Featured
Neurofeedback for Lyme Brain Fog
How neurofeedback retrains dysregulated brainwave patterns in Lyme disease patients. qEEG findings, clinical evidence, and what to expect from treatment.
Condition
Microclots, brain fog, chronic fatigue, dysautonomia. Comprehensive multimodal treatment including apheresis, NAD+ IV, IHHT, and targeted protocols at St. George Hospital.
Clear comparison of neurofeedback and biofeedback. What each measures, how they work, conditions they treat, and how to choose the right one for your situation.
How vagus nerve stimulation restores autonomic balance in chronic fatigue and POTS — the cholinergic anti-inflammatory pathway, clinical evidence, and treatment options.
What causes brain fog in Lyme disease — Johns Hopkins PET scan findings showing glial activation, neuroinflammation mechanisms, and treatment implications.
What to expect during brain fog recovery after Lyme treatment — the multi-system recovery approach, realistic timelines, and evidence-based strategies.

When to get neuropsychological testing for brain fog. What the evaluation measures, how it differs from standard neurology exams, and what the results mean for treatment.
How photobiomodulation targets mitochondrial dysfunction to clear brain fog. Mechanism, clinical evidence, and practical application for chronic illness patients.
Complete guide to quantitative EEG brain mapping. What the test measures, how it identifies brain dysregulation, and why it matters for neurofeedback and brain fog treatment.
Evidence-based review of transcranial photobiomodulation for brain health. Wavelengths, mechanisms, clinical trial data, and what I see in practice.

Brain fog treatment options from neuroinflammation workup to neuromodulation. Causes include post-COVID, Lyme, MCAS, and mold. TPS, NAD+, and hyperbaric oxygen covered.
How H.E.L.P. apheresis filters LDL, fibrinogen, CRP, and microclots from your blood. Mechanism, session details, evidence, and clinical experience at Klinik St. Georg.
Evidence-based guide to nattokinase for fibrinolysis and microclots. Mechanism, dosing, post-COVID relevance, combination with serrapeptase, and safety with blood thinners.
How microclots and damaged blood vessel linings drive post-COVID symptoms. Dr. Beate Jaeger's research and clinical implications.

How we treat Long COVID and post-COVID syndrome. Mechanisms, diagnostics, and a multimodal treatment program grounded in clinical experience.
Why COVID-19 causes persistent cognitive dysfunction and what can be done about it. Mechanisms, assessment, and treatment strategies.
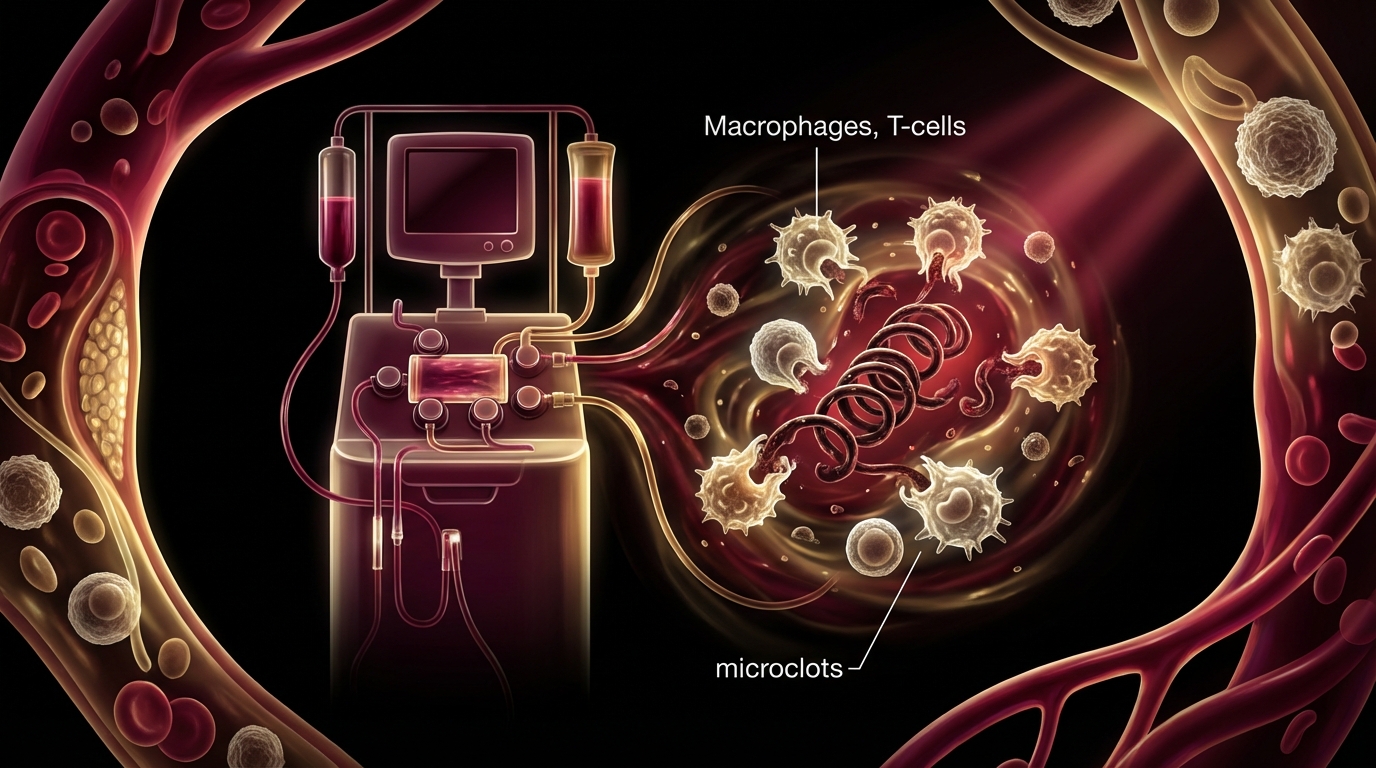
How H.E.L.P. apheresis removes microclots and inflammatory mediators in Long COVID patients. Session protocols, evidence from Pretorius research, and clinical experience at Klinik St. Georg.
A clinical guide to Long COVID symptoms and treatment based on 500+ patients. Microclots, spike protein persistence, endothelial dysfunction, and the therapies that produce measurable results.

An evidence-based recovery protocol addressing inflammation, clotting, energy, and immune regulation after COVID-19. By Dr. Julian Douwes.
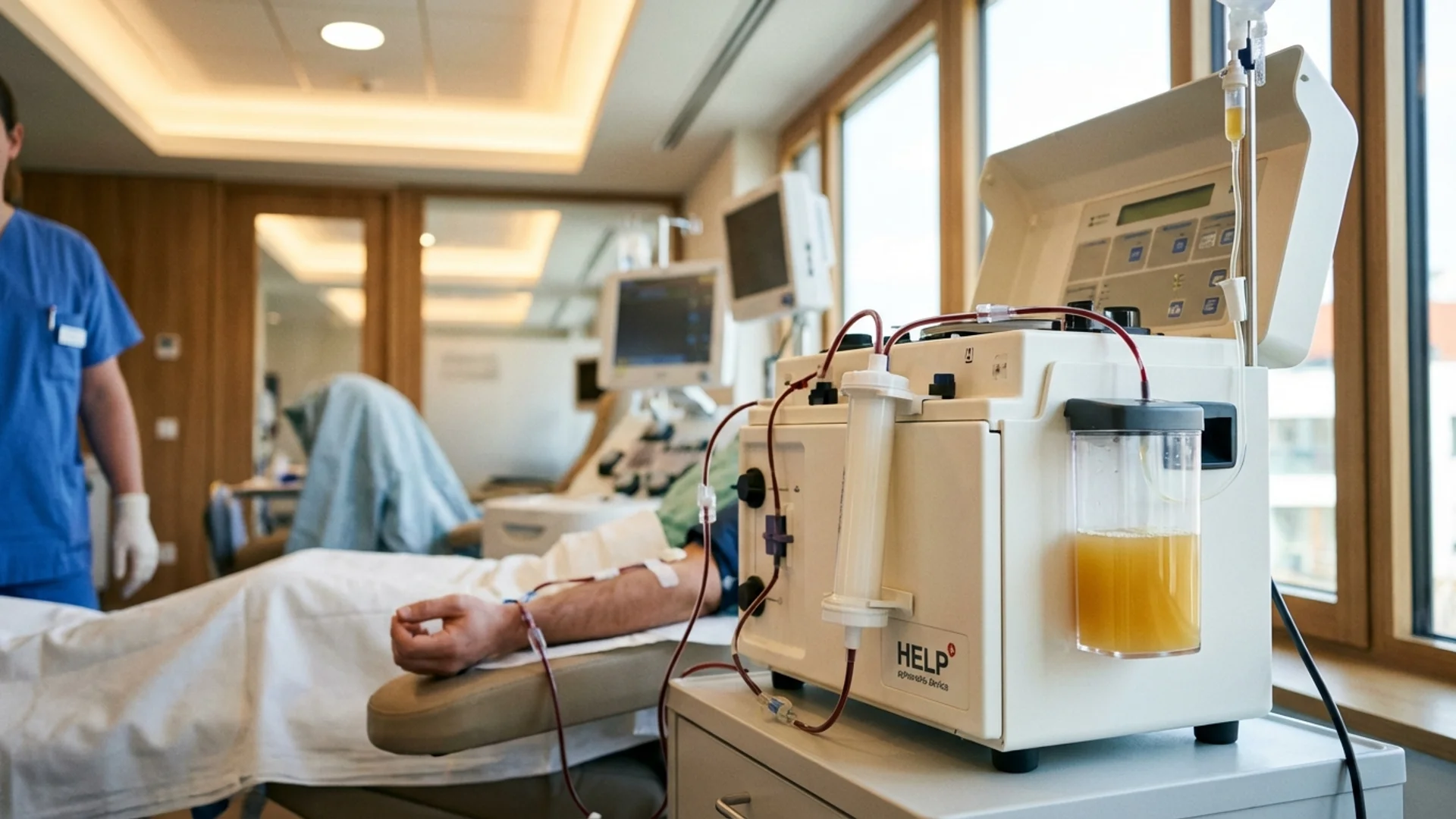
How therapeutic apheresis filters inflammatory mediators, autoantibodies, and microclots. Applications in post-COVID and chronic infection treatment.
Schedule a consultation to discuss your case with Dr. Julian Douwes and the clinical team at St. George Hospital.
Complete Guide
In-depth reference by Dr. Julian Douwes
I want to begin with a statement that should be unnecessary but, given the experience of many patients who find their way to us, apparently is not: Post-COVID syndrome is not in your head. It is not anxiety. It is not deconditioning. It is not a manifestation of pandemic-related stress, though that certainly does not help.
Post-COVID syndrome — also called long COVID, post-acute sequelae of SARS-CoV-2 (PASC), or simply long-haul COVID — is a condition with measurable pathology. We can see it in blood work. We can see it in immune panels. We can see it in microcirculation analysis. We can, in many patients, identify the specific mechanisms driving their symptoms and design treatment accordingly.
The problem is not that the pathology is invisible. The problem is that most standard medical workups are not designed to look for it.
After treating several hundred post-COVID patients at St. George Hospital, and following the evolving research literature closely, we have identified six overlapping pathophysiological mechanisms that account for the majority of symptoms. Most patients have two or more of these operating simultaneously, which is why single-target treatments typically produce limited results.
SARS-CoV-2 spike protein — and in some cases, spike protein fragments generated by the immune response to vaccination — has been detected in human tissue months after acute infection. Studies have identified spike protein in the intestinal mucosa, vascular endothelium, neural tissue, and circulating monocytes well beyond the expected clearance window [1].
Persistent spike protein is not inert. It continues to activate inflammatory pathways, interfere with the ACE2 receptor system, and trigger immune responses. The tissue distribution of persistent spike protein correlates meaningfully with symptom patterns: patients with predominantly neurological symptoms often show evidence of spike persistence in neural tissue, while those with predominantly cardiovascular symptoms show endothelial involvement.
This mechanism helps explain why some patients experience symptoms that wax and wane over months — the spike protein reservoirs may be periodically releasing fragments that trigger inflammatory flares.
This is an area of active and important research. The work of Dr. Beate Jaeger, who collaborates with our hospital, and researchers including Etheresia Pretorius and Douglas Kell, has identified the presence of anomalous fibrin microclots in the blood of post-COVID patients. These microclots are resistant to normal fibrinolysis — they do not dissolve through standard physiological mechanisms — and they appear to obstruct microcirculation, reducing oxygen and nutrient delivery to tissues [2].
I want to frame this carefully. The microclot hypothesis represents an important research direction that has generated compelling preliminary evidence. It provides a plausible mechanistic explanation for many post-COVID symptoms, particularly fatigue, cognitive dysfunction, and exercise intolerance — all of which could result from impaired microcirculation and tissue hypoxia. However, the field is still establishing the precise clinical significance of these findings, and the optimal therapeutic approach to microclot dissolution remains under investigation.
What we can say with confidence is that many of our post-COVID patients demonstrate measurable abnormalities in microcirculation and endothelial function, and that treatments targeting these abnormalities — particularly apheresis — produce consistent clinical improvement.
Endothelial dysfunction extends beyond microclots. SARS-CoV-2 has a particular tropism for endothelial cells via ACE2 receptors, and direct viral damage to the vascular endothelium can persist long after acute infection. This contributes to impaired vascular reactivity, abnormal blood flow regulation, and a pro-thrombotic state that compounds the microclot problem [3].
Post-COVID patients frequently demonstrate a characteristic pattern of immune dysfunction. This is not simple immune suppression or immune overactivation — it is dysregulation, a loss of the balanced coordination that characterizes healthy immune function.
Th1/Th2 imbalance. The immune system’s T-helper cell response becomes skewed, often with suppressed Th1 (cell-mediated) responses and relative Th2 (humoral) dominance. This impairs the body’s ability to clear persistent viral fragments and intracellular infections while maintaining a chronic inflammatory state [4].
T-cell exhaustion. We consistently observe elevated markers of T-cell exhaustion — PD-1 expression, reduced T-cell proliferative capacity, decreased cytotoxic function — in post-COVID patients. These are the same patterns seen in chronic viral infections and certain cancers. The immune system is not inactive; it is functionally impaired, trapped in an exhausted state where it cannot mount an effective response [5].
Autoimmune phenomena. A subset of post-COVID patients develops autoantibodies, including anti-G-protein-coupled receptor antibodies, anti-neuronal antibodies, and anti-phospholipid antibodies. These autoimmune processes may be triggered by molecular mimicry, bystander activation, or the persistent inflammatory state itself. They contribute to symptoms that persist long after viral clearance and may require specific treatment [6].
SARS-CoV-2 does not operate in isolation. The immune disruption it causes creates an environment permissive for the reactivation of latent viruses that most of the population carries asymptomatically.
Epstein-Barr virus (EBV) reactivation is documented in an estimated 60-70% of long COVID patients in some cohorts [7]. EBV reactivation drives fatigue, lymphadenopathy, sore throat, and cognitive dysfunction — symptoms that overlap heavily with post-COVID complaints and may be mistakenly attributed to COVID when EBV is actually the primary driver.
Human herpesvirus 6 (HHV-6) reactivation is less well-studied but clinically significant. HHV-6 has particular neurotropism and can contribute to neurological symptoms including brain fog, mood disturbance, and autonomic dysfunction.
We systematically test for EBV and HHV-6 reactivation in every post-COVID patient. When present, these reactivations must be addressed as part of the treatment plan. Treating post-COVID without addressing concurrent viral reactivation is, in our experience, a common reason for incomplete recovery.
Post-COVID autonomic dysfunction manifests most commonly as postural orthostatic tachycardia syndrome (POTS) — an excessive heart rate increase upon standing, often accompanied by lightheadedness, palpitations, exercise intolerance, and blood pressure instability. But the autonomic nervous system regulates far more than heart rate and blood pressure: it controls digestion, thermoregulation, sleep architecture, bladder function, and pupillary responses. Many seemingly unrelated post-COVID symptoms trace back to autonomic disruption.
The mechanism appears to involve both direct vagal nerve damage from viral neurotropism and autoantibody-mediated dysfunction of autonomic receptors. Some patients demonstrate small fiber neuropathy on skin biopsy, confirming structural damage to the autonomic nerve fibers [8].
Autonomic dysfunction is frequently underdiagnosed because standard cardiac and neurological examinations do not assess it. It requires specific testing — tilt-table examination, heart rate variability analysis, sudomotor function testing — that is not part of routine post-COVID evaluation in most settings.
The cellular energy production system — the mitochondria — appears to be a consistent casualty of SARS-CoV-2 infection. Multiple studies have demonstrated impaired mitochondrial function in post-COVID patients, manifesting as reduced oxidative phosphorylation capacity, increased reactive oxygen species production, and impaired cellular energy metabolism [9].
Clinically, mitochondrial dysfunction produces the profound, characteristic fatigue of post-COVID syndrome — a fatigue qualitatively different from tiredness or deconditioning. It is accompanied by exercise intolerance, post-exertional malaise (symptom worsening after physical or cognitive effort), and a general sense of cellular exhaustion that patients describe as feeling “poisoned” or “running on empty.”
Mitochondrial function can be assessed through functional testing including organic acid profiles, lactate-to-pyruvate ratios, and specialized metabolomic panels. These findings guide our therapeutic approach to mitochondrial restoration.
Before treatment begins, we perform a comprehensive diagnostic assessment designed to identify which of the six mechanisms above are active in each patient. This is not a standardized panel applied identically to every patient — it is a systematic investigation guided by the clinical presentation.
Immune profiling. Extended lymphocyte subsets (CD4/CD8 ratios, NK cell function, regulatory T cells), T-cell exhaustion markers (PD-1, Tim-3), immunoglobulin levels, cytokine panels (IL-6, TNF-alpha, interferon-gamma), autoantibody screening (including anti-GPCR antibodies).
Microclot visualization. We employ fluorescence microscopy of citrated plasma to assess for the presence and density of anomalous fibrin microclots, following protocols developed in the research of Pretorius and colleagues. We also assess endothelial function markers including von Willebrand factor and soluble thrombomodulin.
Autonomic testing. Heart rate variability analysis, active standing test with continuous heart rate and blood pressure monitoring, and where indicated, formal tilt-table testing. Pupillometry and sudomotor testing may be added based on clinical presentation.
Viral reactivation panels. EBV viral capsid antigen IgM, EBV early antigen IgG, EBV DNA PCR, HHV-6 IgG and DNA PCR. These differentiate past infection from active reactivation.
Inflammatory and metabolic panels. High-sensitivity CRP, D-dimer, fibrinogen, ferritin, comprehensive metabolic panel, organic acids, coenzyme Q10, vitamin D, zinc, selenium, and other micronutrients that influence immune and mitochondrial function.
This workup typically requires 1-2 days and provides the foundation for an individualized treatment plan.
Apheresis is frequently the first treatment we initiate in post-COVID patients, and for many patients, it produces the most immediately noticeable improvement. The procedure selectively filters the blood to remove inflammatory mediators, circulating immune complexes, autoantibodies, fibrin fragments, and — we believe, based on the available evidence — microclot components.
Patients typically undergo 3-5 apheresis sessions during their treatment program. The response pattern is characteristic: many patients report improved cognitive clarity, reduced fatigue, and decreased pain within 24-48 hours of their first session. This is consistent with the removal of circulating inflammatory and thrombotic factors that were impairing microcirculation and driving systemic inflammation [10].
Apheresis is not a permanent solution in isolation — the underlying mechanisms that produce inflammatory mediators and microclots must also be addressed. But it provides a rapid improvement in baseline function that facilitates the patient’s ability to engage with and tolerate other treatments.
Nicotinamide adenine dinucleotide (NAD+) is a critical coenzyme in mitochondrial energy production. NAD+ levels decline with age and are further depleted by chronic infection and inflammation. In post-COVID patients with documented mitochondrial dysfunction, IV NAD+ supplementation provides the substrate necessary for mitochondrial recovery.
We administer NAD+ intravenously because oral bioavailability of NAD+ precursors is limited and unpredictable. IV administration achieves supraphysiological levels that can directly support mitochondrial function. Patients typically report improved energy, cognitive clarity, and reduced post-exertional malaise over the course of treatment [11].
NAD+ therapy is not a one-time intervention. We establish a treatment course during the inpatient stay and provide guidance for ongoing oral precursor supplementation (typically NMN or NR) during the recovery period.
IHHT involves alternating periods of breathing hypoxic air (reduced oxygen, approximately 10-14%) with hyperoxic air (enriched oxygen, approximately 36-40%). This controlled oscillation stimulates mitochondrial biogenesis — the creation of new mitochondria — through activation of hypoxia-inducible factor pathways and AMPK signaling [12].
The concept is analogous to altitude training for athletes, but applied therapeutically. By repeatedly exposing cells to mild hypoxic stress followed by oxygen-rich recovery, we trigger adaptive responses that result in a net increase in mitochondrial mass and improved mitochondrial efficiency.
IHHT sessions are conducted while the patient rests comfortably, breathing through a mask connected to an altitude simulation device. Sessions last approximately 40 minutes, and patients typically complete 10-15 sessions during their treatment program. The effects are gradual and cumulative — patients often report the most significant improvement in exercise tolerance 2-4 weeks after completing their IHHT course.
Medical ozone therapy involves the controlled administration of an ozone-oxygen mixture, either through major autohemotherapy (mixing ozone with the patient’s own blood, then reinfusing) or rectal insufflation. The mechanisms of action are multiple: immune modulation (shifting toward Th1-mediated responses), improved oxygen utilization, enhanced antioxidant enzyme systems, and mild antimicrobial activity [13].
In post-COVID patients, ozone therapy serves primarily as an immune modulator. It helps rebalance the skewed Th1/Th2 response, supports natural killer cell function, and may contribute to clearance of persistent viral fragments. It also improves red blood cell deformability, enhancing microcirculation — relevant given the endothelial dysfunction present in many patients.
Ozone therapy has a long history of clinical use in Germany and is well-established within the German medical tradition. The evidence base is stronger for immune modulation than for direct antiviral effects, and we frame it accordingly.
We employ peptide therapies selectively and with transparent communication about their evidence levels.
BPC-157 (Body Protection Compound) is used in patients with significant tissue injury, gastrointestinal dysfunction, or tendon and ligament pathology. Animal studies demonstrate impressive tissue-repair and anti-inflammatory properties. Human clinical data remain limited, and we communicate this clearly while noting the favorable safety profile observed in our clinical experience. BPC-157 is considered investigational.
Thymosin alpha-1 is used in patients with documented immune suppression, particularly those with reduced NK cell function and T-cell exhaustion. This peptide has the strongest evidence base among those we employ, with regulatory approval in multiple countries for immune restoration in chronic viral infections. In post-COVID patients with immune exhaustion profiles, thymosin alpha-1 supports the restoration of balanced immune function [14].
These peptide therapies are supplementary to, not substitutes for, the primary treatment modalities.
For patients with significant autonomic dysfunction and cognitive impairment, we employ neuromodulation techniques.
Transcutaneous auricular vagus nerve stimulation (taVNS) involves gentle electrical stimulation of the auricular branch of the vagus nerve in the ear. This activates vagal afferent pathways, improving parasympathetic tone and helping rebalance the autonomic nervous system. For POTS patients and those with autonomic dysfunction, taVNS can produce measurable improvements in heart rate variability and symptom burden [15].
Neurofeedback uses real-time EEG monitoring to train brain activity patterns. Post-COVID patients with brain fog frequently demonstrate characteristic EEG abnormalities — reduced alpha power, increased theta activity, impaired coherence. Neurofeedback provides the brain with real-time information about its own activity, enabling gradual normalization of these patterns. Sessions are conducted over the course of the inpatient stay, and patients may continue with home-based protocols after discharge.
Exercise in post-COVID patients must be approached with extreme caution. Post-exertional malaise (PEM) — the characteristic worsening of symptoms 24-72 hours after exertion — is a hallmark of the condition, and aggressive exercise programs can cause significant setbacks.
We work with patients to establish their current activity tolerance and prescribe exercise that remains within the “energy envelope” — the level of activity that does not trigger PEM. This often begins with gentle walking, breath work, or seated exercises, and is titrated upward only as mitochondrial function, autonomic stability, and inflammatory markers improve.
This is not deconditioning that can be resolved through willpower and a gym membership. It is cellular energy impairment that requires metabolic recovery before exercise capacity can be rebuilt. We emphasize this strongly because inappropriate exercise prescription remains one of the most common harms inflicted on post-COVID patients by well-meaning clinicians.
Having treated chronic Lyme disease for 30 years and post-COVID for the past several, the parallels are striking and clinically important.
Both conditions involve immune dysregulation with T-cell exhaustion and NK cell suppression. Both produce chronic inflammation that outlasts the initial infection. Both cause autonomic dysfunction, cognitive impairment, and profound fatigue. Both are poorly served by standard diagnostics and frequently dismissed by conventional medicine.
The overlap goes beyond symptom similarity. We see significant numbers of patients whose post-COVID symptoms are actually driven by reactivation of latent Lyme disease or co-infections. The immune disruption caused by SARS-CoV-2 can release previously controlled Borrelia, Bartonella, or EBV from latent reservoirs, producing a clinical picture that appears to be “long COVID” but is actually a chronic infection unmasked by immune compromise.
We test every post-COVID patient for tick-borne diseases and viral reactivation, and we treat what we find. In some patients, addressing the reactivated Lyme disease or EBV produces more improvement than any COVID-specific therapy.
Patients deserve honest expectations. Based on our clinical experience:
Most patients improve. The majority of post-COVID patients who complete our treatment program experience meaningful improvement in their primary symptoms — fatigue, cognitive function, exercise tolerance, autonomic stability. This is not anecdotal impression; we track outcomes systematically.
Recovery is gradual. Apheresis may provide rapid initial improvement, but full recovery of mitochondrial function, immune balance, and autonomic stability takes time. A realistic timeline for substantial improvement is 3-6 months. For patients with severe presentations, 6-12 months is more appropriate. Some patients experience continued improvement for 18-24 months.
Setbacks occur. Recovery is not linear. Patients experience good days and bad days, good weeks and difficult weeks. Viral exposures, stress, overexertion, and hormonal fluctuations can trigger temporary setbacks. This does not mean treatment has failed — it means the recovery process has the variability inherent in complex biological systems.
Some symptoms may persist. We aim for maximal functional recovery, but complete resolution of every symptom in every patient is not realistic. Some patients retain mild residual symptoms — particularly subtle cognitive changes or exercise sensitivity — even after substantial overall improvement.
Lifestyle modification matters. Sleep quality, stress management, anti-inflammatory nutrition, appropriate exercise progression, and avoidance of immune-suppressive exposures all influence recovery trajectory. Treatment at our hospital provides a foundation, but what patients do in the months following discharge significantly affects their outcome.
We have extensive experience supporting international patients. Our international patient coordinator manages the logistics of treatment planning, hotel and accommodation arrangements near the hospital, airport transfers, medical record translation, and coordination with home physicians for post-discharge follow-up.
The initial consultation can be conducted remotely via telemedicine. We review records, assess the clinical picture, and provide a treatment recommendation and cost estimate before any commitment to travel. Most treatment programs are 2-3 weeks in duration.
Bad Aibling is located approximately 60 kilometers southeast of Munich, accessible via Munich International Airport (MUC). The town itself is a traditional Bavarian spa town, and the pace of recovery is well-suited to the therapeutic environment.
For patients who have been struggling for months or years with a condition their physicians cannot explain or effectively treat, the decision to travel for care is significant. We respect that decision by providing thorough, individualized treatment based on the best available evidence and our clinical experience with complex post-infectious illness.
[1] Patterson BK, et al. “Persistence of SARS CoV-2 S1 protein in CD16+ monocytes in post-acute sequelae of COVID-19 (PASC) up to 15 months post-infection.” Front Immunol. 2022;12:746021.
[2] Pretorius E, et al. “Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin.” Cardiovasc Diabetol. 2021;20:172.
[3] Huertas A, et al. “Endothelial cell dysfunction: a major player in SARS-CoV-2 infection (COVID-19)?” Eur Respir J. 2020;56(1):2001634.
[4] Peluso MJ, et al. “Long-term SARS-CoV-2-specific immune and inflammatory responses in individuals recovering from COVID-19 with and without post-acute symptoms.” Cell Rep. 2021;36(6):109518.
[5] Zheng HY, et al. “Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients.” Cell Mol Immunol. 2020;17(5):541-543.
[6] Wallukat G, et al. “Functional autoantibodies against G-protein coupled receptors in patients with persistent Long-COVID-19 symptoms.” J Transl Autoimmun. 2021;4:100100.
[7] Gold JE, et al. “Investigation of Long COVID prevalence and its relationship to Epstein-Barr virus reactivation.” Pathogens. 2021;10(6):763.
[8] Oaklander AL, et al. “Peripheral neuropathy evaluations of patients with prolonged long COVID.” Neurol Neuroimmunol Neuroinflamm. 2022;9(3):e1146.
[9] Guntur VP, et al. “Signatures of mitochondrial dysfunction and impaired fatty acid metabolism in plasma of patients with post-acute sequelae of COVID-19 (PASC).” Metabolites. 2022;12(11):1026.
[10] Achleitner M, et al. “Immunoadsorption in post-COVID patients with neurocognitive symptoms.” Mol Psychiatry. 2022;27:1-3.
[11] Braidy N, et al. “Role of Nicotinamide Adenine Dinucleotide and Related Precursors as Therapeutic Targets for Age-Related Degenerative Diseases.” Antioxid Redox Signal. 2019;30(2):187-207.
[12] Serebrovska TV, et al. “Intermittent hypoxia training in prediabetes patients: beneficial effects on glucose homeostasis, hypoxia tolerance and gene expression relevant to mitochondrial biogenesis.” Front Endocrinol. 2019;10:650.
[13] Bocci V. “Biological and clinical effects of ozone. Has ozone therapy a future in medicine?” Br J Biomed Sci. 2017;56(4):270-279.
[14] Romani L, et al. “Thymosin alpha 1: an endogenous regulator of inflammation, immunity, and tolerance.” Ann N Y Acad Sci. 2012;1270:32-36.
[15] Badran BW, et al. “Neurophysiologic effects of transcutaneous auricular vagus nerve stimulation (taVNS) via electrical stimulation of the tragus.” Brain Stimul. 2018;11(3):492-500.