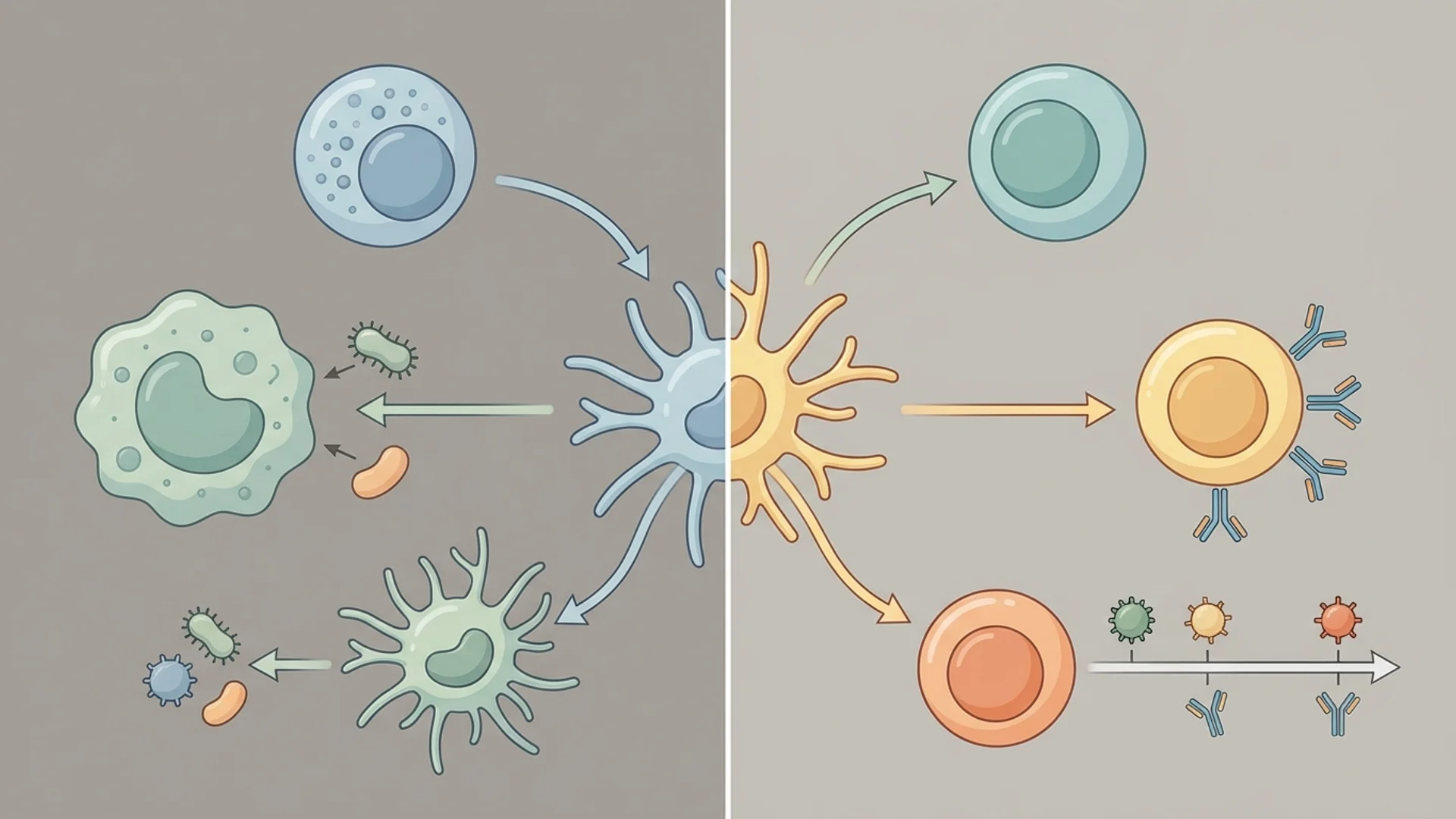
When a patient asks me how their immune system works, I begin with a simple analogy: you have two defense systems, not one. The first is fast, broad, and ready at all times. The second is slower, precise, and builds memory. Both are essential, and most immune dysfunction involves a failure of coordination between the two.
This is not a simplification for the sake of convenience. The innate-adaptive division is the organizing principle of immunology, and understanding it clarifies nearly everything else about immune function, immune disease, and immune treatment.
Innate Immunity: Speed Over Specificity
The innate immune system is evolutionarily ancient — conserved across insects, plants, and vertebrates. It activates within minutes of encountering a pathogen and does not require prior exposure.
How It Recognizes Threats
Innate immune cells carry pattern recognition receptors (PRRs), the most studied being toll-like receptors (TLRs). These receptors recognize pathogen-associated molecular patterns (PAMPs) — molecular structures common to broad categories of microorganisms but absent from human cells.
Examples include:
- Lipopolysaccharide (LPS) — a component of gram-negative bacterial cell walls, recognized by TLR4
- Flagellin — bacterial flagellum protein, recognized by TLR5
- Double-stranded RNA — a hallmark of viral replication, recognized by TLR3
- Unmethylated CpG DNA — bacterial DNA pattern, recognized by TLR9
The innate system also recognizes damage-associated molecular patterns (DAMPs) — molecules released by damaged or dying host cells. This is why tissue injury, even without infection, produces an inflammatory response.
Key Innate Immune Cells
Neutrophils are the most abundant white blood cells and the first responders to bacterial infection. They live only hours to days and kill pathogens through phagocytosis, degranulation, and the formation of neutrophil extracellular traps (NETs). A low neutrophil count is one of the most significant risk factors for bacterial infection.
Macrophages are longer-lived phagocytes that reside in tissues throughout the body. They engulf pathogens, process antigens, and secrete cytokines that recruit other immune cells and influence the adaptive response. Macrophages are remarkably plastic cells — they can polarize toward pro-inflammatory (M1) or anti-inflammatory (M2) states depending on their environment.
Natural killer (NK) cells patrol the body looking for cells that have lost normal surface markers — a sign of viral infection or malignant transformation. Unlike cytotoxic T cells, NK cells do not require prior sensitization. They can kill on first contact. NK cell activity is one of the functional immune markers I check frequently in clinical practice.
Dendritic cells are the critical bridge between innate and adaptive immunity. They capture antigens at sites of infection, migrate to lymph nodes, and present those antigens to T cells, initiating the adaptive response. Without dendritic cells, the adaptive immune system would be largely blind to threats.
The Complement System
The complement system is a cascade of over 30 proteins that can be activated through three pathways (classical, lectin, and alternative). Once activated, complement proteins can directly lyse pathogen membranes, coat pathogens for enhanced phagocytosis (opsonization), and recruit immune cells through the release of anaphylatoxins.
Complement deficiencies, though rare, dramatically illustrate the system’s importance — patients with complement deficiencies are highly susceptible to recurrent bacterial infections, particularly Neisseria species.
Adaptive Immunity: Precision and Memory
The adaptive immune system is unique to vertebrates and requires days to weeks for a primary response but offers two advantages the innate system lacks: exquisite specificity and immunological memory.
T Cells: Cellular Immunity
T cells mature in the thymus (hence the “T”) and undergo a selection process that ensures they can recognize foreign antigens but do not react to self-antigens. This process eliminates approximately 95% of developing T cells — a necessary quality control measure.
Three major T cell populations:
Helper T cells (CD4+) coordinate the immune response by releasing cytokines that activate other immune cells. Different helper T cell subtypes (Th1, Th2, Th17, Tfh) produce different cytokine profiles and are suited to different types of threats. Th1 responses are directed against intracellular pathogens. Th2 responses target parasites and are involved in allergic disease. Th17 responses are important for mucosal defense but, when excessive, contribute to autoimmunity.
Cytotoxic T cells (CD8+) directly kill cells displaying foreign antigens on their surface — virus-infected cells, cancer cells, or transplanted cells. They are the adaptive system’s assassins, and their specificity is remarkable: a cytotoxic T cell recognizes a specific peptide fragment presented by a specific MHC molecule.
Regulatory T cells (Tregs) suppress immune responses and prevent autoimmunity. They are the immune system’s brakes. Treg dysfunction is implicated in virtually every autoimmune disease, and restoring Treg function is an active area of therapeutic research.
B Cells: Humoral Immunity
B cells produce antibodies (immunoglobulins) — soluble proteins that bind to specific antigens. There are five classes of antibody:
- IgM — the first antibody produced in an immune response, effective at complement activation
- IgG — the most abundant antibody in blood, crosses the placenta, provides long-term protection
- IgA — the dominant antibody at mucosal surfaces, critical for gut and respiratory defense
- IgE — involved in allergic responses and parasite defense
- IgD — functions still being elucidated
When a B cell encounters its target antigen and receives appropriate T cell help, it can undergo affinity maturation — a process that refines the antibody’s binding precision through somatic hypermutation in germinal centers. The result is antibodies that bind with extraordinary specificity and high affinity.
Immunological Memory
The defining feature of adaptive immunity is memory. After an immune response resolves, a subset of T and B cells persist as memory cells. These cells can survive for decades, and upon re-encounter with the same pathogen, they mount a secondary response that is faster (hours to days versus days to weeks), stronger (higher antibody titers, more effective T cell responses), and more precisely targeted.
This is why you rarely get the same childhood illness twice, and it is the biological basis for vaccination.
How the Two Systems Cooperate
The innate-adaptive distinction is useful for teaching, but in reality, the two systems operate as an integrated network.
Innate cells activate adaptive responses. Dendritic cells capture antigens and present them to T cells, providing the signals necessary for T cell activation. Without this innate input, adaptive responses do not initiate properly.
Adaptive responses enhance innate function. Antibodies coat pathogens (opsonization), making them more recognizable to macrophages and neutrophils. Th1 cytokines activate macrophages to kill intracellular pathogens more effectively. The adaptive system essentially provides targeting information that the innate system uses.
Innate responses shape the type of adaptive response. The cytokine environment created by the innate response during the initial encounter with a pathogen determines whether the adaptive response skews toward Th1, Th2, or Th17. This is not a trivial point — the wrong type of adaptive response can be worse than no response at all.
Clinical Relevance
Understanding this two-system architecture has direct clinical implications:
Chronic infections like Lyme disease often involve pathogens that have evolved mechanisms to evade both systems — hiding intracellularly to avoid antibodies, downregulating surface markers to avoid T cell recognition, and suppressing innate signaling.
Autoimmune disease represents a failure of the selection and regulation mechanisms that normally prevent adaptive immune cells from attacking self. Restoring this tolerance is the central challenge in autoimmune treatment.
Cancer evades immune surveillance through multiple mechanisms, including downregulating MHC molecules (making tumor cells invisible to T cells), recruiting Tregs to suppress anti-tumor immunity, and creating an immunosuppressive microenvironment.
Aging (immunosenescence) affects both systems but is particularly damaging to the adaptive system through thymic involution and the accumulation of exhausted, senescent T cells.
In my practice, I assess both arms of the immune system routinely. A standard CBC provides limited information. A lymphocyte subset panel with functional assessments tells me far more about what is actually happening in a patient’s immune system and where intervention is needed.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical care. Consult a qualified physician before making decisions about your health based on this information.