You react to foods you used to tolerate without issue. Your skin flushes unpredictably. Your gut alternates between cramping and bloating. You get headaches, brain fog, heart palpitations, and a feeling of being “inflamed” that defies a simple diagnosis. You have been told it is anxiety, IBS, or that nothing is wrong.
If this sounds familiar, you may be dealing with Mast Cell Activation Syndrome — MCAS — and what I can tell you is that it is real, it is diagnosable, and it is treatable.
In my clinical experience, MCAS is one of the most under-recognized conditions in medicine. It is also one of the most frustrating for patients, because the symptoms are so varied that they seem unrelated. A patient who presents with flushing, abdominal pain, tachycardia, and brain fog might see a dermatologist, a gastroenterologist, a cardiologist, and a neurologist — and get four separate diagnoses that miss the unifying mechanism.
The unifying mechanism is the mast cell. Here is what you need to know.
At a Glance
| Property | Value |
|---|---|
| Evidence Level | Emerging to Moderate |
| Primary Use | Diagnosis and management of mast cell hyperactivation |
| Key Mechanism | Excessive mast cell degranulation releasing histamine, tryptase, prostaglandins, leukotrienes |
| Common Overlap | Lyme disease, mold/CIRS, EBV, post-COVID, Ehlers-Danlos Syndrome |
| Treatment Approach | Trigger avoidance, H1/H2 antihistamines, mast cell stabilizers, dietary modification, treat underlying drivers |
What Mast Cells Do — And What Goes Wrong
Mast cells are immune sentinel cells derived from the bone marrow and distributed throughout virtually every tissue in the body — particularly concentrated in skin, gut mucosa, respiratory epithelium, and perivascular connective tissue. They are positioned at the interfaces between your body and the environment.
In normal function, mast cells serve as first responders. When they detect a pathogen, toxin, or physical injury, they degranulate — releasing preformed mediators (histamine, tryptase, heparin) and synthesizing new mediators (prostaglandins, leukotrienes, cytokines) that initiate the inflammatory and immune response. This is appropriate and necessary.
In MCAS, this system becomes dysregulated. Mast cells degranulate excessively and in response to stimuli that should not trigger activation — certain foods, temperature changes, emotional stress, physical pressure, fragrances, or even endogenous hormone fluctuations. The mast cells are not malignant (that would be mastocytosis, a different and much rarer condition). They are structurally normal but functionally hyperreactive.
The result is a chronic, fluctuating state of inappropriate inflammation driven by mediator release. Because mast cells are distributed throughout the body, symptoms can manifest in virtually any organ system — which is exactly why MCAS mimics and overlaps with dozens of other conditions.
The Mediator Cascade
When a mast cell degranulates, it releases a complex mixture of mediators. Understanding these mediators explains the diversity of symptoms:
Histamine: The most well-known mast cell mediator. Histamine acts on four receptor types (H1 through H4), producing effects that include vasodilation (flushing), increased vascular permeability (edema, hives), smooth muscle contraction (bronchospasm, GI cramping), gastric acid secretion, and central nervous system effects (headache, brain fog, anxiety).
Tryptase: A serine protease that is relatively specific to mast cells. Tryptase activates complement, degrades fibrinogen, and contributes to tissue remodeling. Elevated serum tryptase is one of the diagnostic markers for mast cell activation.
Prostaglandins (especially PGD2): Prostaglandin D2 causes vasodilation, bronchoconstriction, and inhibits platelet aggregation. It is also a potent mediator of flushing. PGD2 is metabolized to 11-beta-prostaglandin F2-alpha, which can be measured in urine as a marker of mast cell activation.
Leukotrienes (LTC4, LTD4, LTE4): Potent bronchoconstrictors and promoters of mucous secretion. Leukotriene E4 can be measured in urine.
Cytokines (IL-6, TNF-alpha, IL-1beta): Mast cell-derived cytokines contribute to systemic inflammation, fatigue, and the constitutional symptoms that make MCAS patients feel chronically unwell.
Heparin: Can contribute to unexplained bruising and, paradoxically, both bleeding and clotting tendencies.
Symptoms: Why MCAS Looks Like Everything
The symptom profile of MCAS is broad precisely because mast cells are everywhere and their mediators affect every organ system. Here is what I see in my patients:
Skin
- Flushing (face, chest, neck — often without an obvious trigger)
- Urticaria (hives) — can be dermographic (appearing where the skin is scratched or pressed)
- Angioedema (deep tissue swelling, often around the eyes or lips)
- Itching without visible rash
- Unexplained bruising
Gastrointestinal
- Abdominal cramping and pain
- Diarrhea, sometimes alternating with constipation
- Nausea
- Bloating and distension
- Food intolerances — often multiple and fluctuating
- Gastroesophageal reflux
Cardiovascular
- Tachycardia (often episodic, may meet criteria for POTS)
- Hypotension or blood pressure instability
- Palpitations
- Lightheadedness, especially postural
- Pre-syncope or syncope
Neurological
- Brain fog — difficulty with concentration, word-finding, and processing speed
- Headaches, often migrainous
- Anxiety and panic-like episodes (mediator-driven, not primary psychiatric)
- Insomnia
- Peripheral neuropathic symptoms (tingling, numbness)
Respiratory
- Nasal congestion and rhinitis
- Wheezing or bronchospasm
- Throat tightness
- Chronic cough
Musculoskeletal
- Diffuse pain
- Joint pain without structural abnormality
- Muscle aches
Constitutional
- Fatigue — often severe and disproportionate to activity
- Temperature dysregulation
- Sensitivity to heat, cold, or rapid temperature changes
- Sensitivity to chemicals, fragrances, and environmental exposures
The hallmark pattern is episodic, multi-system symptoms that fluctuate in intensity, are often triggered by identifiable stimuli (but not always), and do not fit neatly into any single organ-specific diagnosis. The patient who has been to 6 specialists and received 6 different diagnoses — irritable bowel syndrome from the gastroenterologist, anxiety from the psychiatrist, chronic urticaria from the dermatologist, POTS from the cardiologist — may have MCAS as the unifying explanation.
The MCAS-Infection Connection
This is where my clinical experience adds important context that you will not find in most MCAS literature.
MCAS and Lyme Disease
In my practice treating over 12,000 Lyme patients, I see MCAS as a frequent complication — not a coincidence, but a mechanistic consequence.
Borrelia burgdorferi (and its co-infections) directly activates mast cells through multiple pathways: toll-like receptor signaling, complement activation, and direct interaction with mast cell surface receptors [1]. Chronic infection produces chronic mast cell activation. Over time, this sustained activation appears to lower the threshold for degranulation — the mast cells become permanently sensitized.
What I observe in practice is that many patients diagnosed with MCAS actually have underlying chronic Lyme disease or tick-borne co-infections driving the mast cell hyperactivation. Treating the underlying infection often reduces or resolves the mast cell symptoms. This is critically important, because MCAS treatment without addressing the infectious trigger produces only partial improvement.
MCAS and Mold/CIRS
Chronic Inflammatory Response Syndrome (CIRS) from mold and biotoxin exposure is another potent driver of mast cell activation. Mold-derived mycotoxins directly trigger mast cell degranulation and amplify the innate immune inflammatory response [2].
Patients with both mold exposure and MCAS present with particularly severe symptom profiles. The combination of mycotoxin-driven inflammation and mast cell mediator release creates a compounding effect. Treatment must address both the mold exposure (removal from the environment, binder protocols, detoxification) and the mast cell activation simultaneously.
MCAS and EBV Reactivation
Epstein-Barr virus reactivation — which I see frequently in immunocompromised patients, including those with chronic Lyme, post-COVID, and chronic fatigue — can trigger and sustain mast cell activation. EBV infects B cells and can produce immune dysregulation that includes mast cell hyperreactivity.
MCAS and Post-COVID
A significant subset of post-COVID patients develop MCAS or MCAS-like symptoms. The mechanism likely involves spike protein interaction with mast cells, complement activation, and immune dysregulation. Patients with pre-existing MCAS tendencies appear to be at higher risk for developing Long COVID — a connection that is being actively researched [3].
Diagnostic Criteria
MCAS diagnosis requires three criteria to be met simultaneously. This is important — no single criterion alone is sufficient:
1. Typical Symptoms
Episodic symptoms consistent with mast cell mediator release affecting two or more organ systems. The symptoms must be recurrent, cannot be explained by another diagnosis, and should follow a pattern consistent with mediator release (episodic, triggered, and involving the organ systems where mast cells are concentrated).
2. Biochemical Evidence of Mast Cell Mediator Elevation
Laboratory documentation of elevated mast cell mediators during or shortly after a symptomatic episode:
Serum tryptase: The most commonly measured marker. An increase of 20% plus 2 ng/mL above the patient’s baseline during a symptomatic episode is considered significant. Note: baseline tryptase in MCAS is often normal (unlike mastocytosis, where it is persistently elevated). The key is the rise from baseline during episodes.
24-hour urine N-methylhistamine: Elevated histamine metabolites in timed urine collection.
24-hour urine prostaglandin D2 metabolites (11-beta-PGF2-alpha): Elevated prostaglandin metabolites. This is often the most sensitive marker.
24-hour urine leukotriene E4: Elevated in some MCAS patients.
Plasma histamine: Difficult to measure reliably due to rapid metabolism, but can be informative when properly collected (chilled specimen, rapid processing).
Practical challenge: The diagnostic testing is technically demanding. Specimens must be collected during or within hours of a symptomatic flare. Improper collection, delayed processing, or testing during asymptomatic periods produces false negatives. I tell patients: a negative mediator test during an asymptomatic period does not rule out MCAS. You must test during a flare.
3. Response to Mast Cell-Targeted Treatment
Symptomatic improvement with medications that target mast cell mediators or mast cell activation (antihistamines, mast cell stabilizers). This is both a diagnostic criterion and part of the treatment approach.
Treatment: A Layered Approach
MCAS treatment is a layered strategy. You build from the foundation up, adding interventions based on response.
Layer 1: Trigger Identification and Avoidance
Before adding medications, identify and reduce triggers. Common triggers include:
- Dietary: High-histamine foods (aged cheese, fermented foods, cured meats, alcohol, vinegar, certain fish), histamine-releasing foods (citrus, strawberries, tomatoes, shellfish), and individual sensitivities identified through careful tracking
- Environmental: Fragrances, cleaning chemicals, mold, temperature extremes, cigarette smoke
- Physical: Friction, pressure, vibration, exercise (particularly in heat), rapid temperature changes
- Emotional: Acute stress, anxiety (which both triggers and is triggered by mast cell activation — a vicious cycle)
- Hormonal: Menstrual cycle fluctuations (estrogen promotes mast cell degranulation), thyroid dysfunction
A low-histamine elimination diet — maintained for 2-4 weeks — is often the most informative initial step. If symptoms improve significantly on a low-histamine diet, this supports the diagnosis and identifies a modifiable trigger.
Layer 2: H1 and H2 Antihistamines
The foundation of pharmacological MCAS treatment:
H1 blockers: Cetirizine (Zyrtec) or loratadine (Claritin) are first-line. Second-generation antihistamines are preferred over first-generation (diphenhydramine) due to fewer CNS side effects. Dosing often needs to exceed standard allergy doses — many MCAS patients require twice-daily dosing or higher than typical doses, titrated under physician guidance.
H2 blockers: Famotidine (Pepcid) is the standard choice. H2 receptors are not just in the stomach — they are present on mast cells and in the CNS. H2 blockers reduce GI symptoms, may reduce flushing, and appear to have mast cell-stabilizing properties at higher doses.
The combination of H1 and H2 blockers is more effective than either alone. This is first-line treatment and should be tried before anything else.
Layer 3: Mast Cell Stabilizers
If antihistamines provide partial but insufficient relief:
Cromolyn sodium (oral): A mast cell stabilizer that works primarily in the GI tract (poorly absorbed systemically). Particularly useful for GI-predominant MCAS. Dose: 100-200mg before meals, titrated gradually.
Ketotifen: Both an antihistamine and a mast cell stabilizer. Available as an ophthalmic preparation in many countries; oral formulation available in some countries (not FDA-approved in the US but available through compounding pharmacies). Often effective for patients who do not respond adequately to standard antihistamines.
Quercetin: A flavonoid with mast cell-stabilizing properties. The evidence is predominantly preclinical, but clinical observation supports its use as an adjunct. Typical dose: 500-1000mg twice daily. Bioavailability-enhanced formulations (phytosome or liposomal) are preferred [4].
Layer 4: Leukotriene Inhibitors and Additional Agents
For patients with persistent symptoms despite layers 1-3:
Montelukast (Singulair): A leukotriene receptor antagonist. Useful for patients with prominent respiratory or flushing symptoms. Some patients report significant benefit; others see minimal effect.
Low-dose naltrexone (LDN): Emerging evidence for immune modulation and potential mast cell stabilization at doses of 1.5-4.5mg nightly. The mechanism is thought to involve endorphin upregulation and microglial modulation. The evidence is limited but the safety profile is favorable.
Vitamin C: Acts as a cofactor for the enzyme (diamine oxidase) that degrades histamine. High-dose vitamin C (1-2g daily or IV) may support histamine clearance.
Diamine oxidase (DAO) supplementation: Taken before meals, DAO supplements help break down dietary histamine in the gut. Useful for patients with prominent dietary triggers.
Layer 5: Address the Underlying Driver
This is where MCAS treatment differs from MCAS management. Layers 1-4 manage symptoms. Layer 5 addresses why the mast cells became hyperreactive in the first place.
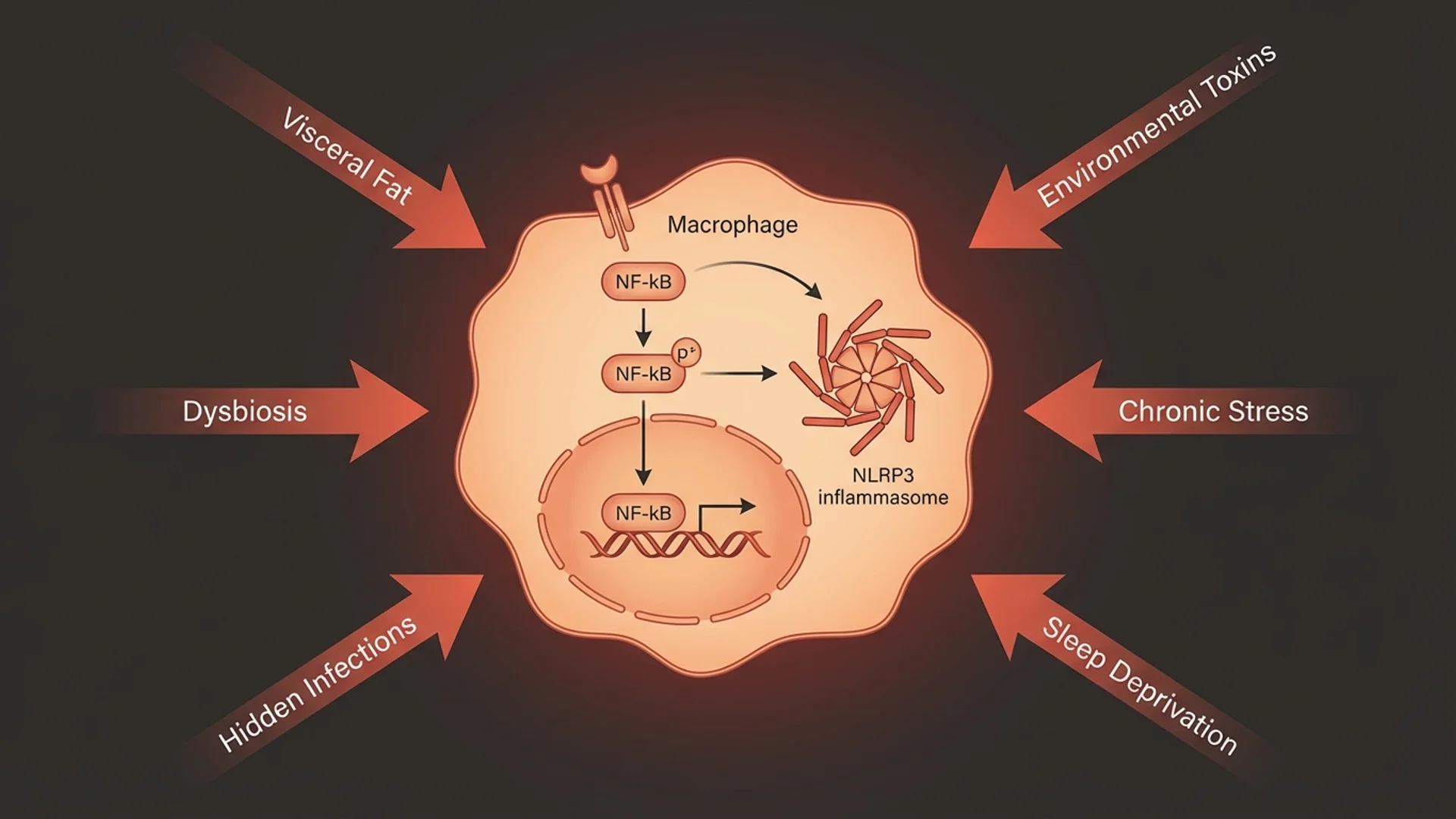
In my clinical experience, the most common underlying drivers are:
- Chronic infections: Lyme disease, Bartonella, Babesia, chronic EBV, mold/CIRS
- Gut dysbiosis: SIBO, candida overgrowth, parasitic infection, intestinal permeability
- Environmental toxins: Mold exposure, heavy metal burden
- Autoimmune activation: Particularly when MCAS co-occurs with autoimmune conditions
Treating the underlying driver is what separates long-term improvement from indefinite symptom management. A patient with MCAS driven by chronic Lyme disease who receives only antihistamines will improve partially but never resolve. Address the Lyme infection, and the mast cell activation often quiets substantially.
The Histamine Intolerance Overlap
Histamine intolerance and MCAS are related but not identical.
Histamine intolerance refers to symptoms caused by an inability to adequately degrade histamine — typically due to reduced diamine oxidase (DAO) activity in the gut. These patients react to high-histamine foods and beverages. The source of the histamine is external (dietary).
MCAS involves endogenous histamine release from the patient’s own mast cells, in addition to potential dietary histamine sensitivity. MCAS patients produce too much histamine; histamine intolerance patients cannot clear normal amounts.
In practice, many patients have both — endogenous mast cell activation and impaired histamine degradation. The dietary management overlaps (low-histamine diet), but the pharmacological approach differs. MCAS requires mast cell-targeted therapy; isolated histamine intolerance may respond to DAO supplementation and dietary modification alone.
What I See in Practice
After years of treating patients with MCAS in the context of complex, multi-system illness, the patterns are clear:
MCAS is almost never the primary diagnosis. It is almost always a downstream consequence of something else — an infection, a toxin exposure, a gut problem, an autoimmune process. The mast cells are reacting to a real problem. Finding and addressing that problem is the difference between managing symptoms and achieving resolution.
The patients who improve most are those who commit to a comprehensive approach: dietary modification, layered pharmacotherapy, and investigation and treatment of underlying drivers. There are no shortcuts.
Symptom fluctuation is normal. MCAS symptoms wax and wane. Good days and bad days are expected, especially early in treatment. Progress is measured over weeks and months, not days.
Environmental control matters more than patients expect. Removing mold from the home environment, reducing chemical exposures, and optimizing air quality often produce more improvement than adding another medication.
The Bottom Line
MCAS is a real, diagnosable condition driven by hyperreactive mast cells releasing inflammatory mediators in response to stimuli that should not trigger such a response. The symptoms are multi-system, episodic, and frequently misdiagnosed. Diagnosis requires symptoms, biochemical evidence of mediator elevation, and response to mast cell-targeted treatment.
Treatment is layered: trigger avoidance, antihistamines (H1 and H2), mast cell stabilizers, and — critically — identification and treatment of the underlying driver. In my clinical experience, MCAS in isolation is uncommon. MCAS in the context of chronic infection, mold exposure, or post-infectious immune dysregulation is the rule, not the exception.
If your symptoms span multiple organ systems, fluctuate unpredictably, and have resisted diagnosis despite thorough specialty evaluation — ask about MCAS. The nuance matters.

References
- Talkington J, Nickell SP. Borrelia burgdorferi spirochetes induce mast cell activation and cytokine release. Infection and Immunity. 1999;67(3):1107-1115. doi:10.1128/IAI.67.3.1107-1115.1999
- Kritas SK, et al. Mast cells contribute to the pathogenesis of chronic inflammatory conditions. Journal of Biological Regulators and Homeostatic Agents. 2018;32(3):467-475. PMID:29921373
- Weinstock LB, et al. Mast cell activation symptoms are prevalent in Long-COVID. International Journal of Infectious Diseases. 2021;112:217-226. doi:10.1016/j.ijid.2021.09.043
- Weng Z, et al. Quercetin is more effective than cromolyn in blocking human mast cell cytokine release and inhibits contact dermatitis and photosensitivity in humans. PLoS ONE. 2012;7(3):e33805. doi:10.1371/journal.pone.0033805
- Afrin LB, et al. Diagnosis of mast cell activation syndrome: a global “consensus-2.” Diagnosis. 2021;8(2):137-152. doi:10.1515/dx-2020-0005
- Molderings GJ, et al. Mast cell activation disease: a concise practical guide for diagnostic workup and therapeutic options. Journal of Hematology & Oncology. 2011;4:10. doi:10.1186/1756-8722-4-10
This content is educational and does not constitute medical advice. MCAS requires individualized medical evaluation and treatment.