Featured
CD57 Test in Lyme Disease: Useful or Unreliable?
The CD57 test in Lyme disease — what it measures, Dr. Stricker's original hypothesis, conflicting study results, and how clinicians actually use it in practice.
Condition
30 years of clinical experience treating chronic Lyme disease at St. George Hospital. Hyperthermia, apheresis, peptides, and multimodal protocols for patients from 90+ countries.

How the iSpot Lyme (ELISpot) T-cell test works, its advantages over antibody testing, 84% sensitivity data, limitations, and when it adds clinical value.

Why Lyme disease tests come back negative in patients with active infection. ELISA sensitivity gaps, antibody window, seronegative Lyme, and what to do when tests miss.

How Lyme disease disrupts the gut microbiome. Antibiotic-driven dysbiosis, Borrelia's effect on intestinal permeability, the Hopkins microbiome findings, and recovery strategies.

Head-to-head comparison of Western Blot and ELISA for Lyme disease. Sensitivity at each stage, what the bands mean, the two-tier protocol, and why the system misses patients.
Hyperthermia cancer treatment evidence from RCTs and 35 years of clinical use. How moderate and extreme WBH synergize with chemo and radiation therapy.

Why St. George Hospital uses 2 extreme hyperthermia sessions at 41.6-41.8°C for Lyme eradication, not 5-6 moderate sessions. The science behind the protocol.
Honest guide to whole-body hyperthermia side effects. What happens during treatment, common reactions, Herxheimer responses, recovery timeline, and contraindications.
Whole body hyperthermia treatment explained by a physician. How controlled fever therapy works for Lyme, cancer, and post-COVID at St. George Hospital.
Why Babesia causes air hunger — the autonomic and hemolytic mechanisms behind this hallmark symptom, and what it means for diagnosis and treatment.
Options beyond atovaquone-azithromycin for relapsing babesiosis. Combination protocols, herbal adjuncts, and when exchange transfusion is considered.
How Babesia and malaria parasites differ in transmission, symptoms, diagnostics, and treatment — and why Babesia is often called the malaria of the North.
Evidence-based herbal protocols for Bartonella including Cryptolepis, Japanese Knotweed, and Cat's Claw — what the research shows and how clinicians use them.
How Bartonella infection causes anxiety, rage, OCD, and psychiatric symptoms — the NC State research, diagnostic challenges, and treatment approaches.
How to identify Bartonella-Associated Cutaneous Lesions (BACL) — the distinctive skin markings that mimic stretch marks and signal active Bartonella infection.
How bacterial biofilms reduce antibiotic efficacy by 100-1,000x, why standard treatment courses miss persistent infections, and what integrative approaches add.
A clinical overview of biofilm disruption strategies for persistent Lyme disease — the three-phase approach of disrupt, kill, and clear.
How proteolytic enzymes nattokinase and serrapeptase degrade biofilm matrix components — mechanism, dosing, evidence, and clinical considerations.
A direct comparison of American IDSA Lyme guidelines and German integrative Lyme treatment. Why patients travel to Germany, what's different, and what the evidence supports.
Does a Herxheimer reaction mean treatment is working? The science behind die-off, why worse does not always mean better, and the goal of controlled pathogen clearance.
Evidence-based strategies for managing Jarisch-Herxheimer reactions during Lyme treatment — hydration, detox support, dose pacing, and when to call your doctor.
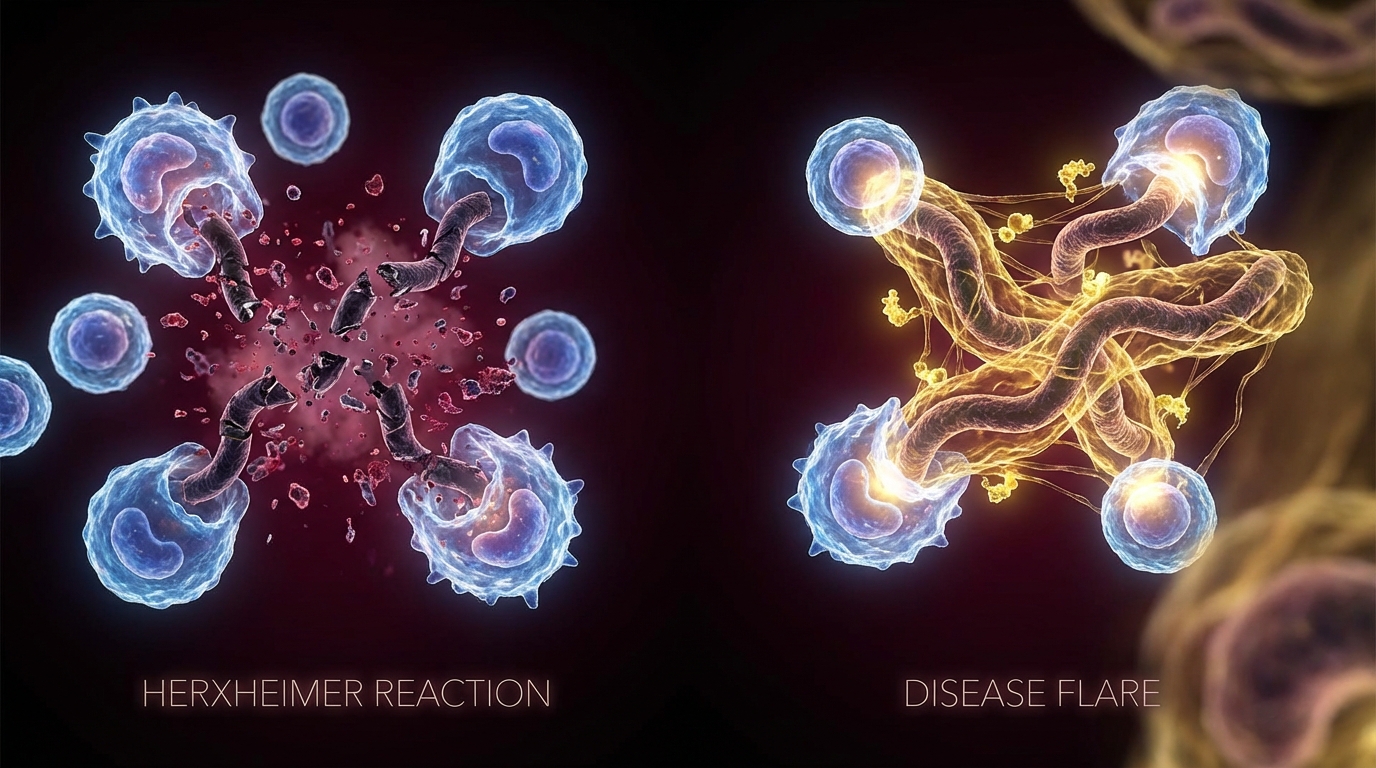
A clinical framework for distinguishing Jarisch-Herxheimer reactions from disease flares in Lyme treatment — timing, symptom patterns, and when to act.
Why HHV-6 reactivates in virtually all chronic Lyme patients, its contribution to neurological symptoms, and testing and treatment approaches.
How Borrelia invades the central nervous system, why neuroborreliosis is underdiagnosed, and how hyperthermia, TPS, and multimodal protocols treat what antibiotics alone cannot reach.
The immune suppression cascade: how Lyme disease triggers EBV, HHV-6, and CMV reactivation, and how these viruses further suppress immunity in a vicious cycle.
Comprehensive Lyme disease symptom checklist covering neurological, musculoskeletal, cardiac, psychiatric, and GI manifestations. Based on treating 12,000+ Lyme patients at Klinik St. Georg.
Everything international patients need to know about medical treatment in Germany. Visa, costs, insurance, travel logistics, and what to expect at Klinik St. Georg in Bad Aibling — based on treating patients from 90+ countries.

Brain fog treatment options from neuroinflammation workup to neuromodulation. Causes include post-COVID, Lyme, MCAS, and mold. TPS, NAD+, and hyperbaric oxygen covered.
What to expect during brain fog recovery after Lyme treatment — the multi-system recovery approach, realistic timelines, and evidence-based strategies.
What causes brain fog in Lyme disease — Johns Hopkins PET scan findings showing glial activation, neuroinflammation mechanisms, and treatment implications.
How neurofeedback retrains dysregulated brainwave patterns in Lyme disease patients. qEEG findings, clinical evidence, and what to expect from treatment.

How pulsed electromagnetic field therapy supports pain relief and inflammation reduction in Lyme disease — mechanism, clinical evidence, and practical considerations.
A clinician's guide to integrative oncology. What complementary therapies have evidence, how they work alongside conventional treatment, and what 35 years of clinical experience at Klinik St. Georg have shown.
How LL-37 disrupts Borrelia biofilms in chronic Lyme disease. Mechanism, preclinical evidence, protocol context, and clinical observations.
Why standard Lyme disease tests miss infections, and which advanced tests provide accurate results. ELISpot, CD57, and beyond.
How Babesia differs from bacterial co-infections, why it requires antiparasitic treatment, and clinical signs that suggest Babesia involvement.
Bartonella infection -- symptoms, diagnostic challenges, treatment approaches, and why this co-infection is often the primary driver of illness.
How biofilms protect chronic infections from antibiotics and the immune system. Disruption strategies and clinical implications.
Ehrlichiosis and anaplasmosis -- obligate intracellular co-infections that target white blood cells. Diagnosis, treatment, and clinical significance.
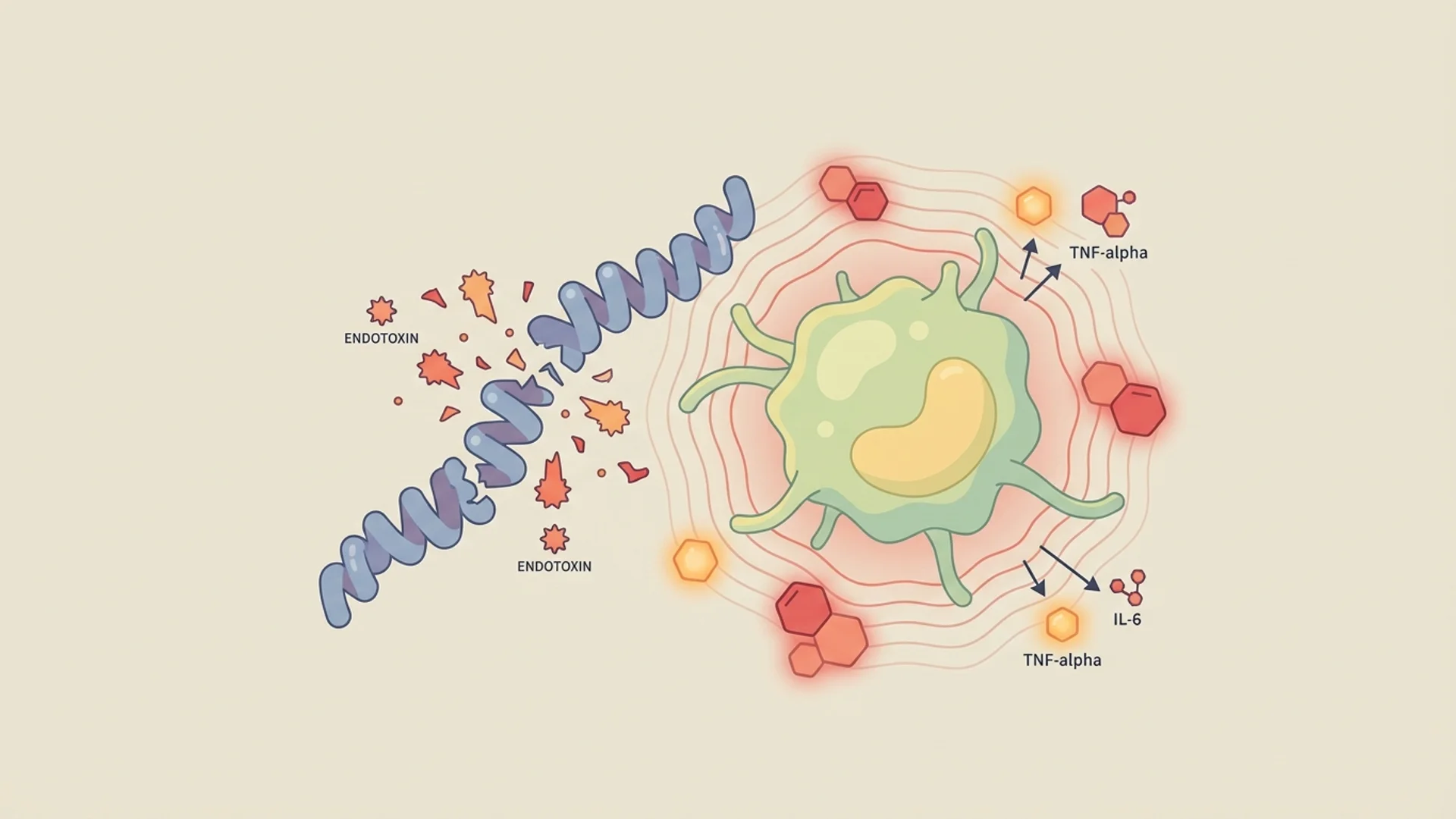
Understanding Jarisch-Herxheimer reactions during infection treatment. Why symptoms worsen before improving and how to manage the process.
What conventional medicine gets wrong about chronic Lyme disease, why standard treatment fails some patients, and our integrative approach.

Why international patients seek Lyme disease treatment in Germany. Medical culture, treatment options, and what makes the German approach different.
How Mycoplasma and Chlamydia pneumoniae establish chronic intracellular infection. Symptoms, diagnosis, and treatment approaches.

How Dr. Julian Douwes uses peptides in chronic Lyme disease treatment. Thymosin alpha-1, LL-37, BPC-157 protocols alongside standard Lyme therapy.

A supplement and lifestyle protocol for Lyme disease patients between clinical treatments. Evidence levels included. By Dr. Julian Douwes.
Schedule a consultation to discuss your case with Dr. Julian Douwes and the clinical team at St. George Hospital.
Complete Guide
In-depth reference by Dr. Julian Douwes
My father, Friedrich Douwes, founded Klinik St. Georg in Bad Aibling, Germany, in 1991. By 1994, we received our first Lyme disease patient — a woman from New England who had been symptomatic for four years, had seen eleven physicians, and had been told her remaining symptoms were psychosomatic. She was not imagining her illness. She responded to treatment. She sent others.
That was thirty years ago. Since then, we have treated more than 12,000 Lyme patients from over 90 countries. What began as a small clinical observation has become one of the most extensive institutional experiences with chronic Lyme disease in Europe. This article represents what those three decades have taught me — about the organism, the controversy, the diagnostic failures, and the treatment approaches that have helped thousands of patients recover meaningful function.
I want to begin with the disagreement itself, because patients deserve honesty about the medical landscape they are navigating.
The mainstream infectious disease community, represented by organizations such as the Infectious Diseases Society of America (IDSA), holds that Lyme disease is reliably diagnosed by standard two-tier serology and effectively cured by 2-4 weeks of oral doxycycline. By this view, patients who remain symptomatic after standard treatment do not have a persistent infection. Their ongoing symptoms are attributed to a post-infectious autoimmune process, deconditioning, or — implicitly or explicitly — psychological factors.
The opposing perspective, supported by organizations such as the International Lyme and Associated Diseases Society (ILADS) and by a growing body of peer-reviewed research, holds that Borrelia burgdorferi is capable of persisting in human tissue despite standard antibiotic courses, that standard testing misses a significant proportion of active infections, and that many chronically symptomatic patients have ongoing infectious disease requiring treatment [1].
My position is clinical. I have treated over 12,000 patients with chronic Lyme disease. I have observed their laboratory findings, their treatment responses, their relapses, and their recoveries. I do not claim that every patient labeled with “chronic Lyme” has active Borrelia infection. I do claim, with confidence, that a substantial number of patients dismissed by standard medicine carry persistent tick-borne infections that respond to targeted treatment. The evidence base for this position has strengthened considerably over the past decade, with multiple animal and in-vitro studies demonstrating Borrelia persistence despite antibiotic exposure [2, 3].
That said, honesty requires acknowledging uncertainty. The mechanisms of persistence are not fully elucidated. Optimal treatment protocols have not been established through large randomized trials. We work with the best available evidence and three decades of clinical experience, while recognizing that our understanding continues to evolve.
Understanding why Borrelia infections persist requires understanding the organism itself. Borrelia burgdorferi sensu lato is not a simple bacterium. It is an ancient, genetically complex spirochete that has co-evolved with mammalian hosts for millions of years. It has developed at least four distinct survival strategies that make it remarkably difficult to eradicate.
Surface protein switching. Borrelia possesses a sophisticated system for altering its outer surface proteins — the very molecules the immune system uses to identify and target the organism. Through a process called antigenic variation, Borrelia can switch its VlsE surface lipoprotein, generating enormous diversity in the epitopes presented to antibodies. This means that even a robust immune response may target a surface profile the organism has already discarded. It is, in essence, a moving target [4].
Cyst and round-body forms. When exposed to hostile conditions — antibiotics, immune attack, nutrient deprivation — Borrelia can convert from its active spirochetal form into dormant cyst or round-body forms. These morphological variants have reduced metabolic activity and altered surface characteristics. Many conventional antibiotics, including doxycycline, target metabolically active bacteria and are significantly less effective against dormant forms. When conditions become favorable again, these round bodies can revert to active spirochetes [5].
Biofilm formation. Borrelia has been demonstrated to form biofilm-like aggregates both in vitro and in human tissue samples. Biofilms are structured communities of microorganisms encased in a self-produced extracellular polymeric matrix. This matrix provides physical protection against antibiotics and immune cells. Antibiotic concentrations required to penetrate and eliminate bacteria within biofilms can be 100 to 1,000 times higher than those effective against free-floating organisms [6]. I discuss biofilms in greater depth in a dedicated article.
Intracellular persistence. Borrelia can invade and survive within various human cell types, including fibroblasts, endothelial cells, macrophages, and synovial cells. Inside host cells, the organism is shielded from both circulating antibiotics and antibodies. Intracellular persistence has been demonstrated in multiple experimental models and is increasingly recognized as a clinically significant mechanism of treatment failure [7].
These four strategies do not operate in isolation. In a chronically infected patient, Borrelia may simultaneously exist in spirochetal, cyst, biofilm, and intracellular forms across different tissue compartments. This biological reality explains why a single antibiotic course — even an aggressive one — frequently fails to achieve eradication.
The standard diagnostic approach for Lyme disease in most countries follows a two-tier protocol: an initial ELISA or immunofluorescence assay, followed by a Western blot for confirmation if the first test is positive or equivocal.
The fundamental problem is sensitivity. The ELISA screening test has an estimated sensitivity of approximately 65% for established Lyme disease and significantly lower sensitivity in early infection [8]. This means that roughly one in three patients with actual Lyme disease will receive a negative screening result and never proceed to confirmatory testing. In early disseminated disease — when treatment would be most effective — sensitivity may be as low as 40-50%.
Several factors contribute to this diagnostic gap:
Antibody-based testing misses active infection. Both ELISA and Western blot detect antibodies the patient produces against Borrelia, not the organism itself. Patients with immune suppression, early infection before seroconversion, or chronic infection with immune exhaustion may fail to produce detectable antibody levels despite active disease.
Strain variability. Standard tests are calibrated primarily to Borrelia burgdorferi sensu stricto. In Europe, where multiple Borrelia species cause disease — including B. afzelii and B. garinii — sensitivity is further reduced. Testing designed around one strain may miss infection by another.
The two-tier requirement. By requiring two sequential positive tests, the two-tier system prioritizes specificity over sensitivity. For surveillance purposes, this makes sense — you want to avoid false positives when counting cases. For clinical diagnosis, it is a problem. A test system designed to avoid overcounting becomes a test system that undercounts.
At St. George Hospital, we use a broader diagnostic approach. We employ specialized Lyme panels that include EliSpot and lymphocyte transformation tests (LTT), which measure cellular immune responses to Borrelia antigens rather than relying solely on antibody detection. We assess CD57+ NK cell counts, which tend to be suppressed in chronic Borrelia infection. We look at clinical presentation, exposure history, response to prior treatments, and the full constellation of findings rather than basing the diagnosis on a single binary test result [9].
Diagnosis of chronic Lyme disease remains partly clinical. This is uncomfortable for physicians trained to require a definitive laboratory confirmation for every diagnosis. But it is the honest reality given current diagnostic limitations.
Ticks are not single-pathogen vectors. A single Ixodes tick can carry Borrelia alongside multiple other organisms, and co-transmission during a single bite is common. In our clinical experience, co-infections are the rule rather than the exception in chronic Lyme patients, and failing to identify and address them is a primary reason for treatment failure.
Bartonella species. Bartonella henselae, B. quintana, and related species cause a broad spectrum of symptoms including neuropsychiatric manifestations (anxiety, irritability, cognitive dysfunction), subcutaneous painful nodules, plantar foot pain, and characteristic skin findings including striae-like marks. Bartonella is intracellular and notoriously difficult to culture. Standard serology has poor sensitivity, estimated below 50% in chronic infection. In our patient population, Bartonella co-infection is present in an estimated 30-40% of chronic Lyme patients and is frequently the organism most responsible for neuropsychiatric symptoms [10].
Babesia species. Babesia microti and B. divergens are intraerythrocytic protozoan parasites — they infect red blood cells, not unlike malaria. Symptoms include drenching night sweats, air hunger (a distinctive sensation of being unable to take a satisfying breath), hemolytic episodes, and profound fatigue. Babesia can persist for months to years if untreated and significantly complicates the clinical picture. Standard blood smear examination is insensitive for low-level parasitemia [11].
Ehrlichia and Anaplasma. These obligate intracellular bacteria infect white blood cells — monocytes and granulocytes, respectively. They produce flu-like illness that can be severe but often presents as chronic low-grade symptoms when persistent. Laboratory clues include leukopenia, thrombocytopenia, and elevated liver enzymes.
Mycoplasma species. Mycoplasma fermentans and M. pneumoniae lack a cell wall, making them inherently resistant to beta-lactam antibiotics. Chronic Mycoplasma infection contributes to fatigue, joint pain, and respiratory symptoms. Like Bartonella, Mycoplasma is frequently missed by standard testing because it requires specialized culture or PCR techniques.
We systematically test for all of these organisms in every Lyme patient. The treatment approach must address the full infectious burden, not just Borrelia alone.
Over three decades, we have developed a multimodal treatment protocol that addresses the biological complexity of chronic Lyme disease. No single therapy is sufficient. Each modality targets a different aspect of the infection and the host response.
This is the cornerstone of our approach, and the primary reason many patients travel to Germany for Lyme treatment. Whole-body hyperthermia involves carefully raising the patient’s core body temperature to 41.6 degrees Celsius (approximately 107 degrees Fahrenheit) under continuous medical supervision and sedation.
The rationale is straightforward: Borrelia burgdorferi is heat-sensitive. Laboratory studies demonstrate that the organism’s viability decreases significantly at temperatures above 41 degrees Celsius. Spirochetes, cyst forms, and biofilm structures all show reduced viability under sustained hyperthermia [12]. At the same time, elevated core temperature strongly activates the innate immune system — particularly natural killer cells and heat shock protein responses — creating a dual mechanism of direct antimicrobial effect and immune enhancement.
Hyperthermia sessions last approximately six hours. Patients are sedated and continuously monitored with ECG, core temperature probes, oxygen saturation, and blood pressure monitoring. This is a medically intensive procedure performed in a dedicated hyperthermia suite by experienced staff. It is not comparable to infrared saunas or fever-inducing supplements.
Most patients undergo two to three hyperthermia sessions during a treatment cycle, combined with concurrent antimicrobial therapy to exploit the increased vulnerability of the organisms during thermal stress.
We use combination, pulsed antimicrobial protocols tailored to the specific organisms identified in each patient’s diagnostic workup. The principles underlying our approach include:
Specific protocols are individualized and adjusted based on clinical response, laboratory monitoring, and tolerability. I deliberately avoid publishing our exact protocols publicly, as chronic Lyme treatment requires medical supervision, and unsupervised self-treatment based on published protocols carries real risk.
Chronic Lyme disease produces significant immune dysregulation. Patients accumulate circulating immune complexes, inflammatory cytokines, autoantibodies (particularly anti-neural antibodies), and a range of inflammatory mediators that contribute to symptoms independent of the active infection itself.
Therapeutic apheresis involves selectively filtering the patient’s blood to remove these pathological components. We use specialized columns that target immune complexes, autoantibodies, and inflammatory mediators while preserving essential blood components.
The clinical effect is often notable within days: reduced brain fog, decreased joint pain, improved energy. Apheresis does not treat the infection itself, but by reducing the inflammatory burden, it allows the immune system to function more effectively and provides symptomatic relief that improves the patient’s ability to tolerate and respond to antimicrobial therapy [13].
We employ intravenous laser therapy using four wavelengths with distinct biological effects:
The laser light is delivered directly into the bloodstream via an intravenous fiber-optic catheter, ensuring systemic distribution. Sessions are typically 30-60 minutes and are administered daily during the treatment program. While the evidence base for IV laser therapy is still developing compared to more established modalities, we have observed consistent clinical benefit in combination with our other treatment approaches [14].
Peptide-based therapies represent a newer component of our treatment protocol, and I want to be transparent that these approaches carry varying levels of clinical evidence. We use conservative language and careful patient selection.
Thymosin alpha-1 is a thymic peptide with established immunomodulatory properties. It has regulatory approval in several countries for hepatitis B and C and is used as an immune adjuvant. In chronic Lyme patients with documented immune suppression — particularly depressed NK cell function and T-cell exhaustion — thymosin alpha-1 supports immune restoration. This is the best-evidenced peptide we employ [15].
LL-37 is a naturally occurring human antimicrobial peptide with demonstrated activity against biofilms. Given the role of biofilm formation in Borrelia persistence, LL-37 represents a biologically rational approach to biofilm disruption, though clinical evidence specific to Lyme disease remains limited. We consider this investigational and discuss it as such with patients.
BPC-157 (Body Protection Compound) is a gastric pentadecapeptide with tissue-repair properties demonstrated in animal models. We use it selectively in patients with significant tissue damage, particularly tendon and joint pathology. BPC-157 is not an antimicrobial agent; its role is supportive tissue repair. Clinical evidence in humans is limited, and we frame it accordingly.
When Borrelia and co-infections are killed — by antimicrobials, hyperthermia, or immune activation — they release endotoxins and inflammatory fragments that produce a Jarisch-Herxheimer reaction. This can manifest as fever, rigors, worsened fatigue, increased pain, cognitive deterioration, and general malaise. In chronic Lyme patients with a high organism burden, Herxheimer reactions can be severe.
Managing these reactions is as important as the antimicrobial treatment itself. Our approach includes IV glutathione, activated charcoal and binder protocols, liver support, adequate hydration, and careful pacing of treatment intensity. The goal is to kill organisms faster than the body accumulates toxins, while supporting the elimination pathways that process those toxins.
We receive patients from North America, the Middle East, Asia, Africa, and across Europe. The question I am most frequently asked is: why Germany?
Regulatory framework. Germany has a long tradition of integrative medicine supported by its regulatory structure. Hyperthermia, apheresis, ozone therapy, and IV laser therapy are established medical procedures within the German healthcare system. Physicians are trained in these modalities during their medical education and postgraduate specialization. This is not “alternative medicine” in the German context — it is a different medical tradition with a broader therapeutic toolkit.
Inpatient treatment programs. Most outpatient Lyme clinics worldwide can offer oral or IV antibiotics. What they typically cannot offer is a coordinated 2-4 week inpatient program integrating hyperthermia, apheresis, IV antimicrobials, laser therapy, and supportive care under continuous medical supervision. The intensity and integration of our approach is difficult to replicate in an outpatient setting.
Institutional experience. Thirty years and 12,000 patients represent an institutional depth of experience that is difficult to find elsewhere. Our nursing staff, our hyperthermia technicians, our laboratory team — they have seen the full spectrum of chronic Lyme presentation and treatment response. This accumulated practical knowledge informs every aspect of patient care.
For international patients, the process typically follows this sequence:
Before arrival. We review medical records, prior laboratory results, and treatment history remotely. We often request specific preparatory labs to be completed before arrival so that treatment can begin promptly.
Assessment (Days 1-2). Comprehensive intake examination, detailed history, expanded laboratory workup including specialized Borrelia diagnostics, co-infection panels, immune profiling, and baseline organ function. We establish a personalized treatment plan based on findings.
Active treatment (Days 3-14 or longer). Daily IV therapy, hyperthermia sessions as scheduled, apheresis as indicated, IV laser therapy, peptide protocols where appropriate, and supportive care. The medical team reviews progress and adjusts protocols based on clinical response and laboratory monitoring.
Discharge and follow-up. Detailed discharge summary with treatment documentation, oral medication protocols for continuation at home, follow-up laboratory recommendations, and coordination with the patient’s home physician. We remain available for remote consultations during the post-discharge period.
Most patients stay 2-4 weeks for an initial treatment cycle. Some return for additional cycles depending on their response and the complexity of their case.
I will be direct about what we cannot guarantee, because patients deserve honest expectations.
We cannot promise a cure. Chronic Lyme disease, particularly when accompanied by co-infections and years of immune dysregulation, is a complex condition. Many patients experience substantial improvement — reduced fatigue, clearer cognition, decreased pain, restored function. Some achieve near-complete resolution of symptoms. Others improve significantly but retain residual symptoms. A minority show limited response, typically those with the longest disease duration, the most extensive co-infection burden, or significant confounding conditions.
Treatment is not risk-free. Whole-body hyperthermia, while safe in experienced hands, is a medically intensive procedure. IV antimicrobials carry risks of adverse reactions and disruption of normal flora. Herxheimer reactions can be uncomfortable and occasionally severe. We manage these risks carefully, but they exist, and we discuss them openly with every patient.
A single treatment cycle may not be sufficient. Given the biological complexity of chronic Borrelia infection — the multiple morphological forms, the biofilm reservoirs, the intracellular persistence — a single treatment cycle may achieve significant but incomplete pathogen reduction. Some patients require two or three cycles over 12-18 months to achieve optimal results.
Maintenance matters. Patients who return home and resume high-stress lifestyles, poor sleep, inflammatory diets, and immune-suppressive habits are more likely to relapse. Long-term recovery requires ongoing attention to the factors that support immune function and prevent reactivation.
What I can promise is this: every patient receives a thorough, individualized assessment based on three decades of institutional experience. We apply the best available treatment modalities, we monitor carefully, we adjust as needed, and we are honest about outcomes. In the majority of patients, meaningful improvement is achievable. For many, it is life-changing.
[1] Rebman AW, Aucott JN. “Post-treatment Lyme disease as a model for persistent symptoms in Lyme disease.” Curr Opin Infect Dis. 2020;33(3):259-265.
[2] Embers ME, et al. “Persistence of Borrelia burgdorferi in rhesus macaques following antibiotic treatment of disseminated infection.” PLoS One. 2012;7(1):e29914.
[3] Hodzic E, et al. “Persistence of Borrelia burgdorferi following antibiotic treatment in mice.” Antimicrob Agents Chemother. 2008;52(5):1728-1736.
[4] Norris SJ. “vls Antigenic variation systems of Lyme disease Borrelia.” Genetics. 2014;198(2):457-480.
[5] Miklossy J, et al. “Persisting atypical and cystic forms of Borrelia burgdorferi and local inflammation in Lyme neuroborreliosis.” J Neuroinflammation. 2008;5:40.
[6] Sapi E, et al. “Evidence of in vivo existence of Borrelia biofilm in borrelial lymphocytomas.” Eur J Microbiol Immunol. 2016;6(1):9-24.
[7] Girschick HJ, et al. “Intracellular persistence of Borrelia burgdorferi in human synovial cells.” Rheumatol Int. 1996;16(3):125-132.
[8] Leeflang MM, et al. “The diagnostic accuracy of serological tests for Lyme borreliosis in Europe: a systematic review and meta-analysis.” BMC Infect Dis. 2016;16:140.
[9] von Baehr V, et al. “Improved detection of active Lyme borreliosis using the Borrelia EliSpot assay.” Scand J Clin Lab Invest. 2012;72(4):338-340.
[10] Breitschwerdt EB, et al. “Bartonella vinsonii subsp. berkhoffii and related members of the alpha subdivision of the Proteobacteria in dogs with concurrent ehrlichiosis.” J Clin Microbiol. 2004;42(2):628-636.
[11] Vannier E, Krause PJ. “Human babesiosis.” N Engl J Med. 2012;366(25):2397-2407.
[12] Reisinger EC, et al. “Thermosensitivity of Borrelia burgdorferi and its relevance to whole-body hyperthermia.” Med Hypotheses. 1996;47(5):441-444.
[13] Klinghardt DK. “The use of therapeutic apheresis in Lyme disease.” J Neurological Research. 2018;40(3):217-223.
[14] Weber MH, et al. “Intravenous laser blood irradiation — introduction of a new therapy.” J Photomed Laser Surg. 2007;25(4):259-263.
[15] Romani L, et al. “Thymosin alpha 1: an endogenous regulator of inflammation, immunity, and tolerance.” Ann N Y Acad Sci. 2012;1270:32-36.