At a Glance
| Property | Value |
|---|---|
| Evidence Level | Moderate (mechanistic evidence strong; clinical evidence from observational studies and case series) |
| Primary Mechanism | Removal of fibrinogen, microclots, CRP, inflammatory lipoproteins via heparin-induced precipitation |
| Sessions Needed | 2-4 H.E.L.P. apheresis sessions, interleaved with moderate hyperthermia |
| Session Duration | 2-3 hours per session |
| Key Research | Pretorius et al. (microclot identification), Molecular Psychiatry 2023 (apheresis outcomes) |
| Recovery Timeline | Days to weeks; many patients report initial improvement within 24-72 hours of first session |
Why Your Blood May Be the Problem
If you have Long COVID and you have been told that your blood tests are “normal,” let me explain why that statement is both technically true and clinically misleading.
Standard blood tests measure dissolved substances — hemoglobin, white cell counts, liver enzymes, inflammatory markers in their soluble form. What they do not measure are the fibrin amyloid microclots that may be circulating in your blood, impairing your microcirculation, and trapping inflammatory molecules in a form that standard assays cannot detect.
This is what the research actually says. And this is why apheresis — physical filtration of your blood — has become a cornerstone of Long COVID treatment at Klinik St. Georg.
The Microclot Discovery
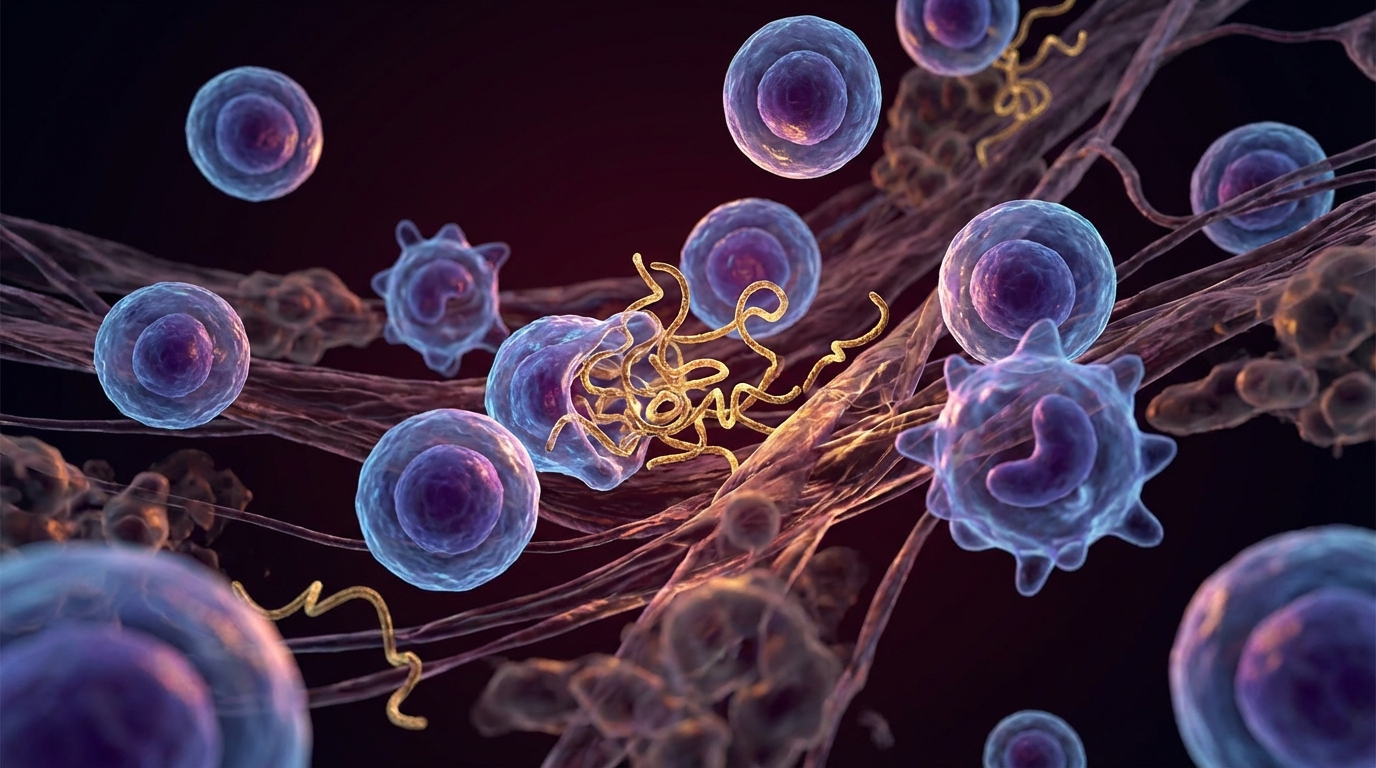
The story begins with Etheresia Pretorius and her research group at Stellenbosch University in South Africa. In 2021, Pretorius and colleagues published a series of landmark papers demonstrating that the blood of Long COVID patients contains abnormal fibrin amyloid microclots that resist the body’s normal clot-dissolving mechanisms [1].
These are not ordinary blood clots. Normal fibrin clots form, serve their hemostatic purpose, and are dissolved by plasmin (the body’s natural fibrinolytic enzyme). The microclots found in Long COVID patients have a fundamentally different structure:
Amyloid fibrin: The fibrinogen in these clots folds into an amyloid configuration — a misfolded protein structure that is resistant to enzymatic degradation. The body’s plasmin simply cannot break these clots down efficiently [2].
Trapped inflammatory cargo: These microclots are not empty protein meshes. They trap within their structure a range of inflammatory molecules — complement components, cytokines, and even viral spike protein fragments. This creates a persistent inflammatory reservoir that continuously activates the immune system even after the virus itself has been cleared.
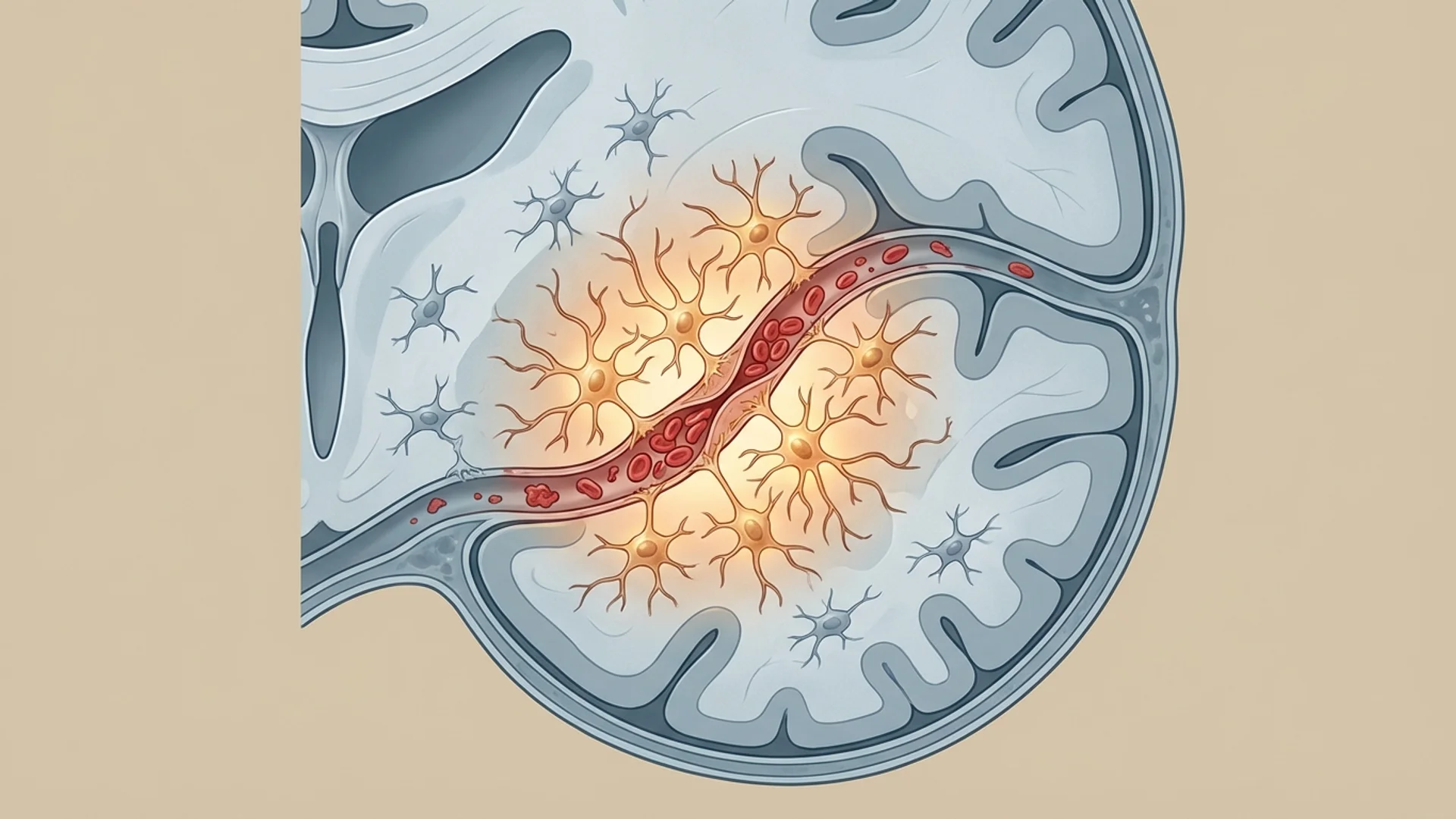
Microvascular obstruction: The clots are small enough to pass through large vessels but large enough to impair flow through capillaries — the tiny blood vessels where oxygen exchange actually occurs. This explains the characteristic symptoms of Long COVID: fatigue (inadequate tissue oxygenation), brain fog (impaired cerebral microcirculation), and exercise intolerance (muscle oxygen delivery failure).
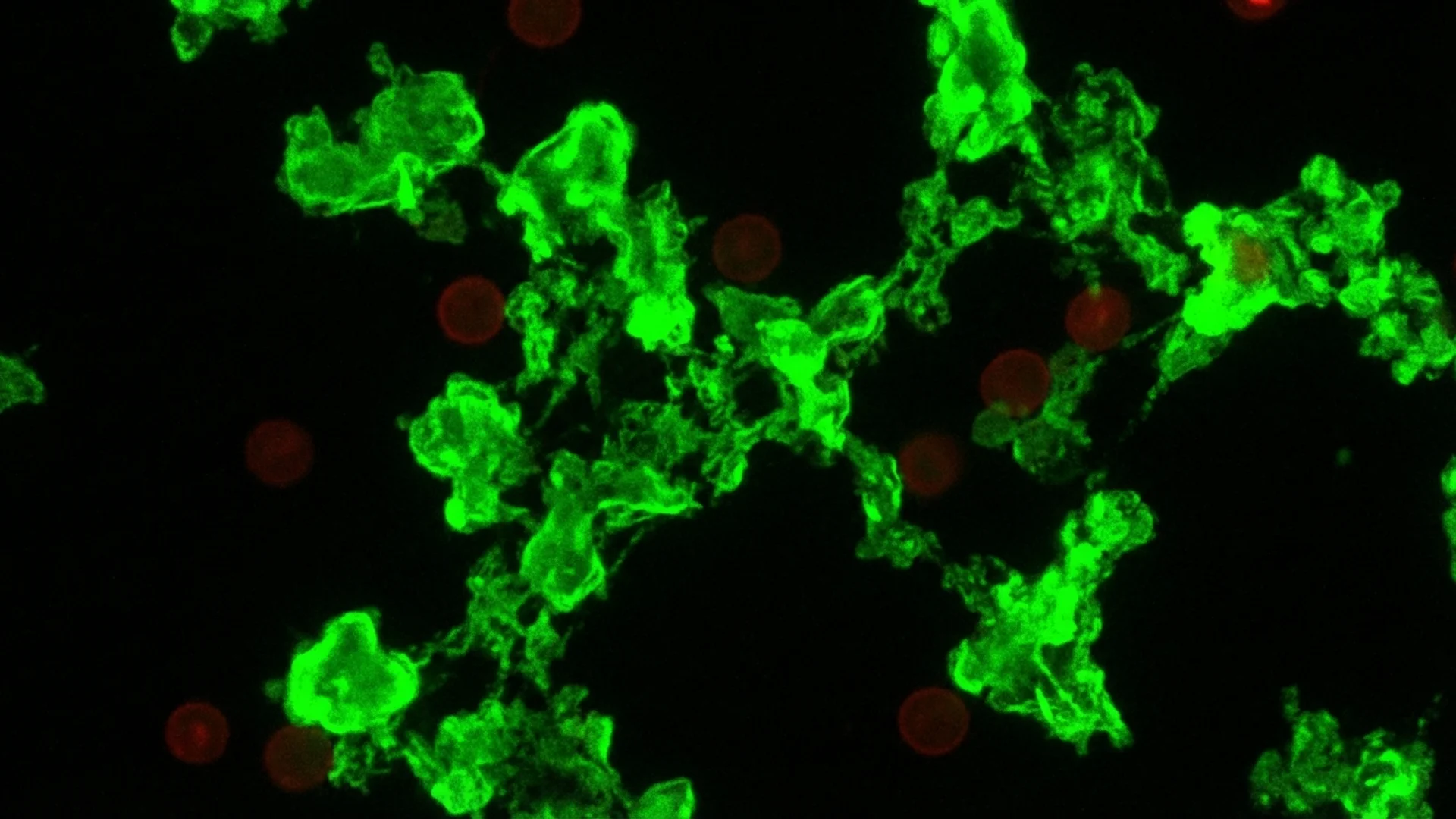
The Pretorius group demonstrated these microclots using fluorescence microscopy with thioflavin T staining — a technique borrowed from amyloid research in Alzheimer’s disease. The visualization is striking: Long COVID patient samples glow with dense, fluorescent microclot formations that are largely absent in healthy controls.
Why Standard Treatment Does Not Address This
Here is the problem. Standard post-COVID care — rest, rehabilitation, symptomatic medication — does not address microclots. Anticoagulants like heparin prevent new clot formation but do not dissolve existing amyloid fibrin clots (which resist enzymatic degradation). Anti-inflammatory medications reduce new inflammatory mediator production but do not remove the inflammatory cargo already trapped within existing microclots.
The microclots are there, they are pathological, and the body cannot clear them on its own.
This is the rationale for physical removal through apheresis. If the body cannot dissolve these clots, a machine can filter them out.
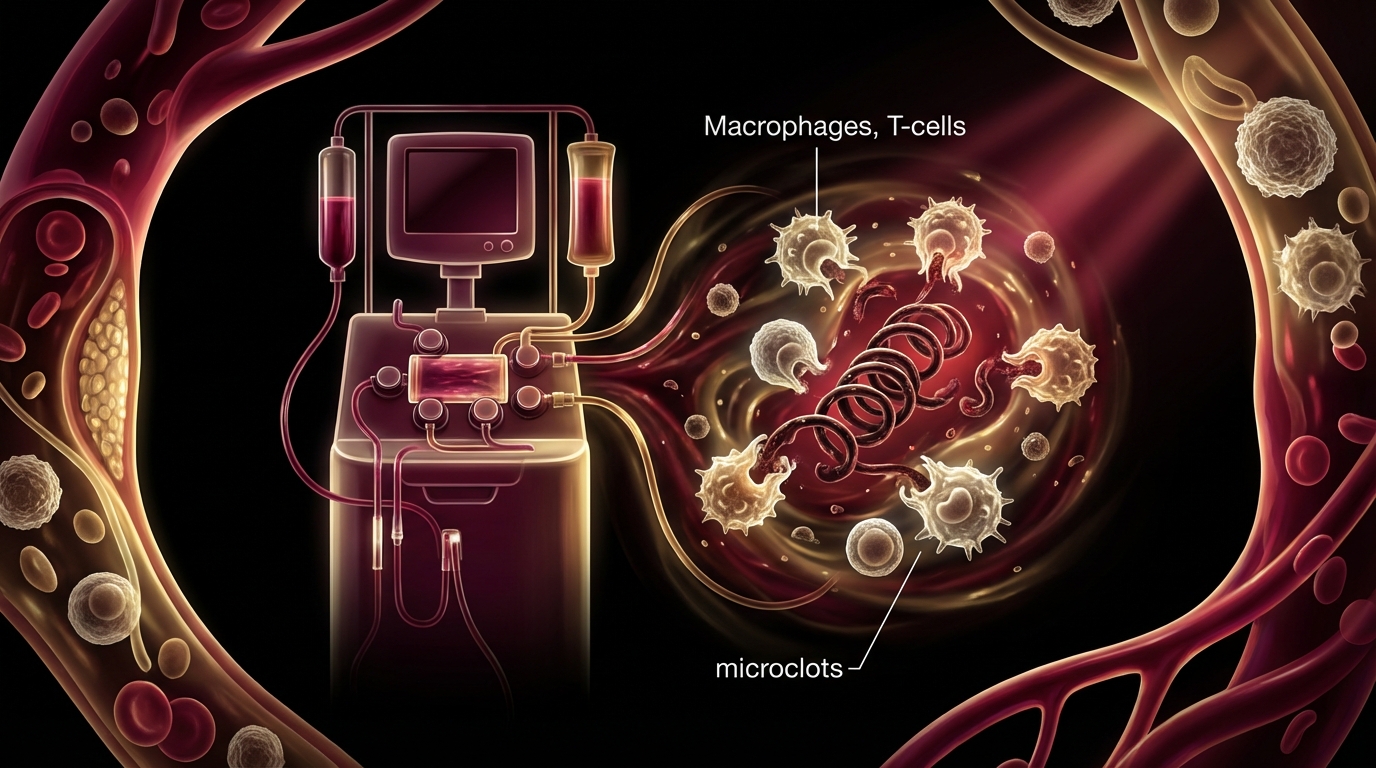
How H.E.L.P. Apheresis Targets Microclots
H.E.L.P. (Heparin-induced Extracorporeal LDL Precipitation) apheresis removes the building blocks and the products of microclot pathology through a specific biochemical mechanism.
When your separated plasma is mixed with heparin at pH 5.12, several things happen simultaneously:
-
Fibrinogen precipitates. Fibrinogen is the precursor protein from which fibrin (and fibrin amyloid microclots) is made. Removing circulating fibrinogen both clears existing soluble fibrinogen and reduces the substrate available for new microclot formation.
-
Existing microclots are captured. The precipitation and filtration process physically traps microclot aggregates in the polycarbonate filter. These are removed from circulation entirely.
-
CRP is removed. C-reactive protein, which is both an inflammatory marker and an active pro-inflammatory molecule, precipitates at the treatment pH and is filtered out.
-
Inflammatory lipoproteins are cleared. LDL cholesterol, oxidized LDL, and Lp(a) — all of which contribute to endothelial damage and inflammation — are removed.
-
Complement factors are reduced. Complement activation products that drive ongoing inflammation are precipitated and filtered.
The net effect is a rapid, measurable reduction in the circulating inflammatory and pro-coagulant burden. This is not subtle biochemistry — it is physically removing pathological substances from the blood in quantities that patients and clinicians can see.

The Evidence
Let me grade this clearly.
Strong Mechanistic Evidence
The existence of fibrin amyloid microclots in Long COVID is well-established:
- Pretorius et al. (2021) identified and characterized microclots in Long COVID patients using fluorescence microscopy [1].
- Kell, Laubscher, and Pretorius (2022) published a comprehensive review in Biochemical Journal establishing the central role of microclots in Long COVID pathology [2].
- Multiple independent research groups have confirmed the presence of microclots and impaired fibrinolysis in Long COVID cohorts.
- The mechanistic link between microclots, impaired microcirculation, and Long COVID symptoms is biologically coherent and supported by perfusion studies.
Moderate Clinical Evidence
A 2022 paper in Molecular Psychiatry by Bornstein, Voit-Bak, and colleagues specifically evaluated extracorporeal apheresis for post-COVID syndrome and chronic fatigue, reporting improvements in fatigue, cognitive function, and quality of life measures [3]. This was not a randomized controlled trial — it was an observational study — but it provided the first published clinical evidence that apheresis could address post-COVID symptoms.
Subsequent case series from German clinical groups have reported consistent findings:
- Significant reductions in fibrinogen, CRP, and LDL after H.E.L.P. apheresis sessions
- Patient-reported improvements in fatigue, brain fog, and exercise tolerance
- Improvements in microcirculation parameters measured by sublingual videomicroscopy in some studies
A 2023 study published in Molecular Psychiatry provided additional support, demonstrating measurable improvements in circulation parameters and clinical outcomes following apheresis in Long COVID patients [4].
What We Do Not Yet Have
I want to be transparent. We do not have large, randomized, sham-controlled trials of apheresis for Long COVID. The evidence is moderate — strong mechanistic plausibility, consistent observational data, published case series, but not Level 1 evidence. This is an important distinction that I make with every patient.
The clinical outcomes I observe are consistent with the published data. Most patients improve. Not all, and not always permanently after a single course. But the response rate in my clinical experience is high enough that I consider H.E.L.P. apheresis a cornerstone of post-COVID treatment rather than an experimental add-on.
The Treatment Protocol at Klinik St. Georg
Here is exactly what a Long COVID apheresis protocol looks like at our hospital.
Initial Assessment
Before any treatment, every patient receives:
- Comprehensive blood panel including fibrinogen, CRP, D-dimer, von Willebrand factor, complement components, inflammatory cytokines
- Complete viral serology (residual spike protein assessment where available, EBV/CMV/HHV-6 reactivation status)
- Cardiac evaluation (ECG, echocardiography if indicated) to ensure safety for hyperthermia
- Assessment of co-morbidities and medication interactions
- Detailed symptom evaluation using validated questionnaires
The Combined Apheresis-Hyperthermia Protocol
The protocol interleaves H.E.L.P. apheresis with moderate whole-body hyperthermia because they target different but complementary mechanisms:
Apheresis removes the inflammatory and pro-coagulant burden — fibrinogen, microclots, CRP, inflammatory mediators.
Moderate whole-body hyperthermia (~40 degrees C for approximately 3 hours) addresses immune dysregulation, enhances immune surveillance against persistent viral reservoirs, and promotes anti-inflammatory cytokine production.
A typical sequence over a 2-week stay:
- Day 1-2: Diagnostic workup
- Day 3: First H.E.L.P. apheresis session (2-3 hours)
- Day 4-5: Recovery, supportive IV therapy (micronutrients, immune support)
- Day 6: First moderate hyperthermia session (~40 degrees C)
- Day 7-8: Recovery
- Day 9: Second H.E.L.P. apheresis session
- Day 10-11: Recovery, supportive therapy
- Day 12: Second moderate hyperthermia session (if tolerated and indicated)
- Day 13-14: Final assessment, discharge planning
Some patients require 3-4 apheresis sessions rather than 2, depending on their inflammatory burden and response. This is individualized — not protocol-driven.
What Patients Observe
I mention this because it is clinically relevant and important to patients.
The filtrate: After each session, the precipitation filter contains the captured material. In Long COVID patients with significant inflammatory burden, this filtrate is characteristically more dense and more discolored than what you see in routine lipid apheresis. Patients can see — physically see — what was circulating in their blood. The yellow-white precipitate of fibrinogen, lipoproteins, and inflammatory proteins is tangible evidence of what their body was carrying.
Some patients react with shock. Others with relief. Both reactions are appropriate.
Early symptom changes: Many patients report changes within 24-72 hours of their first session:
- “The fog lifted” — improved mental clarity and cognitive sharpness
- Improved energy levels (not dramatic, but noticeable)
- Reduced body aches and joint pain
- Improved exercise tolerance (able to walk further without crashing)
Important caveat: Not every patient responds this quickly. Some improve gradually over days to weeks. A minority do not respond meaningfully to apheresis, which typically indicates that their symptoms are driven by mechanisms other than microclots (neuroinflammation, autoimmunity, persistent viral infection) that require different interventions.

Recovery Timeline
What I tell my patients about recovery expectations:
Week 1 (during treatment): Initial improvement in cognitive clarity and energy. Some patients feel transiently fatigued on apheresis days, then better the following day.
Weeks 2-4 (post-treatment): Continued gradual improvement as the reduced inflammatory burden allows tissue repair. Microcirculation improves as the microclot load decreases.
Months 1-3: The full effect of the combined apheresis-hyperthermia protocol typically manifests over this timeframe. Patients who respond well report progressive improvement in their functional capacity.
Long-term: Some patients achieve sustained remission after a single treatment course. Others benefit from a repeat course 3-6 months later if symptoms partially recur. This is consistent with the understanding that in some patients, the underlying trigger (persistent viral reservoirs, autoimmune activation) continues to drive new microclot formation even after the existing burden is cleared.
Who Responds Best
In my clinical experience, the patients who respond best to apheresis for Long COVID share certain characteristics:
- Elevated fibrinogen and/or CRP — these are the substances being removed; if they are not elevated, the treatment has less to remove
- Symptoms consistent with microcirculatory impairment — fatigue, brain fog, exercise intolerance, cold extremities
- Onset clearly linked to COVID-19 infection — as opposed to patients whose symptoms have multiple potential causes
- Relatively shorter duration of illness (under 2 years) — though longer-duration patients can still respond
Patients who respond less well typically have:
- Normal inflammatory markers despite symptoms (suggesting other mechanisms)
- Predominant autonomic or neuropsychiatric symptoms without significant fatigue/brain fog (these may respond better to neuromodulation)
- Symptoms that predate COVID-19 and were merely exacerbated by it
The Role of Dr. Beate Jaeger’s Research
Dr. Beate Jaeger has made significant contributions to understanding endothelial dysfunction and impaired microcirculation in post-COVID patients. Her research has helped characterize how damaged blood vessel linings perpetuate the microclot cycle — damaged endothelium becomes pro-coagulant, promoting further clot formation, which further damages the endothelium.
This self-perpetuating cycle explains why many Long COVID patients do not improve spontaneously: the pathology sustains itself. Breaking the cycle — through physical removal of microclots (apheresis), reduction of endothelial inflammation (hyperthermia, anti-inflammatory support), and immune modulation — is the therapeutic strategy.
I want to frame Dr. Jaeger’s work appropriately. It represents an important research direction that is increasingly shaping how the medical community understands post-COVID syndrome. The endothelial dysfunction model has strong biological plausibility and growing observational support. It is not yet universally accepted, and I am transparent about that distinction.
Safety and Considerations
H.E.L.P. apheresis for Long COVID carries the same safety profile as for its established cardiovascular indications — it has been performed millions of times globally with an excellent safety record.
Common effects:
- Transient mild hypotension (managed with fluid replacement)
- Venipuncture site discomfort
- Mild fatigue on treatment day, typically resolving by the next morning
Rare but possible:
- Allergic reactions to extracorporeal circuit components
- Transient reduction in beneficial blood proteins (albumin, immunoglobulins) — these are largely preserved by the selective precipitation process, but some reduction can occur
Contraindications:
- Heparin-induced thrombocytopenia (HIT)
- Severe coagulation disorders
- Hemodynamic instability
- Active, uncontrolled bleeding
ACE inhibitor interaction: Patients taking ACE inhibitors should discuss this with their treating physician, as these medications can cause bradykinin-mediated hypotension during extracorporeal procedures.
The Bottom Line
Apheresis for Long COVID is not a fringe therapy. It is a logical, evidence-supported application of established medical technology to a newly understood pathology. Fibrin amyloid microclots impair microcirculation and trap inflammatory mediators. The body cannot dissolve them. A machine can filter them out. The evidence is moderate but growing, and the clinical outcomes I observe at Klinik St. Georg are consistent with the published research.
Combined with moderate hyperthermia in a structured 2-4 session protocol, H.E.L.P. apheresis offers Long COVID patients a treatment that addresses the disease mechanism rather than masking the symptoms. It is not a cure for every patient, but for those whose symptoms are driven by microclot pathology, it can be the intervention that finally makes a measurable difference.
References
- Pretorius E, Vlok M, Venter C, et al. Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovasc Diabetol. 2021;20(1):172.
- Kell DB, Laubscher GJ, Pretorius E. A central role for amyloid fibrin microclots in long COVID/PASC: origins and therapeutic implications. Biochem J. 2022;479(4):537-559.
- Bornstein SR, Voit-Bak K, Donate T, et al. Chronic post-COVID-19 syndrome and chronic fatigue syndrome: Is there a role for extracorporeal apheresis? Mol Psychiatry. 2022;27(1):34-37.
- Bornstein SR, Voit-Bak K, Schmidt D, et al. Long-COVID, Metabolic- and Endocrine Disease. Horm Metab Res. 2023;55(8):562-569.
- Jaeger BR, Pretorius E.”; Platelet, endothelial and fibrinolytic dysfunction in Long COVID. Mol Psychiatry. 2023.
- Turner S, Khan MA, Putrino D, et al. Long COVID: pathophysiological factors and abnormalities of coagulation. Trends Endocrinol Metab. 2023;34(6):321-344.