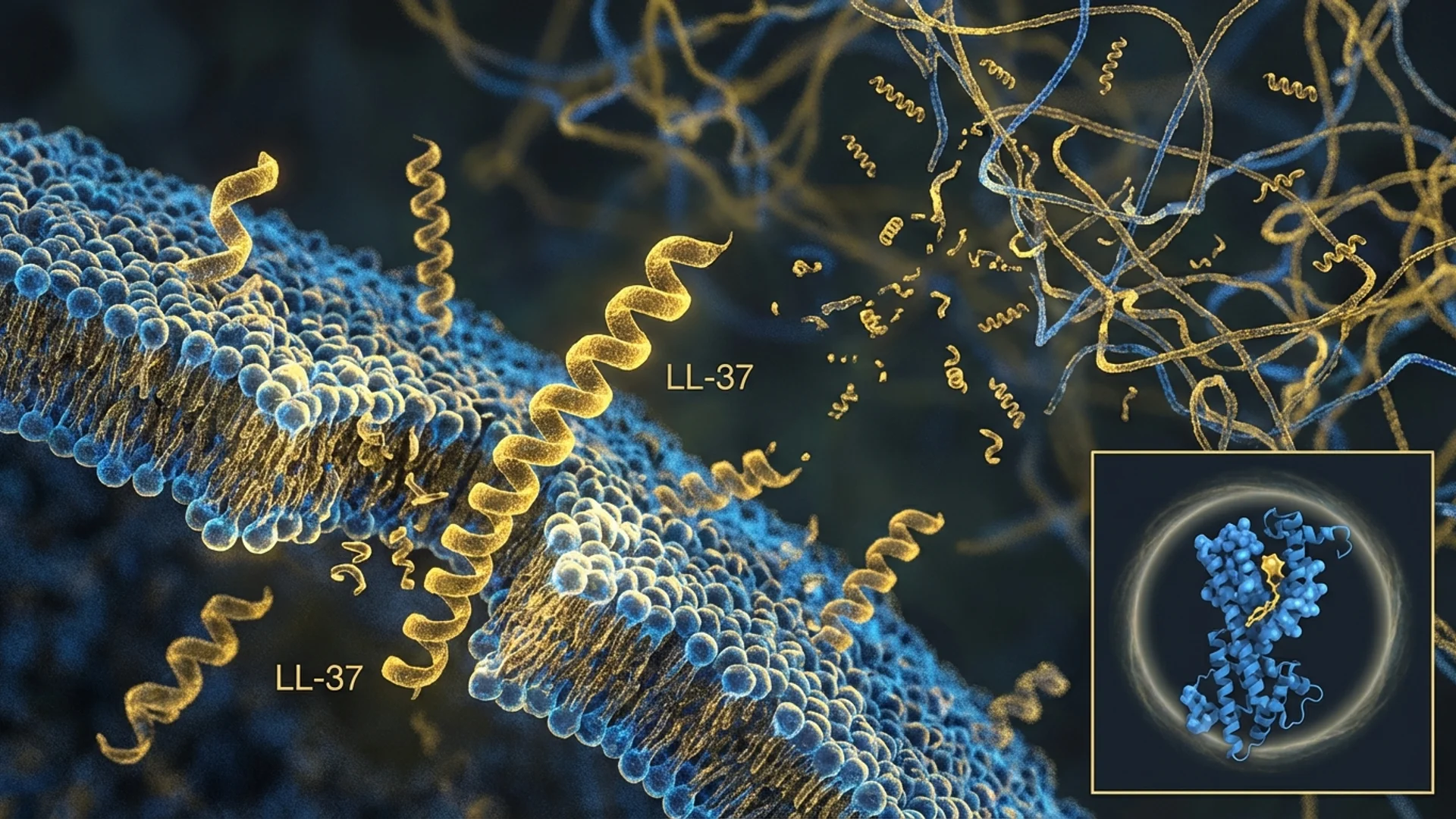
At a Glance
| Property | Detail |
|---|---|
| Evidence Level | Strong — well-characterized molecular pathway with multiple confirming studies |
| Key Discovery | Vitamin D induces LL-37 expression via vitamin D receptor (VDR) activation |
| Optimal Vitamin D Level | 50-80 ng/mL (125-200 nmol/L) for immune optimization |
| Key Pathway | Vitamin D → VDR activation → hCAP18 transcription → LL-37 cleavage |
| Clinical Relevance | Infections, autoimmune disease, chronic Lyme, respiratory immunity |
| First Demonstrated | Liu et al., 2006 — tuberculosis macrophage response |
Why Does Vitamin D Affect Your Immune System?
For decades, the medical establishment treated vitamin D as a bone health nutrient — important for calcium absorption and preventing rickets, but not much else. This was wrong. Spectacularly wrong.
The discovery that vitamin D directly regulates the expression of antimicrobial peptides — particularly LL-37 — fundamentally changed our understanding of how the immune system works. It explained why vitamin D deficiency is associated with increased susceptibility to infections, why infections like tuberculosis are more severe in people with low vitamin D, and why seasonal variations in vitamin D correlate with seasonal patterns of respiratory illness.
This is not speculative biology. This is a well-characterized molecular pathway confirmed by multiple independent research groups. Here is what the evidence shows.
The Vitamin D-LL-37 Pathway
The Molecular Mechanism
The connection between vitamin D and LL-37 is direct and well-characterized:
-
Vitamin D3 (cholecalciferol) is converted to its active form, 1,25-dihydroxyvitamin D3 (calcitriol), by the enzyme 1-alpha-hydroxylase.
-
Calcitriol binds to the vitamin D receptor (VDR), a nuclear receptor present in virtually all immune cells — macrophages, dendritic cells, T-cells, B-cells, and epithelial cells.
-
The activated VDR forms a complex with retinoid X receptor (RXR) and binds to a vitamin D response element (VDRE) in the promoter region of the CAMP gene (cathelicidin antimicrobial peptide gene).
-
This transcriptional activation induces expression of hCAP18, the precursor protein of LL-37.
-
hCAP18 is cleaved by proteinase 3 to produce the active 37-amino-acid antimicrobial peptide LL-37.
The entire pathway — from vitamin D to functional antimicrobial peptide — is dependent on adequate vitamin D levels. When vitamin D is insufficient, the VDR-mediated transcription of hCAP18 is impaired, and LL-37 production drops.
The Tuberculosis Discovery
The pivotal study was published by Liu et al. in 2006 in Science. The researchers investigated why African Americans — who have a higher prevalence of vitamin D deficiency due to melanin-mediated reduction in cutaneous vitamin D synthesis — are more susceptible to tuberculosis.
They discovered that when macrophages encounter Mycobacterium tuberculosis, they upregulate expression of both the vitamin D receptor and the 1-alpha-hydroxylase enzyme. This creates a local amplification loop: the immune cell, upon detecting the pathogen, enhances its own ability to respond to vitamin D. The resulting vitamin D-mediated induction of LL-37 was essential for killing the intracellular mycobacteria.
The critical finding: macrophages from vitamin D-deficient individuals could not mount this LL-37 response effectively. Adding vitamin D to the culture restored it. This was the smoking gun linking vitamin D deficiency to impaired antimicrobial defense through the LL-37 pathway.
Clinical Implications
Respiratory Infections
The vitamin D-LL-37 axis is particularly relevant to respiratory immunity because LL-37 is produced by airway epithelial cells and is present in airway surface liquid — the frontline defense against inhaled pathogens.
Multiple studies have confirmed the relationship:
| Study | Finding |
|---|---|
| Jolliffe et al., 2021 (BMJ meta-analysis) | Vitamin D supplementation reduced risk of acute respiratory infections by 12% overall and 19% in deficient individuals |
| Martineau et al., 2017 (individual patient data meta-analysis, 25 RCTs) | Vitamin D supplementation protected against acute respiratory tract infection, with greatest benefit in those with lowest baseline levels |
| Berry et al., 2011 | Low vitamin D levels associated with increased frequency and severity of respiratory infections |
Chronic Lyme Disease
This connection is directly relevant to the Lyme patient population. Many chronic Lyme patients have suboptimal vitamin D levels, which may compound their immune impairment:
- Impaired LL-37 production — reduced ability to disrupt Borrelia biofilms and kill planktonic bacteria
- Reduced innate immune activation — vitamin D is required for optimal macrophage and dendritic cell function
- Impaired T-cell function — vitamin D modulates both regulatory and effector T-cell activity
In my clinical practice, vitamin D optimization is one of the first interventions in any chronic Lyme protocol. It is simple, safe, inexpensive, and supported by strong evidence.
For more on LL-37’s role in Lyme disease, see LL-37 for Biofilm Disruption in Chronic Lyme.
Autoimmune Disease
The relationship between vitamin D, LL-37, and autoimmunity is more complex. LL-37 has been implicated in the pathogenesis of certain autoimmune conditions — it can form complexes with self-DNA and self-RNA that activate plasmacytoid dendritic cells through TLR9 and TLR7, driving type I interferon production. This mechanism has been demonstrated in psoriasis and systemic lupus erythematosus (SLE).
However, vitamin D itself is broadly immunoregulatory and generally suppresses autoimmune activity. The paradox — vitamin D increases LL-37, but LL-37 can contribute to autoimmunity while vitamin D suppresses it — has not been fully resolved. The current understanding is that vitamin D’s overall immunoregulatory effects are net-positive even in autoimmune conditions, and that the LL-37-mediated autoimmune mechanisms occur primarily when LL-37 encounters specific triggers (damaged tissue releasing self-nucleic acids) rather than from circulating LL-37 alone.
Optimal Vitamin D Levels for LL-37 Production
Standard vs. Optimal
| Level (25-OH-D) | Classification | LL-37 Implication |
|---|---|---|
| Below 20 ng/mL | Deficient | Significantly impaired LL-37 induction |
| 20-30 ng/mL | Insufficient | Suboptimal LL-37 production |
| 30-50 ng/mL | Sufficient (standard guidelines) | Adequate baseline LL-37 production |
| 50-80 ng/mL | Optimal (immune-focused) | Robust LL-37 response; target range for immune optimization |
| Above 100 ng/mL | Potentially excessive | Risk of hypercalcemia; no additional LL-37 benefit demonstrated |
The standard medical definition of “sufficient” vitamin D (30 ng/mL) was established primarily for bone health. The evidence suggests that immune-related functions — including LL-37 production — may require higher levels. In vitro studies show that LL-37 induction increases in a dose-dependent manner with vitamin D concentration up to approximately 50-80 ng/mL, above which the response plateaus.
What I Target in Practice
For patients whose primary concern is immune optimization — chronic infections, recurrent infections, chronic Lyme, autoimmune conditions — I target serum 25-hydroxyvitamin D levels of 50-80 ng/mL. This is above the standard “sufficient” threshold but well below the level at which toxicity occurs.
Vitamin D Supplementation Protocol
Getting to Optimal Levels
| Starting Level | Loading Dose | Maintenance Dose | Recheck |
|---|---|---|---|
| Below 20 ng/mL | 10,000 IU daily for 8-12 weeks | 5,000-7,000 IU daily | At 12 weeks |
| 20-30 ng/mL | 7,000 IU daily for 8 weeks | 4,000-5,000 IU daily | At 8 weeks |
| 30-50 ng/mL | 5,000 IU daily | 3,000-5,000 IU daily | At 8-12 weeks |
| 50-80 ng/mL | Maintenance | 2,000-4,000 IU daily | Every 6 months |
Critical Co-Factors
Vitamin D does not work in isolation. For optimal vitamin D metabolism and LL-37 production, the following co-factors are important:
| Co-Factor | Role | Recommendation |
|---|---|---|
| Vitamin K2 (MK-7) | Directs calcium to bones, prevents arterial calcification | 100-200 mcg daily with vitamin D |
| Magnesium | Required for vitamin D activation (enzyme cofactor) | 400-600 mg daily (glycinate or malate) |
| Zinc | Supports immune function; works synergistically with vitamin D | 15-30 mg daily |
| Vitamin A (retinol) | VDR function requires adequate retinol; RXR is a retinoid receptor | Ensure adequate dietary intake; supplement only if deficient |
Vitamin D3 vs. D2
Use vitamin D3 (cholecalciferol), not D2 (ergocalciferol). D3 is more effective at raising and maintaining serum 25-hydroxyvitamin D levels, has a longer half-life, and is the form naturally produced by human skin in response to UV-B radiation.
Synergy: Vitamin D + Exogenous LL-37
For patients using exogenous LL-37 (subcutaneous injection), optimizing vitamin D levels is not optional — it is foundational. The rationale:
-
Endogenous amplification. Adequate vitamin D ensures your body’s own LL-37 production is maximized. Exogenous LL-37 supplementation addresses the gap between what your body produces and what is needed therapeutically.
-
Immune priming. Vitamin D primes immune cells — macrophages, dendritic cells, T-cells — to respond more effectively. LL-37 then provides direct antimicrobial activity and biofilm disruption. The combination is synergistic.
-
Sustained effect. Exogenous LL-37 has a limited half-life. Vitamin D-driven endogenous production provides a continuous baseline of LL-37 between exogenous doses.
In practice, I ensure vitamin D levels are optimized before or concurrently with initiating exogenous LL-37 therapy. Administering LL-37 to a vitamin D-deficient patient is like refueling one engine of a two-engine airplane — it helps, but it is not the full solution.
Beyond Infections: The Broader Vitamin D-LL-37 Connection
Cancer
LL-37 has demonstrated anti-tumor activity in several models, and vitamin D deficiency is associated with increased risk of several cancers. The vitamin D-LL-37 axis may represent one mechanism through which vitamin D exerts its observed anti-cancer effects — through LL-37-mediated immune surveillance.
Wound Healing
Vitamin D and LL-37 are both involved in wound healing. LL-37 promotes keratinocyte migration and angiogenesis at wound sites. Vitamin D deficiency impairs wound healing in clinical studies. The pathway is clear: adequate vitamin D ensures adequate LL-37 at wound sites.
Mental Health
Emerging research links vitamin D deficiency to depression and cognitive decline. LL-37 has neuroprotective properties. Whether the vitamin D-LL-37 axis contributes to the neurological effects of vitamin D deficiency is speculative but mechanistically plausible.
The Bottom Line
Vitamin D directly and dose-dependently regulates LL-37 production through a well-characterized molecular pathway. This connection explains much of vitamin D’s observed immune-protective effects and has direct clinical implications for infections, chronic Lyme disease, and immune optimization. Targeting serum vitamin D levels of 50-80 ng/mL — above standard sufficiency thresholds but well within the safe range — optimizes LL-37 production. Co-supplementation with vitamin K2, magnesium, and zinc supports the pathway. For patients using exogenous LL-37, vitamin D optimization is a foundational, not optional, component of the protocol.
For LL-37’s role in biofilm disruption, see LL-37 for Biofilm Disruption in Chronic Lyme. For the full overview, see LL-37: The Body’s Antimicrobial Peptide.
References
- Liu PT, et al. “Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response.” Science. 2006;311(5768):1770-1773. PMID: 16497887.
- Wang TT, et al. “Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression.” J Immunol. 2004;173(5):2909-2912. PMID: 15322146.
- Martineau AR, et al. “Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data.” BMJ. 2017;356:i6583. PMID: 28202713.
- Gombart AF, et al. “Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D3.” FASEB J. 2005;19(9):1067-1077. PMID: 15985530.
- Jolliffe DA, et al. “Vitamin D supplementation to prevent acute respiratory infections: a systematic review and meta-analysis of aggregate data from randomised controlled trials.” Lancet Diabetes Endocrinol. 2021;9(5):276-292. PMID: 33798465.
Disclaimer: This article is for educational purposes and reflects current published research and clinical observation. It is not medical advice. Vitamin D supplementation should be guided by serum level testing and physician oversight. Consult a qualified physician before pursuing any supplementation or peptide therapy.