If there is a single concept that transforms how you think about chronic infection, it is biofilms.
Most people — and many physicians — think of bacteria as free-floating individual organisms (planktonic bacteria) that encounter an antibiotic, are either susceptible or resistant, and live or die accordingly. This is how antibiotics are tested in the laboratory: individual bacteria in a petri dish exposed to various drugs.
The problem is that this model represents only a fraction of how bacteria actually live in the human body. An estimated 65-80% of all human infections involve biofilms [1]. In biofilm communities, bacteria behave fundamentally differently than their planktonic counterparts.
What Biofilms Are
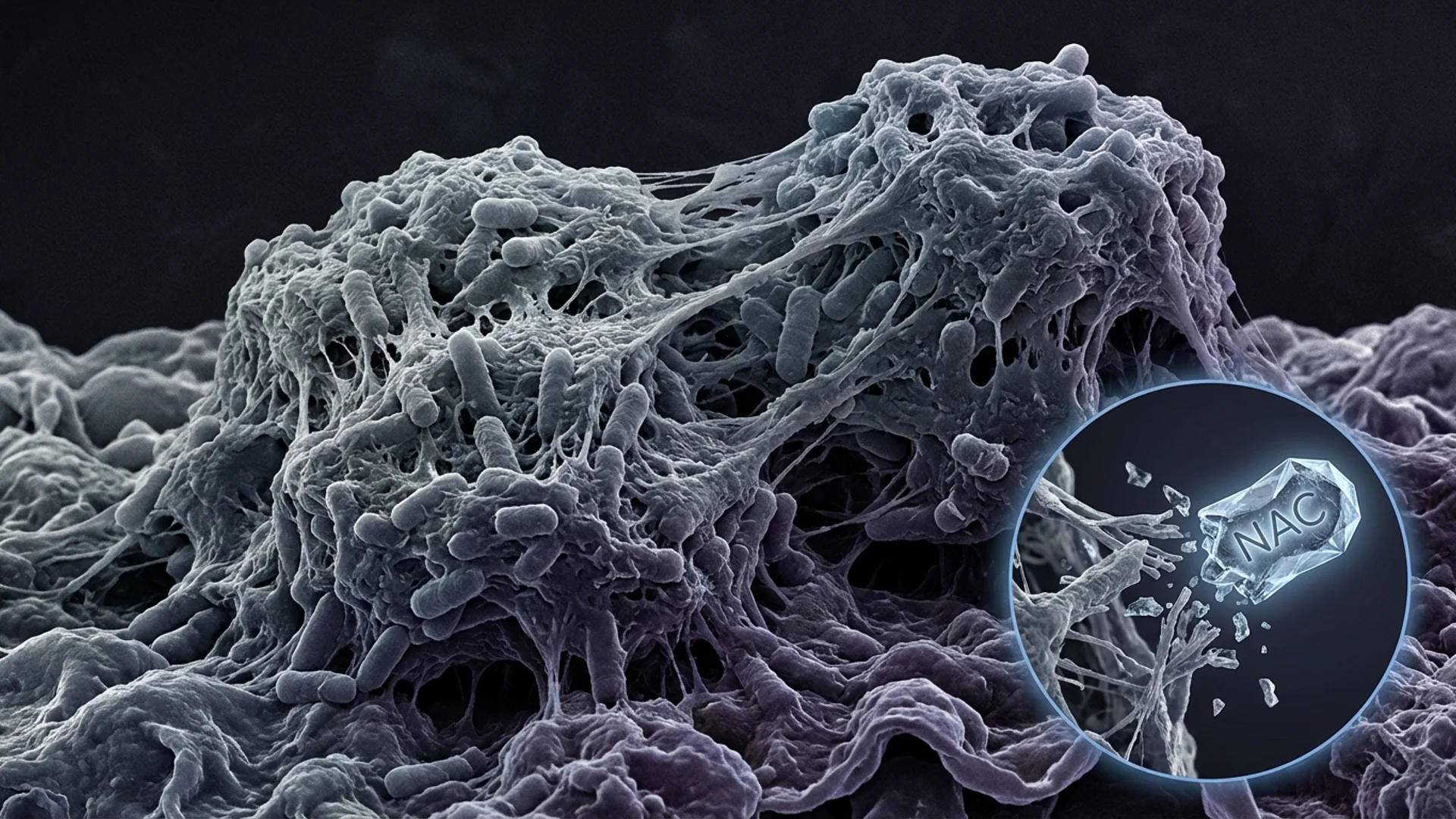
A biofilm is a structured community of microorganisms embedded in a self-produced extracellular matrix — a protective layer composed of polysaccharides, proteins, lipids, and extracellular DNA. Think of it as a fortress that bacteria build around themselves.

Within this matrix:
- Bacteria communicate via quorum sensing (chemical signaling) to coordinate behavior
- Nutrients are shared and distributed through channels
- Gene transfer occurs, including the transfer of antibiotic resistance genes
- Different organisms can coexist in polymicrobial communities (Borrelia alongside Bartonella, for example)
- Metabolic activity varies — bacteria at the core may enter a dormant, metabolically inactive state
Why Biofilms Matter Clinically
Antibiotic tolerance. Biofilm-embedded bacteria can tolerate antibiotic concentrations 100 to 1,000 times higher than planktonic bacteria of the same species [2]. This is not genetic resistance (the bacteria are not mutated) — it is phenotypic tolerance. The same bacterium, if released from the biofilm, would be fully susceptible to the antibiotic.
The mechanisms of this tolerance include:
- Physical barrier: the extracellular matrix impedes antibiotic penetration
- Metabolic dormancy: many antibiotics require active bacterial metabolism to work; dormant bacteria are effectively invisible to these drugs
- Efflux pumps: increased expression of drug efflux mechanisms within biofilms
- Persister cells: a subpopulation of cells within biofilms enters a dormant state specifically resistant to antibiotic killing
Immune evasion. The biofilm matrix protects organisms from immune cells. Neutrophils and macrophages cannot effectively penetrate or phagocytose biofilm-embedded bacteria. Antibodies bind to the biofilm surface but cannot reach the organisms within.
Chronic inflammation. The immune system recognizes biofilm components and mounts an inflammatory response — but because it cannot clear the organisms, the inflammation becomes chronic. This is one mechanism driving the persistent inflammation seen in chronic tick-borne disease.
Biofilms in Tick-Borne Disease
Borrelia burgdorferi forms biofilms both in vitro and in vivo. Studies have demonstrated Borrelia biofilm aggregates in human skin biopsies and in laboratory conditions [3]. These biofilm structures contain Borrelia in multiple morphological forms — spirochetes, round bodies, and cystic forms — creating a reservoir that standard antibiotic monotherapy is unlikely to fully address.
Bartonella species form biofilms, particularly on vascular endothelium, which may explain their persistence and vascular tropism.
Polymicrobial biofilms are common in the context of co-infections. Multiple pathogen species can coexist within a single biofilm, sharing resources and resistance mechanisms. This is one reason why comprehensive co-infection treatment is more effective than sequential, single-pathogen approaches.
Biofilm Disruption Strategies
In my clinical practice, biofilm disruption is an integral component of chronic infection treatment. The rationale is simple: if antibiotics cannot penetrate the biofilm effectively, we need to compromise the biofilm matrix to improve antibiotic access.
Enzymatic Disruption
NAC (N-acetylcysteine): Disrupts the polysaccharide component of biofilm matrices. Has demonstrated antibiofilm activity in vitro and is well-tolerated orally. Typical dose: 600-1,200 mg twice daily, taken away from antibiotic doses.
Serrapeptase and nattokinase: Proteolytic enzymes that degrade the protein components of biofilm matrices. The clinical evidence is primarily in vitro and in dental biofilm contexts, but the mechanistic rationale for broader application is sound. Taken on an empty stomach.
EDTA and lactoferrin: Chelate the metal ions (particularly iron and calcium) that stabilize biofilm structure. Used clinically in wound care and implant-associated infections.
Botanical Agents
Several botanicals have demonstrated antibiofilm activity in laboratory studies:
- Stevia leaf extract (whole leaf, not refined sweetener) has shown activity against Borrelia biofilms in vitro
- Cat’s claw (Uncaria tomentosa) has demonstrated anti-spirochetal and potential antibiofilm effects
- Oregano oil has broad antimicrobial and antibiofilm properties
I want to be transparent about the evidence level here. Most of these findings come from in vitro studies. Whether the concentrations achieved in vivo at relevant tissue sites are sufficient for meaningful biofilm disruption has not been established in controlled human trials. I use these agents as adjuncts to conventional antimicrobial therapy, not as replacements.
Physical Methods
Hyperthermia: Elevated temperature can disrupt biofilm integrity and increase antibiotic penetration. This is one of the mechanistic rationales for our use of therapeutic hyperthermia in chronic infection treatment programs.
Protocol Integration
In practice, I time biofilm disruption agents to precede antibiotic dosing by 30-60 minutes. The rationale is to compromise the biofilm matrix first, then deliver antibiotics to the newly exposed organisms. This is a logical approach based on the known biology, though it has not been validated in randomized controlled trials for tick-borne disease specifically.
The Clinical Implications
Understanding biofilms changes how we think about treatment failure. A patient who does not respond to a course of antibiotics may not have a “resistant” infection. They may have an infection that is fully antibiotic-susceptible but protected by a biofilm that prevents antibiotic access.
This reframing has practical consequences:
- Single-agent, short-course antibiotic therapy is unlikely to eradicate biofilm-associated chronic infections
- Combination approaches that address biofilm disruption alongside antimicrobial therapy are more rational
- Treatment duration matters — persistent biofilms require sustained therapeutic pressure
- Intermittent or pulsed dosing may be effective because organisms released from disrupted biofilms are susceptible to antibiotics
The Bottom Line
Biofilms are not a fringe concept. They are mainstream microbiology. The NIH estimates that biofilms account for over 80% of chronic infections. Understanding their role in chronic tick-borne disease — and incorporating biofilm disruption into treatment protocols — is essential for improving outcomes in patients who have not responded to conventional approaches.
References
- National Institutes of Health. Research on Microbial Biofilms. PA-03-047.
- Hoiby N, et al. Antibiotic resistance of bacterial biofilms. International Journal of Antimicrobial Agents. 2010;35(4):322-332.
- Sapi E, et al. Evidence of In Vivo Existence of Borrelia Biofilm in Borrelial Lymphocytomas. European Journal of Microbiology and Immunology. 2016;6(1):9-24.
This content is educational and does not constitute medical advice. Chronic infection treatment, including biofilm disruption strategies, should be managed by a physician experienced in complex infectious disease.