At a Glance
| Property | Value |
|---|---|
| Topic | IV Laser Therapy and Immune Function |
| Evidence Level | Moderate (clinical studies available) |
| Primary Function | Intravenous laser blood irradiation delivers specific light wavelengths directly to circulating blood through a fiber-optic catheter. |
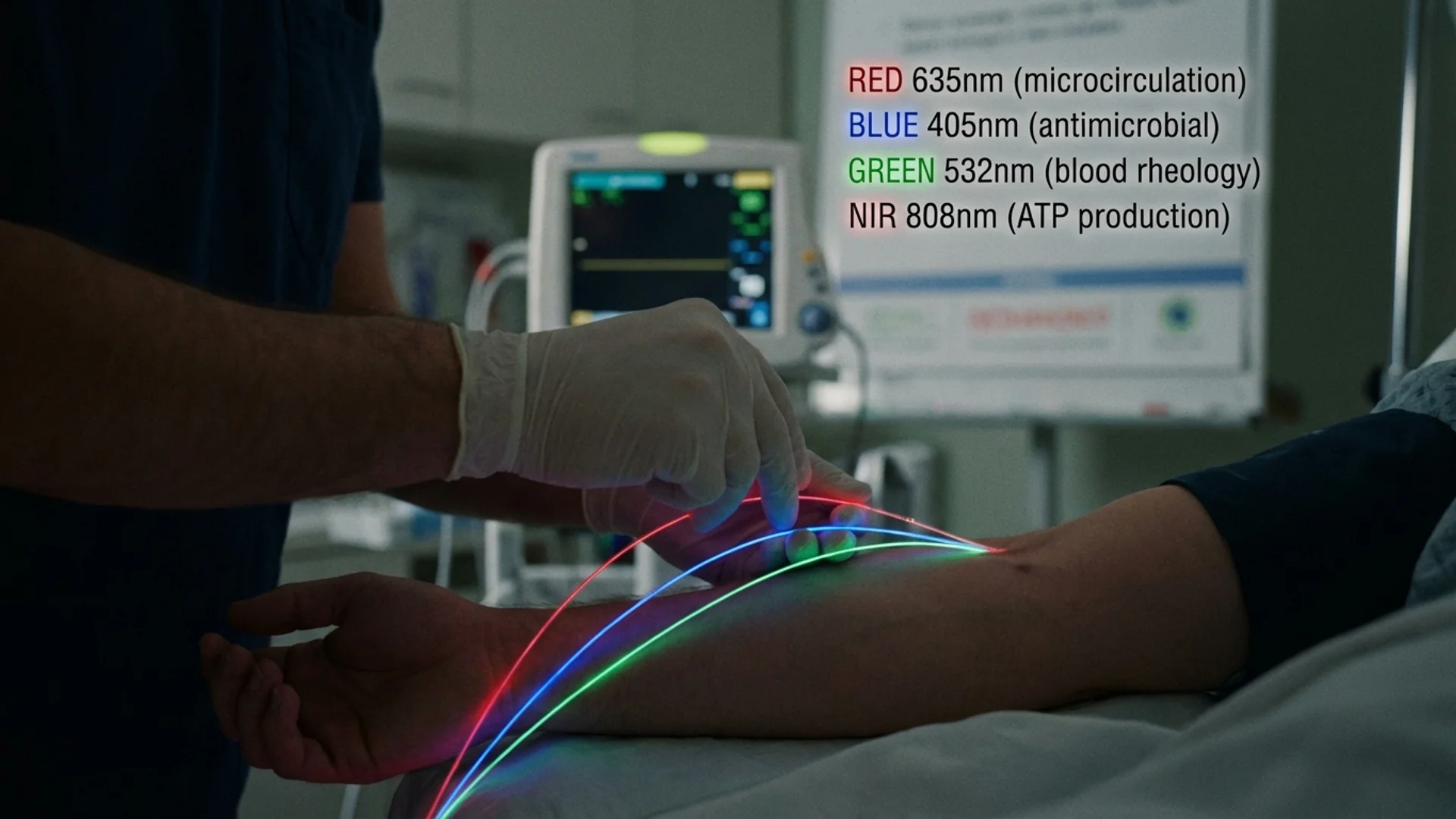
Intravenous laser blood irradiation (ILIB) involves the insertion of a thin fiber-optic catheter into a peripheral vein, through which laser light of specific wavelengths is delivered directly to circulating blood. The therapy has been used in Germany and Eastern Europe for over 30 years, and its effects on immune cell function are among the most interesting — and least widely known — aspects of photobiomodulation.
I want to set expectations clearly: this is an area where mechanistic understanding is strong, clinical observation is extensive, but large-scale randomized controlled trials remain limited. The evidence hierarchy matters here, and I will distinguish between what is well-established and what is observed but not yet confirmed by RCTs.
The Mechanism: Photobiomodulation
All cells contain photoreceptive molecules — chromophores — that absorb light at specific wavelengths. The primary chromophore in mammalian cells is cytochrome c oxidase, the terminal enzyme in the mitochondrial electron transport chain. When this enzyme absorbs light in the red and near-infrared spectrum, it releases nitric oxide (which was inhibiting the enzyme), increasing mitochondrial electron transport, ATP production, and reactive oxygen species (ROS) signaling.
This is not alternative medicine speculation. The mechanism of photobiomodulation through cytochrome c oxidase has been characterized in peer-reviewed publications in journals including Photochemistry and Photobiology, PNAS, and Lasers in Surgery and Medicine. Evidence level for the basic mechanism: strong (in vitro and in vivo studies).
The clinical question is whether these cellular effects translate to meaningful clinical outcomes when applied through intravenous laser irradiation of circulating blood.
Wavelength-Specific Effects
Different wavelengths of light penetrate different depths, are absorbed by different chromophores, and produce different biological effects. In IV laser therapy, the most commonly used wavelengths are:
Red (630-635 nm)
Red light is the most studied wavelength in ILIB. Its primary effects include:
- Enhanced microcirculation — red light promotes the release of nitric oxide from hemoglobin, improving blood flow and oxygen delivery. This has been documented using laser Doppler flowmetry in controlled studies.
- Improved red blood cell deformability — red light reduces aggregation and improves the flexibility of red blood cells, facilitating their passage through capillaries. Evidence level: controlled studies.
- immune modulation strategies — in vitro and clinical studies suggest red light increases phagocytic activity of monocytes and macrophages, and may promote lymphocyte proliferation. Evidence level: controlled studies and clinical observation.
Blue (405 nm)
Blue light has distinct photochemical properties due to its absorption by porphyrins and flavins:
- Antimicrobial effects — blue light activates endogenous porphyrins in bacteria, generating singlet oxygen that destroys microbial cells. This has been well-documented in vitro and is the basis for photodynamic antimicrobial therapy. Evidence level: strong in vitro data; clinical application is emerging.
- Anti-inflammatory effects — blue light has been shown to reduce pro-inflammatory cytokine production (TNF-alpha, IL-1beta) in activated monocytes. Evidence level: in vitro studies with preliminary clinical data.
Green (532 nm)
Green light is absorbed by hemoglobin and has effects on:
- Blood rheology — green light may improve blood viscosity and reduce platelet aggregation. Evidence level: controlled studies (limited).
- Mood and circadian regulation — while this relates to transcranial rather than IV application, melanopsin-containing retinal ganglion cells are maximally sensitive to green light, with implications for circadian rhythm regulation.
Yellow (589 nm)
Yellow light has the deepest penetration into blood and is absorbed by bilirubin and flavins:
- Serotonin metabolism — yellow light may influence serotonin synthesis, though the evidence for this effect via IV application (as opposed to transcutaneous) is preliminary.
- Detoxification support — yellow light activates cytochrome P450 enzymes involved in hepatic metabolism. Evidence level: mechanistic data with limited clinical confirmation for the IV application route.
Near-Infrared (808 nm)
Near-infrared light has the deepest tissue penetration and the strongest effect on mitochondrial function:
- Mitochondrial activation — near-infrared is the optimal wavelength for cytochrome c oxidase activation, with the most robust evidence for increased ATP production. Evidence level: strong in vitro and in vivo data.
- Neuroprotective effects — transcranial photobiomodulation at this wavelength has RCT evidence for traumatic brain injury and is under investigation for neurodegenerative disease. The relevance to IV application specifically requires further study.
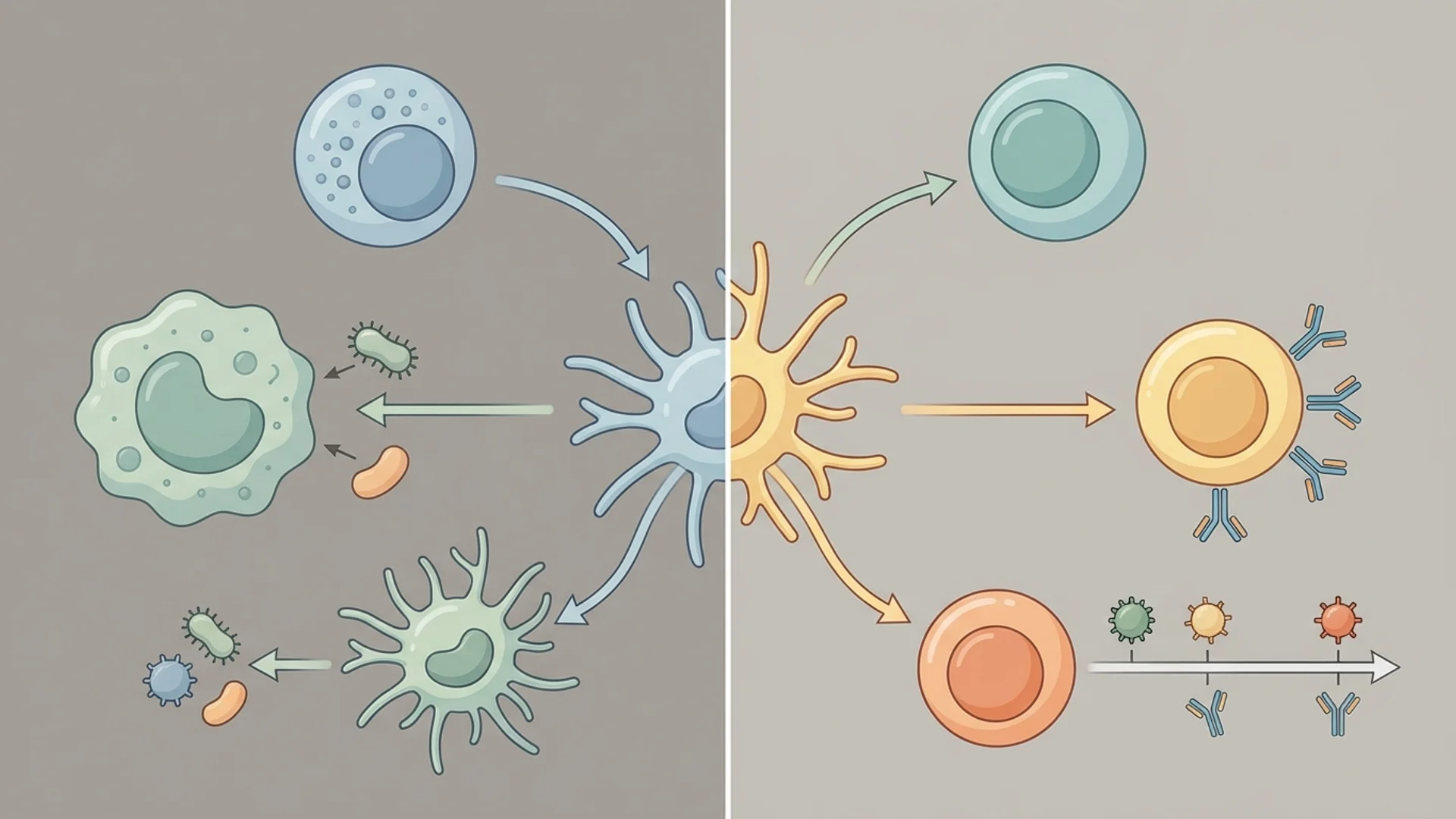
Immune-Specific Effects
The immune effects of IV laser therapy that are most consistently reported across the literature include:
NK cell activation. Several studies have documented increases in NK cell activity following IV laser therapy. In a clinical study by Weber (2007), red and near-infrared ILIB increased NK cell activity in patients with chronic fatigue. Evidence level: controlled clinical studies.
Cytokine modulation. ILIB has been shown to modulate cytokine production — generally reducing pro-inflammatory cytokines in patients with elevated baseline levels while supporting immune activation in immunodeficient patients. This bidirectional effect is consistent with immune modulation rather than simple stimulation. Evidence level: controlled studies.
Lymphocyte proliferation. In vitro studies demonstrate that low-level laser irradiation enhances lymphocyte proliferation in response to mitogens. The clinical translation of this finding is supported by observation of improved lymphocyte counts in immunosuppressed patients, though RCTs specifically addressing this endpoint are limited.
Improved phagocytosis. Monocytes and macrophages exposed to photobiomodulation show enhanced phagocytic capacity. This has been documented in both in vitro studies and clinical measurements of monocyte function after ILIB sessions.
Clinical Application at Klinik St. Georg
In our practice, IV laser therapy is used as a component of comprehensive treatment protocols, not as a standalone intervention. Common applications include. In oxidative medicine protocols, IV laser is frequently combined with major autohemotherapy ozone therapy — each modality targets overlapping but distinct photobiomodulatory and redox pathways, and the combination produces more consistent immune activation than either treatment alone in our clinical observation. For a detailed side-by-side breakdown of mechanisms, indications, and when to sequence both, see Ozone vs. IV Laser Therapy: A Physician’s Comparison.
Common applications include:
- chronic Lyme disease treatment disease — as part of a multi-modal approach including antimicrobials, hyperthermia, and immune support. The rationale is improved microcirculation (enhancing antibiotic delivery to poorly perfused tissues) and immune modulation.
- post-COVID syndrome — addressing endothelial dysfunction, impaired microcirculation, and immune dysregulation.
- Oncology support — as an adjunct to enhance immune surveillance and mitigate treatment-related immunosuppression.
- Chronic fatigue — targeting mitochondrial function through near-infrared activation of cytochrome c oxidase.
A typical treatment series involves 10 sessions over two to three weeks, with each session lasting 30-60 minutes. Wavelengths are selected based on clinical indication.
The Honest Assessment
IV laser therapy is a modality with strong mechanistic rationale, substantial clinical experience (particularly in German and Eastern European medicine), and growing but still limited controlled trial evidence. It is not a treatment I would recommend in isolation for any condition, and I would not present it as supported by the same level of evidence as, for example, IV vitamin C in sepsis or hyperthermia in oncology. For clinicians and patients seeking a complete orientation to the therapy — covering wavelength selection, treatment protocols, ideal patient profiles, and combination strategies — the IV Laser Therapy: A Physician’s Complete Guide provides that broader framework.
What I observe clinically is that patients who receive IV laser therapy as part of a comprehensive protocol tend to report improvements in energy, pain, and overall well-being — and that these improvements correlate with measurable changes in inflammatory markers and immune cell function. Whether this is attributable specifically to the laser therapy or to the combined protocol is difficult to isolate without controlled trials.
This is an area where continued research is warranted and where the clinical experience of physicians using these tools should be documented more rigorously.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical care. IV laser therapy should be administered by trained medical professionals in a clinical setting.
Related Reading
- Innate vs. Adaptive Immunity Explained
- Mistletoe Therapy (Iscador): Evidence in Integrative Oncology
References
References are cited inline throughout the article. A compiled reference list for this article is being prepared.