At a Glance
| Property | Value |
|---|---|
| Name | Peptide Therapy for Lyme Disease |
| Class | Antimicrobial peptide |
| Route | Subcutaneous injection, Oral, IV |
| Typical Dose | 250-500 mcg |
| Evidence Level | Strong |
| Key Uses | A multi-phase peptide protocol for chronic Lyme disease combining thymosin alpha-1 for immune restoration, LL-37 for biofilm disruption… |
Key Takeaways:
- Peptide therapy for Lyme disease addresses two key challenges: immune exhaustion and biofilm-associated persistence
- Thymosin alpha-1 has the strongest evidence base for immune restoration in chronic infection
- LL-37 offers a mechanistic rationale for biofilm disruption strategies, though clinical data is observational
- Peptides complement rather than replace antimicrobial therapy and other standard interventions
- This protocol is a clinical framework, not a prescription — individual variation requires physician-guided adjustment
Chronic Lyme disease presents one of the most complex clinical challenges in infectious disease medicine. Patients often present after months or years of symptoms despite antibiotic treatment, with a constellation of findings that includes persistent fatigue, cognitive dysfunction, musculoskeletal pain, and autonomic instability. The immunological profile in these patients frequently shows evidence of T-cell exhaustion, elevated inflammatory markers, and sometimes an inappropriate shift toward Th2-dominant immune responses.
At Klinik St. Georg, chronic Lyme disease has been treated for decades. My father, Dr. Friedrich Douwes, was among the first physicians in Germany to recognize chronic Lyme as a significant clinical entity. The peptide protocol I describe here has evolved from that clinical experience, combined with current research on immune modulation strategies and biofilm-associated infection.
I want to be clear about the evidence basis: this protocol is built on a combination of published research (primarily for thymosin alpha-1), preclinical data (for LL-37 and BPC-157), and clinical observation from our practice. It has not been validated in a randomized controlled trial for chronic Lyme disease specifically. What I tell my patients is this: the individual components have evidence, the rationale for combining them is sound, and the clinical observations are encouraging — but this is not yet guideline-level medicine.
Who This Protocol Is For
- Patients with confirmed or clinically diagnosed chronic Lyme disease
- Patients who have completed or are undergoing appropriate antimicrobial therapy
- Patients with documented evidence of immune dysregulation (low CD4/CD8 ratios, reduced NK cell function, elevated inflammatory markers)
- Patients under the supervision of a physician experienced in Lyme disease management
Who Should NOT Follow This Protocol Without Medical Supervision
- Patients with active autoimmune disease (thymosin alpha-1 could exacerbate autoimmune flares)
- Patients on immunosuppressive therapy
- Patients with active malignancy
- Pregnant or lactating women
- Patients who have not had appropriate diagnostic workup for Lyme disease (do not assume Lyme — confirm it)
Phase 1: Foundation and Assessment (Weeks 1-2)
Goal
Establish baseline immune and inflammatory status. Optimize foundational factors before initiating peptide therapy.
Actions
Diagnostic Assessment:
- Complete blood count with differential
- Lymphocyte subset panel (CD4, CD8, NK cell count and function)
- Inflammatory markers (CRP, ESR, cytokine panel if available)
- Vitamin D level (25-OH-D)
- Comprehensive metabolic panel
- Borrelia-specific testing (IgM/IgG Western blot, PCR if indicated)
- Co-infection screening (Babesia, Bartonella, Ehrlichia, Anaplasma)
Foundational Optimization:
Supplement Dose Timing Evidence Level Purpose
Magnesium glycinate 400 mg Evening Controlled studies Muscle relaxation, sleep support, enzymatic cofactor
Omega-3 (EPA/DHA) 2-3 g/day With meals RCTs for inflammation Anti-inflammatory support
NAC (N-acetylcysteine) 600 mg twice daily With meals Controlled studies Glutathione support, biofilm disruption potential
Lifestyle:
- Sleep optimization (8+ hours, consistent schedule, no screens 1 hour before bed)
- Anti-inflammatory diet (eliminate processed foods, sugar, alcohol during treatment)
- Gentle movement only (no intense exercise during active treatment — Herxheimer reactions can be exacerbated by exercise)
What to Expect
This phase is about establishing the foundation. Patients should not expect symptom improvement from these measures alone. The goal is to optimize the physiological substrate that the peptide therapy will act upon.
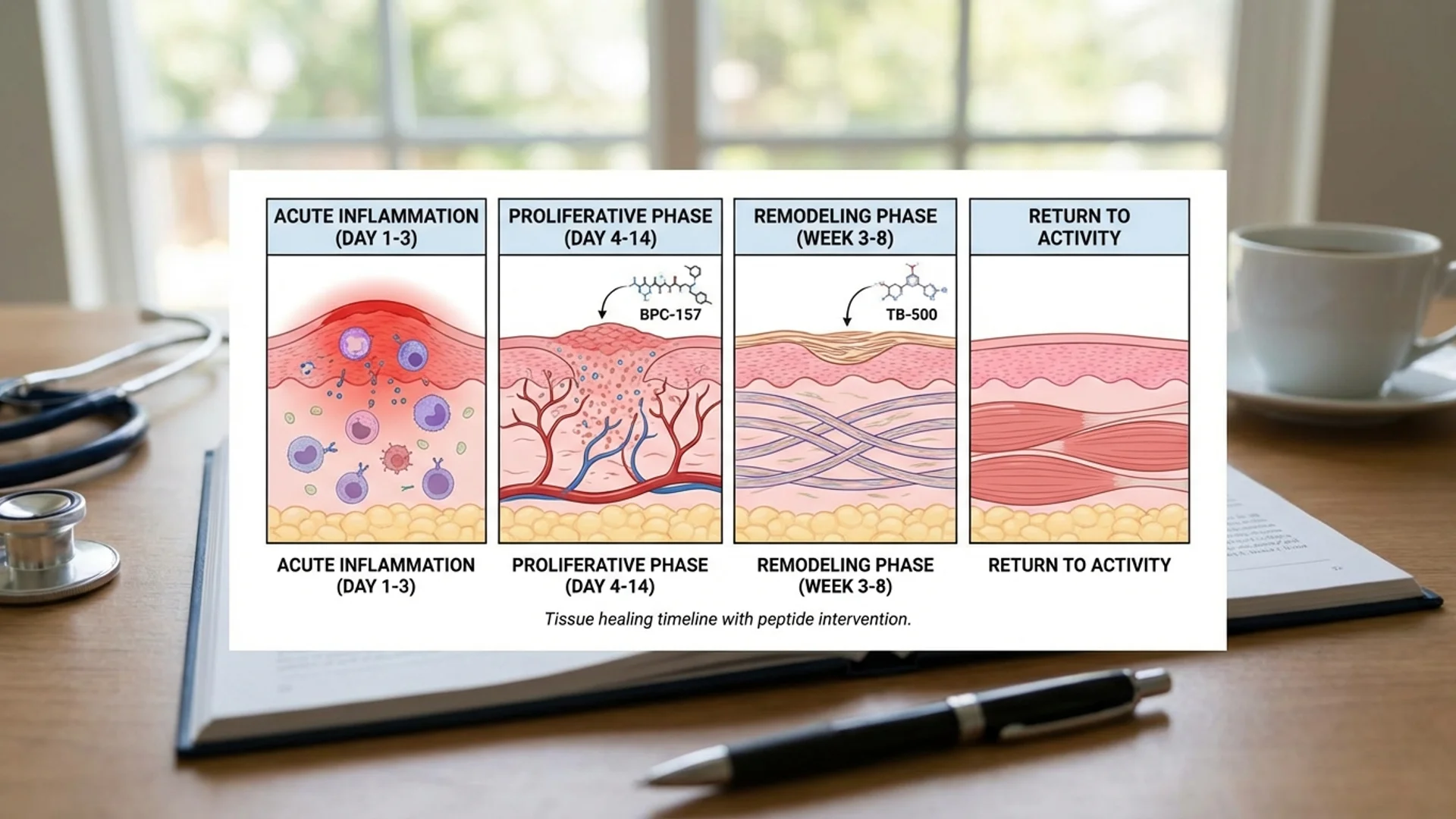
Phase 2: Immune Restoration (Weeks 3-14)
Goal
Restore immune competence through thymosin alpha-1, addressing the T-cell exhaustion and Th1/Th2 imbalance commonly seen in chronic Lyme patients.
Actions
Peptide Protocol:
Peptide Dose Route Frequency Duration Evidence Level Purpose
Antimicrobial Therapy: Continue or initiate antimicrobial therapy as directed by the treating physician. Peptide therapy is designed to complement, not replace, appropriate antibiotic treatment. The specific antimicrobial regimen depends on the clinical situation and should be individualized.
Monitoring (Week 6):
- Repeat lymphocyte subset panel
- Repeat inflammatory markers
- Clinical symptom assessment
- Adjust Ta1 dosing if immune parameters are not responding (some patients require 3.2 mg twice weekly)
What to Expect
Immune restoration is gradual. Most patients begin to notice improvement in fatigue and cognitive function between weeks 4 and 8. Some patients experience a temporary worsening of symptoms in the first 1-2 weeks (Herxheimer-like reaction as immune function improves and the immune system re-engages with persistent organisms). This is typically self-limiting.
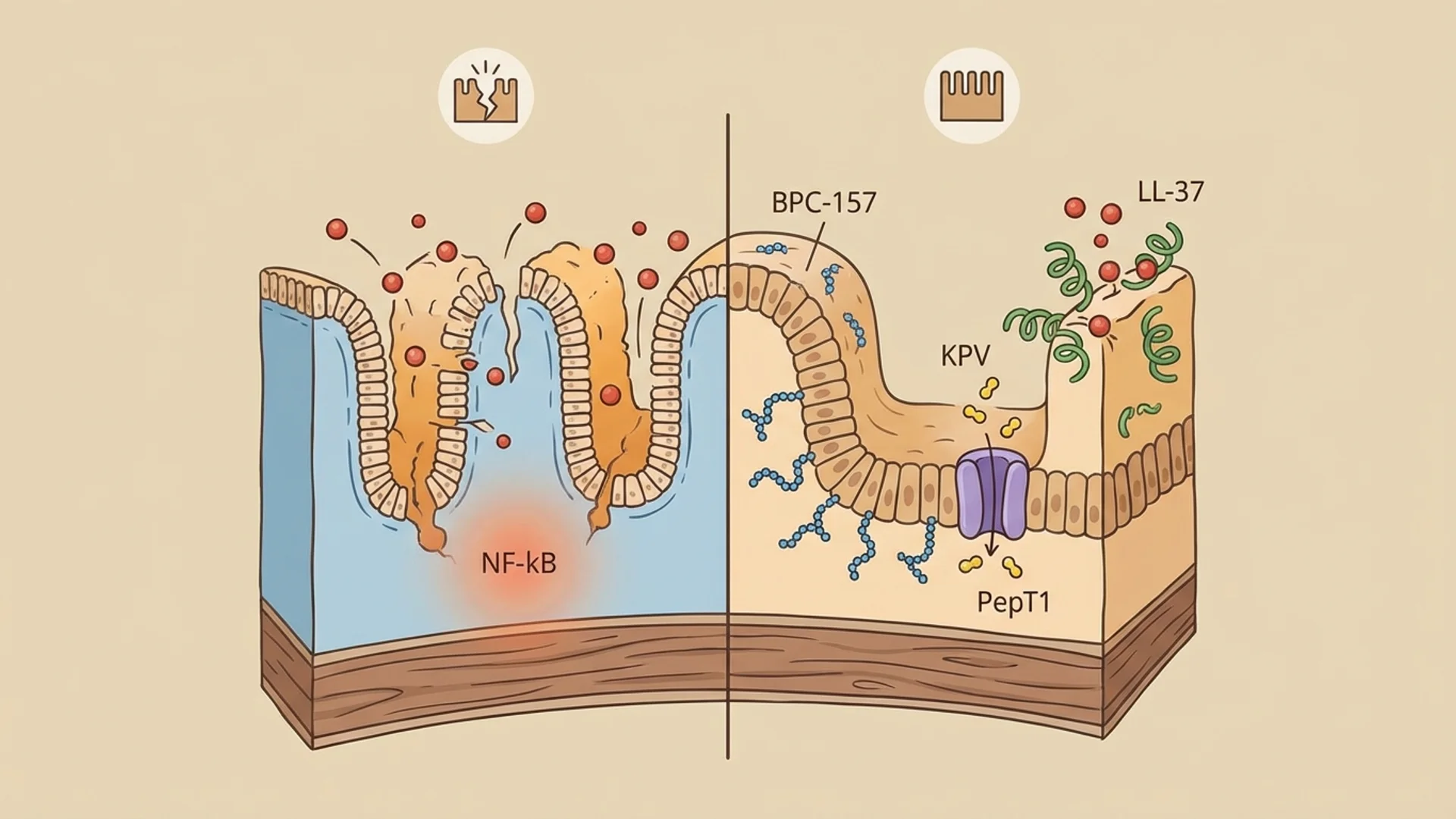
Phase 3: Biofilm and Tissue Support (Weeks 6-14, overlapping with Phase 2)
Goal
Address biofilm-associated persistence and support tissue repair in affected systems.
Actions
Peptide Protocol (added to ongoing Ta1):
Peptide Dose Route Frequency Duration Evidence Level Purpose
BPC-157 250-500 mcg SC injection Daily 6 weeks Animal studies (tissue repair); clinical observation Tissue repair, gut protection (if on antibiotics), neuroprotection
Biofilm Support:
Supplement Dose Timing Evidence Level Purpose
Serrapeptase 120,000 SPU Empty stomach Limited clinical data Biofilm enzyme support
Stevia extract (whole leaf) 200 mg With meals In vitro only Preliminary anti-biofilm data against Borrelia
What to Expect
This phase often coincides with the period of greatest symptom fluctuation. Some patients experience what appears to be a Herxheimer reaction as biofilm-associated organisms are exposed to antimicrobial therapy. Symptoms may temporarily worsen before improving. Close communication with the treating physician is essential during this phase.
Phase 4: Consolidation and Recovery (Weeks 15-20)
Goal
Consolidate immune gains, complete tissue repair support, and transition to maintenance.
Actions
Peptide Protocol:
Peptide Dose Route Frequency Duration Evidence Level Purpose
BPC-157 250 mcg SC injection or oral Daily (4 weeks, then stop) 4 weeks Clinical practice Complete tissue repair phase
Continue:
- Foundational supplements (Vitamin D, magnesium, omega-3)
- Anti-inflammatory diet
- Gradual reintroduction of exercise (start with walking, progress slowly)
What to Expect
Most patients who respond to this protocol report the most significant improvements during Phases 3 and 4. Energy returns, cognitive function improves, and musculoskeletal pain diminishes. Complete resolution of all symptoms is not always achieved — some patients retain residual symptoms that may reflect tissue damage from prolonged infection rather than active disease.
When to Seek Medical Help
- Fever above 38.5C (101.3F) during treatment
- Severe headache with neck stiffness (evaluate for CNS involvement)
- New neurological symptoms (numbness, weakness, vision changes)
- Signs of allergic reaction to any peptide (hives, angioedema, difficulty breathing)
- Significant worsening of symptoms that does not improve within 48-72 hours
- Joint swelling, redness, or warmth suggesting acute arthritis
- Chest pain or shortness of breath
Monitoring and Assessment
- Lymphocyte subsets at baseline, week 6, week 14, and week 20
- Inflammatory markers at baseline and every 6 weeks
- Lyme-specific markers as clinically indicated
- Clinical symptom scoring using a standardized Lyme symptom checklist at each visit
- Vitamin D levels every 8 weeks until stable in target range
Long-Term Maintenance
After completing the protocol, I recommend:
- Continued vitamin D optimization (maintain 60-80 ng/mL)
- Ongoing anti-inflammatory dietary principles
- Regular exercise (as tolerated, gradually increasing intensity)
- Annual immune panel to monitor sustained restoration
- Repeat Ta1 course (4-6 weeks) if immune parameters decline or symptoms recur
- Ongoing co-management with Lyme-literate physician
Clinical Perspective
This protocol reflects my clinical approach to chronic Lyme disease over the past several years. It is not a fixed recipe — every patient is different, and the specific peptides, doses, and timing are adjusted based on individual response, immune status, and co-infections.
The patients who respond best to this approach share certain characteristics: they have documented immune dysfunction (measurable on laboratory testing, not just clinical impression), they are compliant with the full protocol (including dietary and lifestyle modifications), and they are receiving concurrent antimicrobial therapy appropriate to their infection profile.
What I want to be transparent about is that not every patient responds to this protocol. Chronic Lyme disease is a heterogeneous condition, and some patients have co-infections, immune genetic variants, or tissue damage patterns that require different or additional interventions. The protocol I have described is a starting framework, not a guaranteed solution.
The evidence hierarchy for this protocol is mixed: thymosin alpha-1 has robust human clinical data for immune restoration (though not specifically for Lyme), LL-37 has strong in vitro anti-biofilm data (but limited clinical data for systemic use), and BPC-157 has strong preclinical tissue repair data (but limited human trials). I am transparent with patients about where each component sits in the evidence hierarchy.
What I can say with confidence is that addressing immune dysfunction in chronic Lyme patients — rather than relying solely on antimicrobial therapy — has been a meaningful addition to our treatment approach at Klinik St. Georg. Whether peptides are the optimal tool for achieving this, or whether other immunomodulatory approaches would be equally effective, remains an open question that deserves continued investigation.

References
- Goldstein AL, et al. “Thymosin alpha 1: isolation and sequence analysis.” Proc Natl Acad Sci USA. 1977;74(2):725-729.
- Garaci E, et al. “Thymosin alpha 1 in the treatment of infections and cancer.” Int J Immunopharmacol. 2000;22(12):1067-1076.
- Overhage J, et al. “Human host defense peptide LL-37 prevents bacterial bacterial biofilm formation.” Infect Immun. 2008;76(9):4176-4182.
- Sapi E, et al. “Evaluation of in-vitro antibiotic susceptibility of different morphological forms of Borrelia burgdorferi.” Infect Drug Resist. 2011;4:97-113.
- Sikiric P, et al. “Brain-gut Axis and Pentadecapeptide BPC 157.” Curr Neuropharmacol. 2016;14(8):857-865.
- Stricker RB, Johnson L. “Lyme disease: the next decade.” Infect Drug Resist. 2011;4:1-9.
- You J, et al. “Thymosin alpha-1 for chronic hepatitis B: a meta-analysis.” World J Gastroenterol. 2006;12(46):7503-7506.
Disclaimer: This protocol is provided for educational purposes and reflects one physician’s clinical approach. It is not a substitute for individualized medical care. Chronic Lyme disease requires comprehensive evaluation and treatment by a qualified physician. Do not self-treat with peptides or antibiotics without medical supervision.