At a Glance
| Property | Value |
|---|---|
| Evidence Level | Emerging |
| Primary Use | Improving antimicrobial efficacy against biofilm-associated chronic infections |
| Key Mechanism | Sequential disruption of extracellular polymeric matrix followed by targeted antimicrobial therapy and toxin clearance |
Why Standard Treatment Misses Biofilm-Protected Infections
You have taken the antibiotics. You have followed the protocol. And yet the symptoms persist — or they improve temporarily, only to return when treatment stops. If this pattern sounds familiar, you are likely dealing with organisms that have learned to hide.
Here is what I tell my patients: one of the most important reasons antibiotics alone may fail against chronic infections like Lyme disease is biofilm formation. Borrelia burgdorferi, Bartonella, and many other organisms do not simply float around in your bloodstream waiting to be killed. They form structured communities protected by a self-produced extracellular matrix — a biofilm — that can reduce antibiotic penetration by 1,000-fold or more.
Breaking through that shield requires a deliberate, sequenced approach.
What a Biofilm Actually Is
A biofilm is not a wall. It is a community. Bacteria within a biofilm exist in a structured, three-dimensional architecture embedded in a self-produced extracellular polymeric substance (EPS) matrix composed of:
- Polysaccharides — the structural scaffold
- Extracellular DNA (eDNA) — provides structural integrity
- Proteins and glycoproteins — including fibrin and collagen-like proteins
- Lipids — contribute to the hydrophobic barrier
- Calcium, iron, and other minerals — provide structural rigidity and serve as metabolic resources
Within this matrix, organisms exist in different metabolic states — from actively dividing cells at the surface to metabolically dormant persister cells deep within the structure. This metabolic heterogeneity is precisely why single-agent antibiotic therapy fails: the dormant cells survive treatment and repopulate the biofilm when the antibiotic is discontinued [1].
In Lyme disease, biofilm formation by Borrelia burgdorferi has been demonstrated both in vitro and in vivo. Sapi et al. at the University of New Haven documented Borrelia biofilm formation containing all three morphological forms — spirochetes, round bodies, and biofilm aggregates — protected within an extracellular matrix containing alginate, a polysaccharide also found in Pseudomonas biofilms [2].

The Three-Phase Protocol
Phase 1: Disrupt the Matrix
The goal of the first phase is to degrade the protective EPS matrix, exposing the organisms within to antimicrobial agents. This phase uses enzymes that break down specific components of the biofilm matrix.
Proteolytic enzymes:
- Nattokinase and serrapeptase — degrade fibrin and protein components
- Lumbrokinase — fibrinolytic enzyme with additional protease activity
- Bromelain — pineapple-derived protease with anti-inflammatory properties
DNase (deoxyribonuclease):
- Targets extracellular DNA that provides structural integrity to biofilms
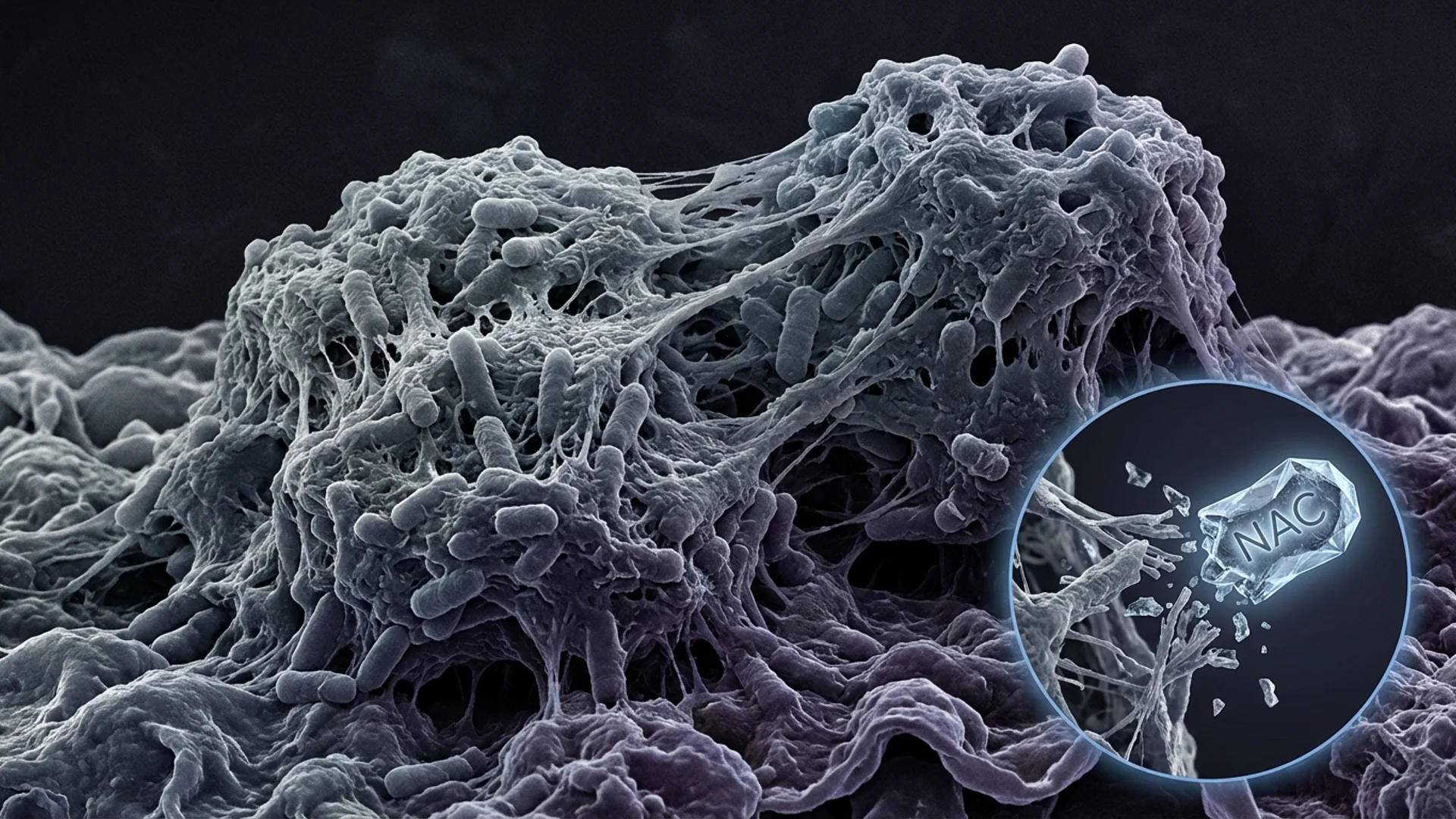
- NAC (N-acetylcysteine) has mucolytic properties that can disrupt the polysaccharide matrix and reduce eDNA-mediated structure
EDTA and chelating agents:
- Chelate calcium and other divalent cations essential for biofilm structural integrity
- EDTA at sub-inhibitory concentrations has been shown to disrupt biofilm matrix in multiple organisms
- Must be used cautiously — systemic chelation has its own risk profile
Timing: Disruption agents are taken on an empty stomach, typically 30-60 minutes before antimicrobials. The rationale is straightforward: degrade the shield first, then send in the killing agents while the organisms are exposed.
Duration of Phase 1: Typically 2-4 weeks of enzyme pre-treatment before adding antimicrobials, though some protocols use concurrent administration.
Phase 2: Kill the Exposed Organisms
Once the biofilm matrix is disrupted, antimicrobials can access the previously protected organisms. This phase combines conventional and, in some protocols, botanical antimicrobials.
Pharmaceutical options:
- Tinidazole or metronidazole — effective against anaerobic and cyst-form Borrelia within biofilms
- Doxycycline — tetracycline with demonstrated anti-biofilm properties in addition to antimicrobial activity
- Ceftriaxone — IV beta-lactam with CSF penetration for neurological involvement
- Azithromycin — concentrates intracellularly, relevant for intracellular co-infections
- Dapsone — emerging evidence for anti-persister activity in Borrelia (Horowitz protocol)
Botanical antimicrobials:
- Stevia rebaudiana whole-leaf extract — in vitro activity against Borrelia biofilm forms (University of New Haven, 2015)
- Cistus incanus — polyphenol-rich herb with biofilm-disrupting properties
- Cryptolepis sanguinolenta — activity against both planktonic and biofilm-associated organisms
Combination strategy: Single-agent antimicrobial therapy against biofilm infections is consistently less effective than combinations. The principle is to target organisms in different metabolic states simultaneously — actively dividing cells (doxycycline, ceftriaxone), intracellular forms (azithromycin), and persister/cyst forms (tinidazole, dapsone).
Phase 3: Clear the Debris
Biofilm disruption and organism killing release significant amounts of bacterial debris, endotoxins, and inflammatory mediators. This phase supports clearance of these toxins and manages the Herxheimer reaction that typically accompanies effective biofilm-targeted treatment.
Binders:
- Activated charcoal — broad-spectrum toxin binding
- Cholestyramine — bile acid sequestrant that binds biotoxins
- Modified citrus pectin — gentler binder option
- Bentonite clay — mineral-based binding
Detoxification support:
- Glutathione (liposomal or IV) — master antioxidant supporting hepatic detoxification
- NAC — glutathione precursor
- Epsom salt baths — magnesium sulfate transdermal absorption
- Infrared sauna — supports toxin elimination through sweat
Anti-inflammatory support:
- Curcumin (bioavailable formulation) — NF-kB modulation
- Omega-3 fatty acids — resolution of inflammation
- Adequate hydration — critical for renal clearance of toxins
Timing: Binders should be taken 2 hours apart from all other supplements and medications to avoid binding the therapeutic agents.
The Evidence
What We Know (Human Data)
The evidence for biofilm disruption protocols in Lyme disease is limited to case series, observational data, and extrapolation from biofilm research in other fields (wound care, cystic fibrosis, device-related infections).
NAC has been shown to disrupt biofilms in multiple clinical contexts. A randomized trial in chronic rhinosinusitis demonstrated that NAC irrigation significantly reduced biofilm burden compared to saline alone. In vitro studies specific to Borrelia have confirmed that NAC at clinically achievable concentrations reduces biofilm formation and enhances doxycycline penetration [3].
Combination antimicrobial protocols targeting Borrelia persisters have shown superior efficacy over single agents in laboratory models. Feng et al. at Johns Hopkins identified that daptomycin, cefoperazone, and doxycycline in combination eradicated Borrelia persisters in vitro — though this specific combination has not been tested in clinical trials [4].
What I See in Practice
In our clinical experience, adding biofilm disruption to antimicrobial protocols has improved treatment response in patients who previously failed standard antibiotic regimens. The improvement is not subtle — patients who were plateau’d on antibiotics alone often report a clear clinical shift within 2-4 weeks of adding enzyme-based disruption.
What I observe in practice is that the Herxheimer reactions during biofilm disruption phases tend to be more intense than during standard antibiotic therapy alone. I interpret this as evidence that previously protected organisms are being exposed and killed — which is the intended effect, but requires active management.
I also observe that patients with mold/CIRS need their detoxification pathways optimized before aggressive biofilm disruption. The toxin burden from biofilm disruption combined with existing biotoxin load can overwhelm Phase I/II hepatic detoxification, producing severe symptoms.

Practical Application
Sample Protocol Sequence
Weeks 1-2 (Pre-treatment — enzyme loading):
- Nattokinase 2,000 FU twice daily (empty stomach)
- Serrapeptase 120,000 SPU twice daily (empty stomach)
- NAC 600mg twice daily
- Binders: Activated charcoal 500mg at bedtime (away from other agents)
Weeks 3-12+ (Combined disruption + antimicrobials):
- Continue enzymes (morning, empty stomach)
- Add antimicrobials 60-90 minutes after enzymes (as prescribed by treating physician)
- Continue binders at bedtime
- Add glutathione support (liposomal 500mg or IV per clinical protocol)
- Monitor for Herxheimer reactions — adjust pace as needed
Ongoing (Cycling):
- Some clinicians cycle 4 weeks on / 2 weeks off to reduce bacterial adaptation
- Others maintain continuous treatment with periodic reassessment
- Treatment duration depends on clinical response and laboratory markers
Monitoring
- Symptom tracking with standardized questionnaire at each visit
- Liver function tests every 4-6 weeks during intensive treatment
- CBC and inflammatory markers (CRP, ESR)
- Lyme testing at 3-month intervals to assess treatment response
- C3a and C4a (complement markers) if available
Safety and Considerations
- Biofilm disruption can release significant pathogen burden simultaneously, producing severe Herxheimer reactions. Start slow and titrate.
- Enzyme supplements affect coagulation. Nattokinase and serrapeptase have fibrinolytic activity — avoid or use cautiously in patients on anticoagulants (warfarin, DOACs) or with bleeding disorders.
- EDTA chelation has significant risks including hypocalcemia and renal impairment. Only use under medical supervision with appropriate monitoring.
- Binders can reduce absorption of medications and supplements. Maintain a minimum 2-hour separation.
- This is an emerging treatment approach. Controlled clinical trials specific to Lyme biofilm disruption are needed. Evidence is derived from in vitro data, other biofilm-forming infection research, and clinical observation.
The Bottom Line
Biofilm disruption is a logical and increasingly evidence-supported addition to chronic infection treatment protocols. The three-phase approach — disrupt, kill, clear — addresses the fundamental reason why antibiotics alone often fail against persistent organisms. The nuance matters: timing, sequencing, and monitoring are critical. This is not a supplement regimen to add casually; it is a structured medical protocol that requires clinical supervision and individualized management.
References
- Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science. 1999;284(5418):1318-1322. PMID: 10334980
- Sapi E, Bastian SL, Mpoy CM, et al. Characterization of biofilm formation by Borrelia burgdorferi in vitro. PLoS One. 2012;7(10):e48277. PMID: 23110225
- Aslam S, Trautner BW, Ramanathan V, Darouiche RO. Combination of tigecycline and N-acetylcysteine reduces biofilm-embedded bacteria on vascular catheters. Antimicrob Agents Chemother. 2007;51(4):1556-1558. PMID: 17220422
- Feng J, Auwaerter PG, Zhang Y. Drug combinations against Borrelia burgdorferi persisters in vitro: eradication achieved by using daptomycin, cefoperazone and doxycycline. PLoS One. 2015;10(3):e0117207. PMID: 25806811