Vaccination is one of the most effective medical interventions in human history. The immunological principles behind it are elegant: expose the immune system to a non-pathogenic version of a threat so that it can develop memory without the risks of actual infection.
I am writing this because my patients frequently ask about how vaccines work at the cellular level, what adjuvants do, why some people respond differently than others, and what factors influence vaccine efficacy. These are legitimate questions that deserve straightforward, evidence-based answers.
The Immunological Basis
Vaccines exploit the adaptive immune system’s capacity for immunological memory. The process follows a well-characterized sequence:
-
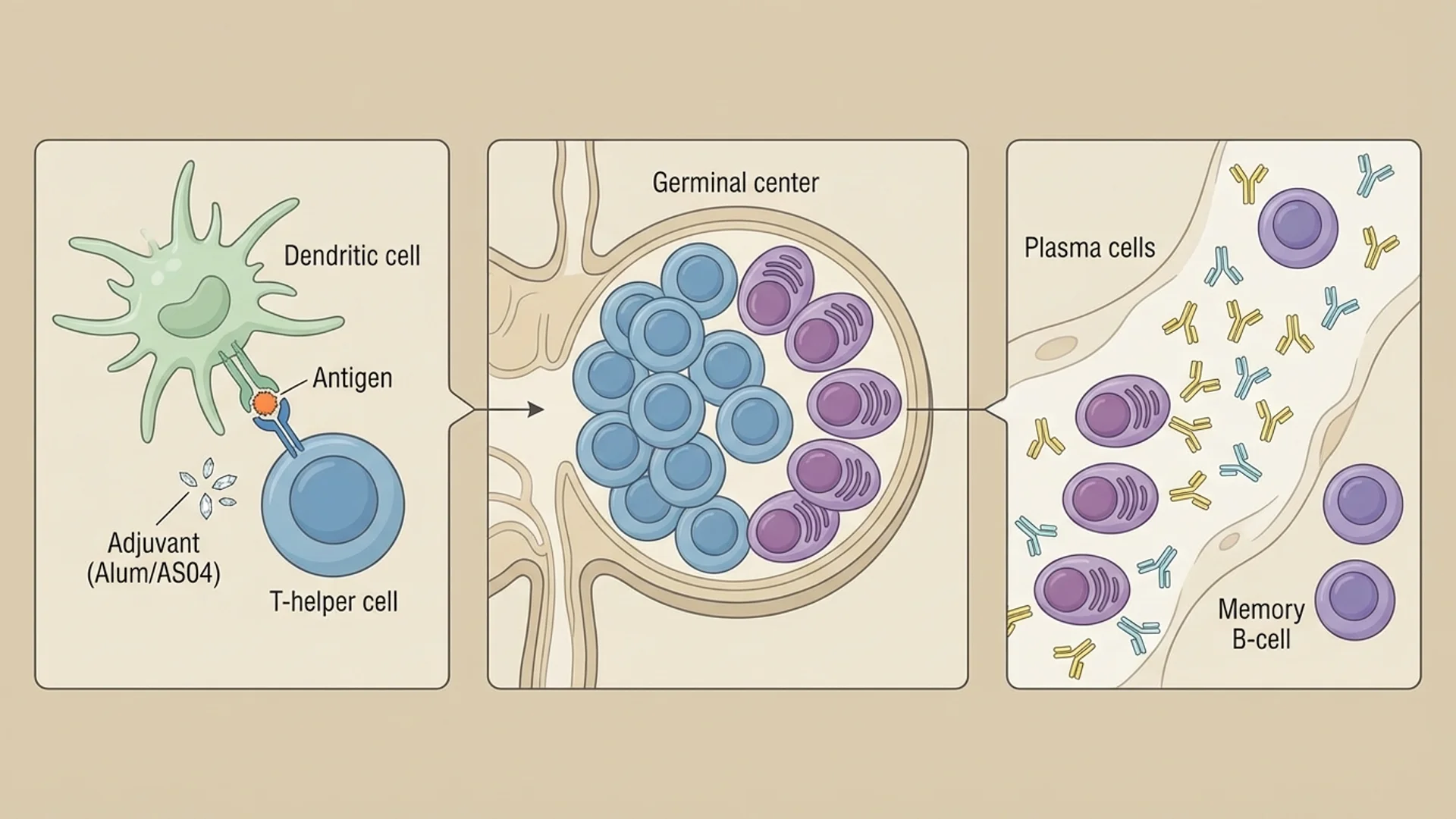
Antigen presentation. The vaccine delivers antigens — molecular fragments from the target pathogen — to the immune system. Dendritic cells at the injection site capture these antigens, process them, and migrate to the draining lymph node, where they present them to T and B cells.
-
Lymphocyte activation. In the lymph node, antigen-specific T and B cells are activated. Helper T cells (CD4+) provide signals that support B cell activation and differentiation. Cytotoxic T cells (CD8+) are primed to recognize and kill cells displaying the antigen.
-
Germinal center reaction. Activated B cells enter germinal centers within lymph nodes, where they undergo somatic hypermutation and affinity maturation. This process refines antibody binding — the antibodies produced become increasingly precise and effective over days to weeks.
-
Memory cell generation. A subset of activated T and B cells differentiate into long-lived memory cells that persist in lymphoid tissue, bone marrow, and circulation. These cells can survive for decades and mount a rapid, robust response upon re-encounter with the same antigen.
-
Secondary response. When the vaccinated individual encounters the actual pathogen, memory cells are activated within hours, producing a response that is faster, larger, and more targeted than the primary response. This is typically sufficient to prevent clinical disease.
Vaccine Types and Immune Responses
Different vaccine platforms engage the immune system differently:
Live attenuated vaccines (measles, mumps, rubella, varicella) use weakened versions of the pathogen that replicate in the host but do not cause significant disease. They produce broad immune responses — both antibody and cellular — and typically generate strong, long-lasting immunity, often with a single dose. They are generally contraindicated in immunosuppressed patients.
Inactivated vaccines (influenza, hepatitis A, polio-Salk) use killed pathogens. They primarily stimulate antibody responses and typically require multiple doses and periodic boosters to maintain protection.
Subunit/conjugate vaccines (hepatitis B, HPV, pneumococcal) use specific protein or polysaccharide antigens from the pathogen. They produce highly targeted antibody responses with good safety profiles but typically require adjuvants and multiple doses.
mRNA vaccines (COVID-19 Pfizer-BioNTech, Moderna) deliver genetic instructions for cells to produce the target antigen endogenously. Because the antigen is produced intracellularly, mRNA vaccines generate both antibody responses and robust CD8+ T cell responses. This is immunologically advantageous for pathogens where cellular immunity is important for protection.
Viral vector vaccines (COVID-19 AstraZeneca, Johnson & Johnson) use modified viruses to deliver the target antigen gene. They produce both humoral and cellular responses.
The Role of Adjuvants
Adjuvants are substances added to vaccines to enhance the immune response. Without adjuvants, many subunit vaccines would not generate sufficient immunity.
Aluminum salts (alum) are the most widely used adjuvant, present in vaccines for over 90 years. They work by creating a depot effect (slow antigen release) and by activating the NLRP3 inflammasome, which enhances antigen presentation. Safety data is extensive — billions of doses administered with well-characterized adverse event profiles. Evidence level: strong (decades of safety data and mechanistic studies).
AS04 (alum plus monophosphoryl lipid A) is used in the hepatitis B vaccine Fendrix and the HPV vaccine Cervarix. MPL activates TLR4, enhancing both innate and adaptive responses.
MF59 (squalene oil-in-water emulsion) is used in some influenza vaccines. It enhances antigen uptake by dendritic cells and promotes a broader immune response. Over 100 million doses have been administered with a well-established safety record.
AS01 (liposome-based) is used in the shingles vaccine Shingrix and the malaria vaccine RTS,S. It produces strong cellular immune responses.
Adjuvants are necessary because purified antigens alone are often poor immunogens. The innate immune system needs danger signals — provided by the adjuvant — to mount an effective adaptive response. In the absence of adjuvant, the immune system may recognize the antigen but generate tolerance rather than immunity.
Factors That Influence Vaccine Response
Individual variation in vaccine response is well-documented and clinically significant:
Age. Immune responses to vaccination decline with age due to thymic involution, reduced naive T cell production, and accumulated immune senescence. This is why older adults often receive higher-dose formulations (high-dose influenza vaccine) or more potent adjuvant systems (Shingrix versus Zostavax).
Nutritional status. Zinc deficiency impairs T cell function and antibody production, reducing vaccine efficacy. Vitamin D deficiency is associated with poorer vaccine responses. Protein malnutrition impairs essentially every aspect of immune function. Evidence level: controlled human studies.
Sleep. Sleep deprivation around the time of vaccination reduces antibody production. In a controlled study (Spiegel et al., JAMA), participants who slept four hours per night for six nights before influenza vaccination produced less than half the antibodies compared to well-rested controls.
Stress. Chronic psychological stress and elevated cortisol suppress vaccine responses. Caregivers of patients with dementia, for example, produce significantly weaker antibody responses to influenza vaccination than matched controls. Evidence level: controlled human studies.
Gut microbiome. Emerging evidence suggests that the gut microbiome influences vaccine responses, potentially through its effects on innate immune activation and adjuvant response. Antibiotic-treated individuals show reduced responses to certain vaccines. Evidence level: controlled studies (emerging).
Genetics. HLA polymorphisms influence antigen presentation and thus vaccine responses. Some individuals are genetically low responders to specific vaccines (5-10% of people do not seroconvert after hepatitis B vaccination, for example, regardless of dose or schedule).
Optimizing Vaccine Response
Based on the evidence above, several practical steps may support an effective vaccine response:
- Ensure adequate sleep (seven to nine hours) in the days surrounding vaccination
- Address vitamin D and zinc status before vaccination if levels are known to be low
- Avoid vaccination during periods of acute illness or severe stress when possible
- For older adults, discuss adjuvanted or high-dose vaccine formulations with your physician
These are modest interventions, but they address documented factors that influence immune response to vaccination.
A Physician’s Perspective
I approach vaccines the way I approach any medical intervention: based on the evidence, with attention to individual circumstances. The evidence for the efficacy of core vaccines — measles, polio, tetanus, hepatitis B, and others — is among the strongest in medicine. The public health impact of vaccination programs is not debatable in any meaningful scientific sense.
At the same time, I take seriously the questions patients raise about individual risk, adjuvant safety, and optimal timing. These are not unreasonable concerns, and they deserve evidence-based answers rather than dismissal.
What I tell my patients: vaccination is a tool. Like any medical tool, it works best when used thoughtfully — in the right patient, at the right time, with appropriate preparation. The immunological principles are sound, the evidence base is robust, and the benefits, for the vast majority of patients, clearly outweigh the risks.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical advice. Discuss vaccination decisions with your physician based on your individual health circumstances.