Acute inflammation is essential. It is the immune system’s response to injury and infection — vasodilation, immune cell recruitment, pathogen destruction, tissue repair. Without it, a simple wound could be fatal.
Chronic inflammation is a different phenomenon entirely. It is low-grade, systemic, persistent, and largely silent. It does not produce the redness, swelling, and pain of acute inflammation. Instead, it smolders beneath the threshold of clinical detection, driving tissue damage over years and decades.
Here is what the evidence shows: chronic inflammation is implicated as a contributing factor in cardiovascular disease, type 2 diabetes, neurodegenerative disease (Alzheimer’s, Parkinson’s), cancer, depression, chronic fatigue, and accelerated aging. It is arguably the most significant modifiable driver of chronic disease.
The Molecular Machinery
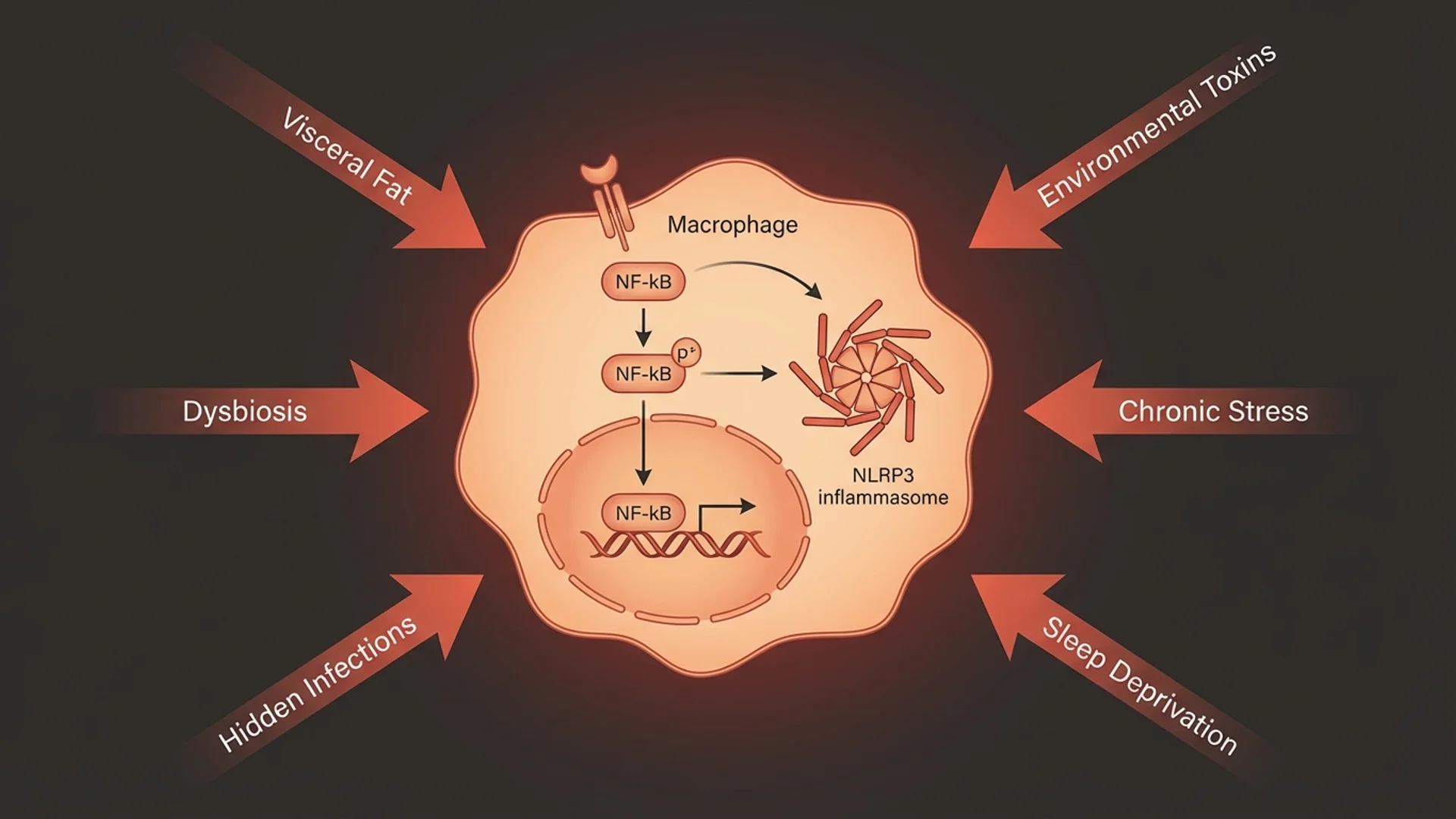
NF-kB: The Master Switch
Nuclear factor kappa-B (NF-kB) is the central transcription factor that controls inflammatory gene expression. When activated, NF-kB translocates to the nucleus and upregulates the production of pro-inflammatory cytokines (TNF-alpha, IL-1beta, IL-6), adhesion molecules, enzymes (COX-2, iNOS), and chemokines.
In acute inflammation, NF-kB activation is self-limiting. In chronic inflammation, the pathway remains constitutively active, driven by persistent stimuli. Understanding NF-kB is important because many effective anti-inflammatory interventions — from omega-3 fatty acids to curcumin to exercise — work at least partially through NF-kB inhibition.
Pro-Inflammatory Cytokines
The cytokines most consistently elevated in chronic inflammatory states include:
- TNF-alpha — drives systemic inflammation, insulin resistance, and tissue destruction
- IL-6 — has both pro- and anti-inflammatory properties depending on context; chronically elevated IL-6 is associated with cardiovascular risk and depression
- IL-1beta — activates the inflammasome pathway; elevated in metabolic syndrome and autoinflammatory conditions
- CRP (C-reactive protein) — not a cytokine but an acute phase protein produced by the liver in response to IL-6; widely used as a clinical marker of systemic inflammation
The Inflammasome
The NLRP3 inflammasome is an intracellular signaling complex that, when activated, triggers the release of IL-1beta and IL-18. NLRP3 activation has been implicated in atherosclerosis, type 2 diabetes, gout, Alzheimer’s disease, and age-related macular degeneration.
What activates NLRP3? A range of stimuli including cholesterol crystals, uric acid crystals, glucose, palmitate (a saturated fat), silica, asbestos, and amyloid-beta. The breadth of activating stimuli helps explain why chronic inflammation is associated with such a diverse range of diseases.
What Drives Chronic Inflammation
Visceral Adiposity
Adipose tissue is not an inert storage depot. It is an endocrine organ that produces adipokines, and visceral (abdominal) fat is particularly metabolically active. Excess visceral fat produces TNF-alpha, IL-6, and leptin while reducing production of the anti-inflammatory adipokine adiponectin.
This is one of the primary reasons obesity is associated with virtually every inflammatory disease. The adipose tissue itself is a source of chronic inflammatory signaling. Evidence level: robust human data from multiple large studies.
Gut Dysbiosis and Barrier Dysfunction
Lipopolysaccharide (LPS) from gram-negative gut bacteria that crosses a compromised intestinal barrier activates TLR4 on immune cells, triggering NF-kB activation and pro-inflammatory cytokine release. This process — metabolic endotoxemia — has been documented in controlled human studies and is associated with metabolic syndrome, depression, and cardiovascular risk.
Chronic Infections
Persistent infections maintain chronic immune activation. This is particularly relevant in my practice: patients with chronic Lyme disease, Epstein-Barr virus reactivation, or chronic periodontal disease often have persistently elevated inflammatory markers driven by ongoing immune stimulation.
Environmental Toxins
Heavy metals (mercury, lead, cadmium), air pollution (particulate matter, nitrogen dioxide), and pesticides activate inflammatory pathways through oxidative stress and direct immune cell stimulation. The evidence for air pollution as a driver of systemic inflammation is Level 1 (large epidemiological studies with mechanistic confirmation).
Chronic Psychological Stress
Chronic stress activates the hypothalamic-pituitary-adrenal (HPA) axis, leading to cortisol dysregulation. While acute cortisol is anti-inflammatory, chronic cortisol exposure produces glucocorticoid resistance in immune cells — the cells stop responding to cortisol’s anti-inflammatory signal, and inflammatory pathways become unrestrained.
This is one of the most well-characterized pathways linking psychological stress to physical disease. The evidence is Level 1 (controlled human studies and meta-analyses).
Sleep Deprivation
Even partial sleep deprivation (sleeping six hours instead of eight) increases CRP, IL-6, and TNF-alpha levels. The effect is rapid — measurable after a single night of poor sleep — and cumulative. Evidence level: controlled human studies (multiple).
Diet
The standard Western diet — high in refined carbohydrates, seed oils, processed meats, and sugar — is pro-inflammatory through multiple mechanisms: direct NF-kB activation by glucose spikes, omega-6/omega-3 imbalance promoting pro-inflammatory eicosanoid production, and gut microbiome disruption.
Measuring Inflammation
Standard Markers
- hs-CRP (high-sensitivity C-reactive protein) — the most widely available inflammatory marker. Optimal is below 1.0 mg/L; above 3.0 mg/L indicates significant systemic inflammation.
- ESR (erythrocyte sedimentation rate) — less specific but useful in combination with CRP.
- Fasting insulin — elevated fasting insulin is both a consequence and driver of inflammation. Often overlooked in standard panels.
Advanced Markers
- Cytokine panels (TNF-alpha, IL-6, IL-10) — provide more specific information about the type and balance of inflammatory signaling.
- Homocysteine — elevated levels indicate methylation dysfunction and are associated with cardiovascular inflammation.
- Fibrinogen — an acute phase protein that contributes to the hypercoagulable state associated with chronic inflammation.
- Fecal calprotectin — specific to intestinal inflammation.
- LPS-binding protein — a marker of endotoxemia from gut barrier dysfunction.
In my practice, I assess inflammatory status using a combination of these markers, not relying on CRP alone. A patient can have a normal CRP and still have significant compartmentalized inflammation — in the gut, the brain, or within specific tissues.
Addressing Chronic Inflammation
The most effective approach addresses multiple drivers simultaneously. Targeting a single pathway while ignoring others produces incomplete results.
Foundational interventions (strongest evidence):
- Anti-inflammatory dietary pattern (Mediterranean diet has Level 1 evidence for CRP reduction)
- Regular moderate exercise (Level 1 evidence for anti-inflammatory effects)
- Sleep optimization (7-9 hours, consistent timing)
- Stress management (measurable reductions in inflammatory markers with meditation, yoga, and cognitive behavioral therapy)
- Weight management (visceral fat reduction is one of the most impactful anti-inflammatory interventions)
Targeted supplementation (moderate to strong evidence):
- Omega-3 fatty acids (EPA/DHA, 2-4 g daily) — RCTs demonstrate CRP and cytokine reduction
- Curcumin (400-1000 mg daily, bioavailable formulation) — RCTs demonstrate NF-kB inhibition and CRP reduction
- Vitamin D (optimize to 40-60 ng/mL) — RCTs demonstrate anti-inflammatory effects
- Magnesium (400 mg daily) — associated with lower CRP in controlled studies
Advanced interventions (clinical context required):
- IV therapies (vitamin C, glutathione) for acute inflammatory episodes
- Hyperthermia for chronic infection-driven inflammation
- Ozone therapy for modulating oxidative-inflammatory balance
The honest answer is that chronic inflammation rarely has a single cause, which means it rarely has a single solution. The patients who achieve the best outcomes are those who address diet, sleep, stress, gut health, and toxin exposure simultaneously, with targeted supplementation based on their specific inflammatory profile.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical care. Consult a qualified physician before beginning any anti-inflammatory protocol.