At a Glance
| Property | Value |
|---|---|
| Evidence Level | Moderate |
| Primary Use | Restoring autonomic balance and reducing inflammation in ME/CFS and POTS |
| Key Mechanism | Activation of the cholinergic anti-inflammatory pathway via vagus nerve afferent signaling, restoring sympathovagal balance |
The Nerve That Controls Your Energy
If you have chronic fatigue — whether from Lyme disease, post-COVID, ME/CFS, or an undefined chronic illness — you have probably heard that your autonomic nervous system is dysregulated. Sympathetic overdrive. Parasympathetic underactivity. Poor heart rate variability. These are the clinical terms.
But here is what those terms actually mean: the nerve that should be telling your body to rest, recover, digest, and heal is not functioning properly. That nerve is the vagus nerve — the longest cranial nerve in your body, running from your brainstem to your gut, touching nearly every major organ along the way.
And there is growing evidence that stimulating this nerve can meaningfully improve fatigue, autonomic dysfunction, and the chronic inflammation that drives both.
The Vagus Nerve and the Autonomic Problem
Normal Function
The vagus nerve (cranial nerve X) is the primary parasympathetic pathway in the body. It regulates:
- Heart rate (slows it)
- Respiratory rate (calms it)
- Digestion (activates it)
- Inflammatory response (modulates it)
- Stress response (counterbalances sympathetic activation)
Healthy autonomic function is a balance between sympathetic (“fight or flight”) and parasympathetic (“rest and digest”) activity. The vagus nerve is the brake pedal. Sympathetic activation is the gas pedal.
What Goes Wrong in Chronic Fatigue and POTS
In ME/CFS, POTS (Postural Orthostatic Tachycardia Syndrome), and chronic infection-related fatigue, this balance is disrupted:
Sympathetic overdrive: The sympathetic system is chronically activated — heart rate is elevated, blood pressure is labile, cortisol is dysregulated, and the body is in a constant state of low-level stress response.
Vagal underactivity: The vagus nerve is not providing adequate parasympathetic counter-regulation. Heart rate variability (HRV) is low — one of the most consistent biomarkers of autonomic dysfunction in chronic fatigue [1].
Impaired inflammation control: The vagus nerve controls the cholinergic anti-inflammatory pathway — a neural circuit that modulates TNF-alpha, IL-6, and other inflammatory cytokines. When vagal tone is low, this inflammatory brake is released, and chronic low-grade inflammation persists.
This creates a self-reinforcing cycle: low vagal tone → poor inflammation control → more inflammation → more sympathetic activation → lower vagal tone.
The POTS Connection
POTS is a clinical manifestation of this autonomic imbalance. Upon standing, the heart rate increases excessively (30+ bpm above baseline or to >120 bpm within 10 minutes) because the cardiovascular system lacks adequate vagal counter-regulation and baroreceptor sensitivity is impaired.
In our clinical experience, POTS is extremely common in chronic Lyme patients — present in 30-40% of our population — and it directly contributes to both fatigue and brain fog through impaired cerebral perfusion during upright posture.
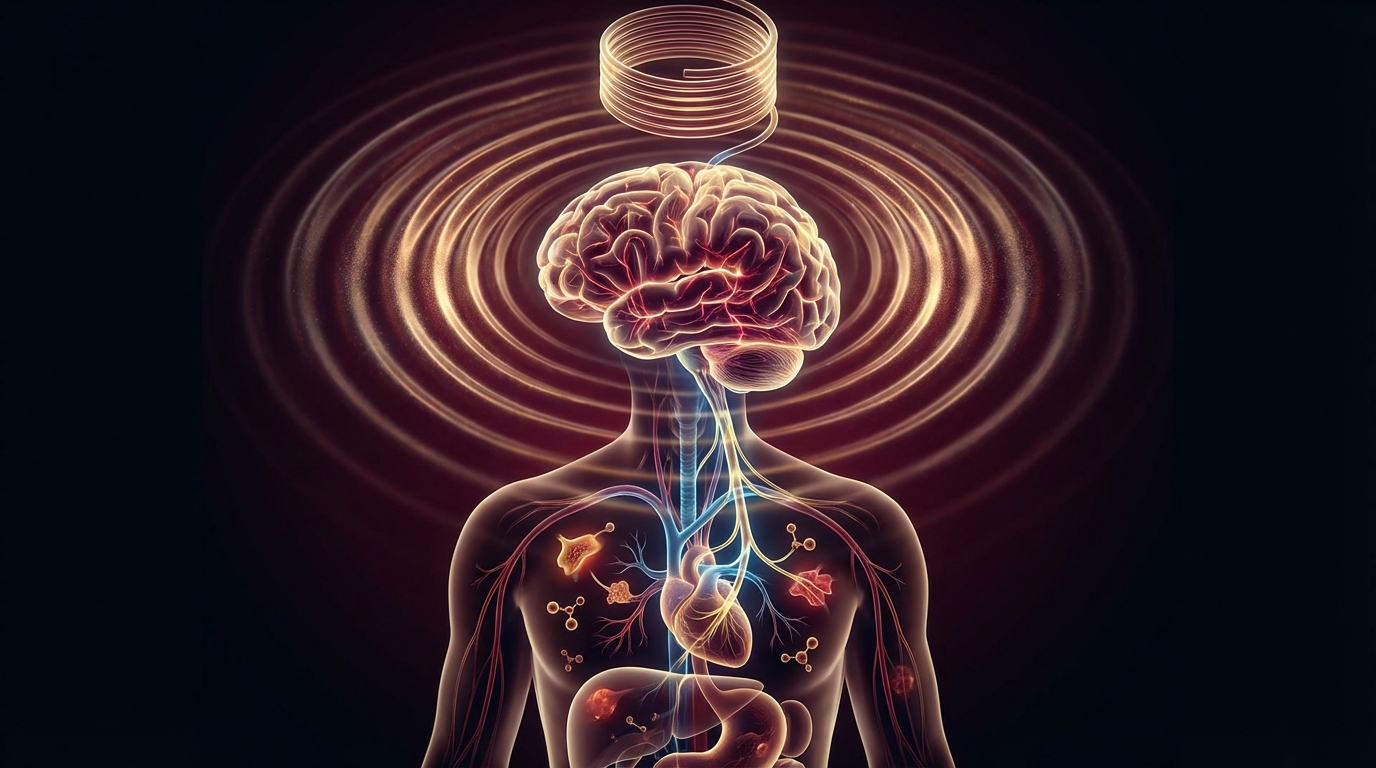
The Cholinergic Anti-Inflammatory Pathway
This is the mechanism that makes vagus nerve stimulation relevant to chronic fatigue and inflammatory illness:
- Vagus nerve afferents detect peripheral inflammation (via cytokine sensing)
- Signal travels to the brainstem (nucleus tractus solitarius)
- Efferent vagal signal descends to the spleen via the splenic nerve
- Acetylcholine is released onto macrophages expressing alpha-7 nicotinic receptors
- Alpha-7 receptor activation suppresses NF-kB signaling in macrophages
- TNF-alpha, IL-1, and IL-6 production is reduced [2]
This is not subtle modulation — in animal models, vagus nerve stimulation reduces TNF-alpha production by up to 75%. The clinical translation: stimulating the vagus nerve directly reduces the inflammatory signaling that drives fatigue.

The Evidence
What We Know (Human Data)
Vagus nerve stimulation for fatigue: A pilot study at Johns Hopkins demonstrated that transcutaneous auricular VNS (taVNS) — non-invasive stimulation of the vagus nerve branch in the ear — reduced fatigue severity by approximately 30% in women with POTS over an 8-week treatment period [3].
Multiple studies in rheumatoid arthritis (another inflammatory fatigue condition) have shown that VNS reduces both inflammatory markers and fatigue severity. The landmark study by Koopman et al. using an implanted VNS device demonstrated sustained TNF-alpha reduction and clinical improvement in RA patients who had failed standard therapy [4].
Vagus nerve stimulation for POTS: A Johns Hopkins study using taVNS in POTS patients showed improvements in orthostatic tachycardia, symptom burden, and quality of life. The mechanism appears to involve improved baroreceptor sensitivity and sympathovagal rebalancing.
Heart rate variability as a biomarker: HRV is consistently reduced in ME/CFS and POTS patients. Interventions that increase vagal tone (VNS, breathing exercises, exercise) consistently improve HRV, and HRV improvement correlates with symptom improvement.
VNS for Long COVID: A randomized controlled trial published in Bioelectronic Medicine demonstrated that taVNS improved fatigue, brain fog, and autonomic symptoms in Long COVID patients — a population with significant clinical overlap with chronic Lyme fatigue.
What I See in Practice
In our clinical experience, vagus nerve interventions are among the most impactful additions to chronic fatigue treatment protocols. What I observe:
1. HRV improvement is measurable within weeks. Patients using taVNS devices or implementing structured vagal exercises show measurable HRV improvement within 2-4 weeks — one of the earliest objective improvements we can track.
2. Fatigue improvement follows HRV improvement. Clinical fatigue severity typically begins to improve 4-8 weeks after vagal tone begins improving. The autonomic normalization appears to precede the symptomatic improvement.
3. Patients with the lowest baseline HRV benefit most. This makes physiological sense — those with the most autonomic dysfunction have the most to gain from vagal activation.
4. Combining device-based VNS with vagal exercises produces better results than either approach alone. The device provides consistent, calibrated stimulation; the exercises build intrinsic vagal tone over time.
Practical Application
Device-Based Vagus Nerve Stimulation
Transcutaneous auricular VNS (taVNS):
- Non-invasive device clips to the ear (tragus or cymba conchae — where the auricular branch of the vagus nerve is accessible)
- FDA-cleared devices available (gammaCore for migraine/cluster headache; other devices available in EU/Germany)
- Typical protocol: 20-30 minutes, twice daily
- Well-tolerated; mild tingling at stimulation site
Transcutaneous cervical VNS:
- gammaCore device (FDA-cleared)
- Applied to the neck over the vagus nerve
- Originally approved for migraine and cluster headache
- Off-label use for autonomic dysfunction and inflammation
Exercise-Based Vagal Activation
At-home vagus nerve exercises provide a no-cost, no-device approach to improving vagal tone:
- Diaphragmatic breathing (extended exhale)
- Cold exposure (face immersion, cold shower)
- Humming and gargling
- Meditation and yoga
- Social engagement (polyvagal theory)
Combined Protocol for Chronic Fatigue and POTS
Phase 1 (Weeks 1-4): Foundation
- Begin taVNS: 20 minutes twice daily
- Implement daily diaphragmatic breathing: 5 minutes, 3 times daily
- Track HRV with wearable device (baseline measurement)
- Address sleep optimization (vagal tone is highest during sleep)
Phase 2 (Weeks 5-12): Building
- Continue taVNS
- Add gentle exercise (walking, yoga — with HR monitoring for POTS patients)
- Add cold exposure (30-second cold finish to shower)
- Increase breathing practice: 10 minutes, twice daily
- Anti-inflammatory nutrition (Mediterranean-type diet, high omega-3)
Phase 3 (Months 3+): Integration
- Continue daily vagal activation (device + exercises)
- Progressive exercise increase (guided by HRV and symptom response)
- Address concurrent factors: viral reactivation, brain fog, hormonal optimization
- Monthly HRV tracking to document progress
POTS-Specific Considerations
POTS patients require additional management alongside VNS:
- Salt and fluid loading: 2-3L water daily with 3-5g sodium supplementation
- Compression garments: Waist-high compression (30-40mmHg) for venous return
- Counter-maneuvers: Leg crossing, muscle tensing during prolonged standing
- Medications (if needed): Midodrine, fludrocortisone, ivabradine — under physician guidance
- Recumbent exercise initially: Swimming, recumbent cycling to avoid orthostatic stress

Safety and Considerations
- taVNS is generally well-tolerated. Rare side effects include headache, ear discomfort, and dizziness. Discontinue if side effects persist.
- Do NOT use VNS with active cardiac arrhythmias or implanted cardiac devices (pacemakers, defibrillators) without cardiology clearance.
- POTS patients should start vagal exercises slowly. Some exercises (cold exposure, breath holds) can transiently worsen orthostatic symptoms in severely dysautonomic patients. Start with the gentlest approach (extended exhale breathing) and progress gradually.
- VNS is an adjunct therapy, not a standalone treatment for chronic fatigue. It addresses the autonomic component but does not treat the underlying infection, mold exposure, or other triggers.
- HRV measurements from consumer wearables are approximations. They are useful for tracking trends but should not replace clinical autonomic testing (tilt table, Valsalva maneuver, deep breathing testing) for diagnosis.
The Bottom Line
Vagus nerve stimulation targets a core mechanism of chronic fatigue and POTS — the sympathovagal imbalance and impaired cholinergic anti-inflammatory pathway that perpetuates inflammation, orthostatic intolerance, and energy depletion. The evidence supports both device-based taVNS and exercise-based vagal activation for improving fatigue, HRV, and autonomic function. In my clinical experience, vagal interventions are one of the most cost-effective and impactful additions to a comprehensive chronic fatigue treatment plan — and they address a mechanism that antimicrobials alone cannot reach.
References
- Naviaux RK, Naviaux JC, Li K, et al. Metabolic features of chronic fatigue syndrome. Proc Natl Acad Sci U S A. 2016;113(37):E5472-E5480. PMID: 27573827
- Tracey KJ. The inflammatory reflex. Nature. 2002;420(6917):853-859. PMID: 12490958
- Stavrakis S, Stoner JA, Humphrey MB, et al. TREAT AF (Transcutaneous Electrical Vagus Nerve Stimulation to Suppress Atrial Fibrillation). JACC Clin Electrophysiol. 2020;6(3):282-291. PMID: 32192678
- Koopman FA, Chavan SS, Miljko S, et al. Vagus nerve stimulation inhibits cytokine production and attenuates disease severity in rheumatoid arthritis. Proc Natl Acad Sci U S A. 2016;113(29):8284-8289. PMID: 27382171