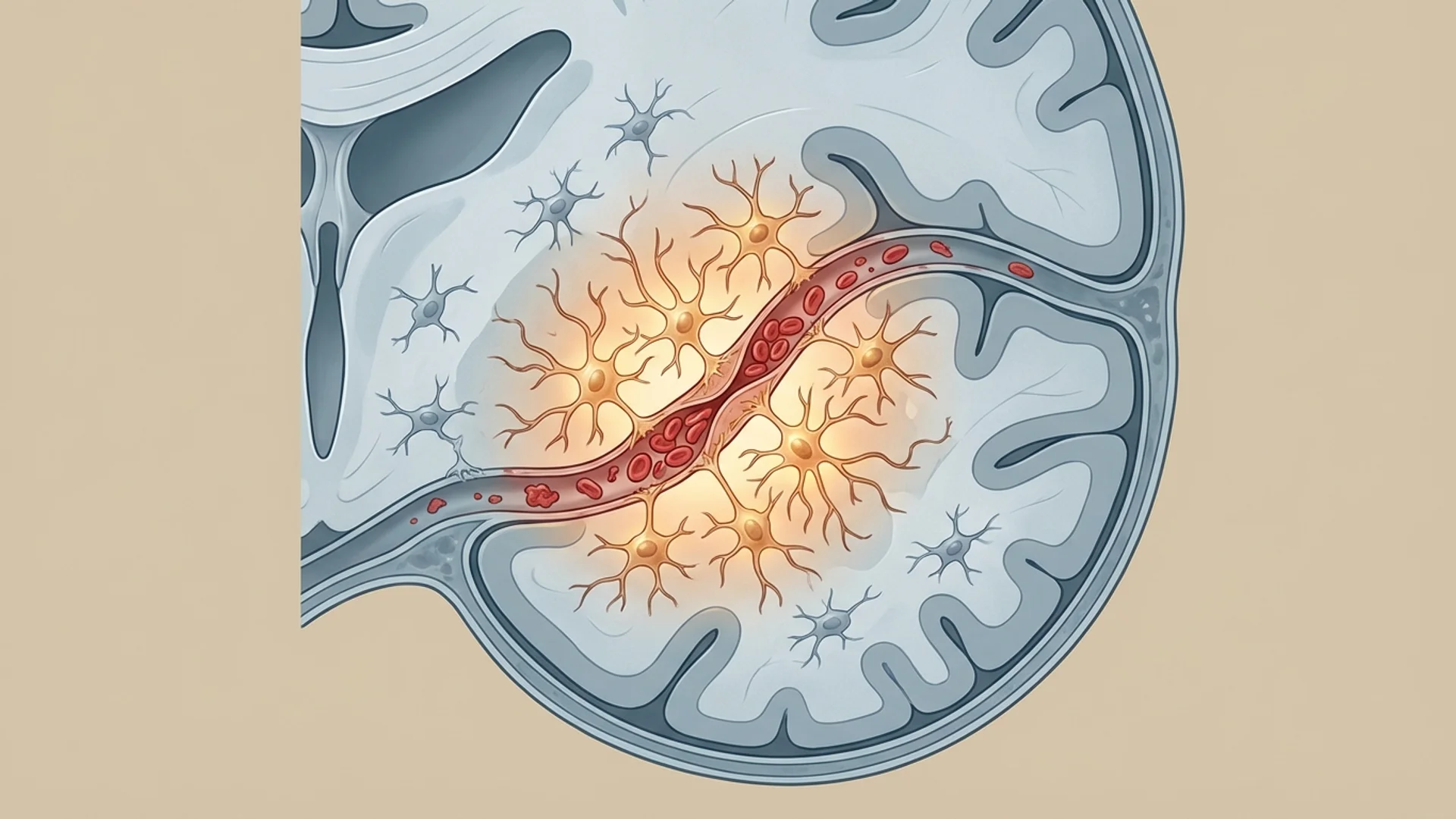
- Microscopic size: These clots are too small to cause strokes or pulmonary emboli, but they are large enough to impair flow through capillaries and small blood vessels — the microcirculation.
How Microclots Cause Symptoms
The implications of impaired microcirculation are broad. Every organ depends on capillary-level blood flow for oxygen and nutrient delivery. When microclots obstruct this flow:
Fatigue: Reduced oxygen delivery to muscles and other tissues produces fatigue disproportionate to activity level. Patients describe exhaustion after minimal exertion — a hallmark of post-COVID.
Cognitive dysfunction: The brain is exquisitely sensitive to microcirculatory impairment. Even modest reductions in capillary flow can produce the “brain fog” that post-COVID patients describe — difficulty concentrating, word-finding problems, slowed processing speed. I discuss this further in my brain fog article.
Exercise intolerance: Impaired microcirculation in muscle tissue reduces exercise capacity and prolongs recovery time. Patients who were previously fit find themselves unable to perform even moderate physical activity.
Autonomic dysfunction: Reduced microcirculatory perfusion may contribute to the autonomic dysregulation (POTS, blood pressure instability) commonly seen in post-COVID.
Endothelial Dysfunction
The endothelium — the single-cell layer lining all blood vessels — is not merely a passive barrier. It actively regulates vascular tone, coagulation, inflammation, and immune cell trafficking. SARS-CoV-2 directly infects endothelial cells via the ACE2 receptor, causing endothelial damage and dysfunction that can persist long after the acute infection resolves [2].
Damaged endothelium:
- Becomes pro-coagulant (promoting clot formation)
- Becomes pro-inflammatory (recruiting immune cells and releasing cytokines)
- Loses its normal vasodilatory function (impairing blood flow regulation)
- Increases vascular permeability (contributing to tissue edema)
Dr. Beate Jaeger’s research has been particularly important in characterizing the relationship between endothelial dysfunction, impaired microcirculation, and the clinical presentation of post-COVID. Her work suggests that endothelial dysfunction is not merely a consequence of the acute infection but an ongoing, self-perpetuating process that drives persistent symptoms.
This is an important research direction, and one that I believe will increasingly shape how the medical community understands and treats post-COVID syndrome.
Measuring Microcirculatory Impairment
Assessing microcirculation is not part of standard medical evaluation, which is one reason this mechanism has been overlooked. Tools that can identify microcirculatory problems include:
- Sublingual videomicroscopy: Direct visualization of capillary flow under the tongue. Impaired flow, reduced capillary density, and microthrombi can be observed in post-COVID patients.
- Fluorescence angiography: Assessment of perfusion patterns in the retinal or nail-fold capillary beds.
- D-dimer and fibrinogen: Elevated levels suggest ongoing coagulation activation, though normal levels do not exclude microclot pathology.
- Von Willebrand factor: Often elevated in endothelial dysfunction, serving as a marker of endothelial damage.
- Specialized microclot assays: Fluorescence microscopy of platelet-poor plasma with thioflavin T staining can identify amyloid microclots directly. This is not widely available but is used in research settings.
Treatment Implications
If microclots and endothelial dysfunction are central mechanisms in post-COVID, then treatment must address them directly. The approaches we use include:
Anticoagulation
Carefully managed anticoagulation to reduce ongoing microclot formation. This is not standard aspirin therapy — it requires individualized assessment of coagulation status and titration based on response. I use a combination of antiplatelet and anticoagulant agents, adjusted based on D-dimer, fibrinogen, and clinical response.
Apheresis
therapeutic apheresis — specifically, techniques that filter inflammatory mediators, autoantibodies, and microclots from the blood — has shown promise in post-COVID patients. I discuss the evidence and our clinical experience in my apheresis article.
Endothelial Support
Supporting endothelial repair and function through:
- Targeted nutrients (L-arginine, vitamin C, vitamin E, omega-3 fatty acids)
- Addressing ongoing endothelial insults (inflammation, oxidative stress)
- Physical activity (gentle, graduated — exercise is the strongest stimulus for endothelial health, but post-COVID patients need careful pacing)
Fibrinolytic Support
Enzymes that support fibrinolysis (nattokinase for fibrinolytic support, lumbrokinase) may assist in breaking down the fibrin component of microclots. These are used as adjuncts, not replacements for medical anticoagulation, and their efficacy in this specific context is based on mechanistic rationale and clinical observation rather than controlled trials.
The Honest Assessment
The microclot hypothesis explains much of what we observe clinically in post-COVID patients. It provides a mechanistic bridge between the acute viral insult and the chronic symptoms. And it suggests specific, testable treatment strategies.
However, I want to be transparent about the limitations. Large, randomized controlled trials of microclot-targeted treatments for post-COVID have not been completed. The diagnostic tools for assessing microclots are not widely standardized. And not every post-COVID patient’s symptoms are driven primarily by microclots — immune dysregulation, viral reactivation, and autonomic dysfunction contribute independently.
What I can say from clinical experience is that patients whose workup suggests significant microcirculatory impairment and who receive targeted treatment often improve substantially. This is consistent enough across hundreds of patients that I am confident in the approach, even while acknowledging that the evidence base continues to develop.

References
- Pretorius E, et al. Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovascular Diabetology. 2021;20(1):172.
- Varga Z, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417-1418.
This content is educational and does not constitute medical advice. Post-COVID treatment, particularly anticoagulation therapy, requires careful medical supervision and individualized assessment.