Selank presents an unusual situation in the peptide landscape: it is an approved medication in one country (Russia), with clinical trial data published primarily in Russian-language journals, and yet it remains largely unknown to Western regulatory bodies. This creates an epistemic challenge that I want to address directly — the data exists, but its accessibility and the degree to which it has undergone international peer review require us to hold it with appropriate nuance.
At a Glance
Property Detail
Type Synthetic analog of endogenous peptide (tuftsin)
Amino acid count 7
Primary mechanism BDNF modulation, monoamine neurotransmitter regulation, immunomodulation
Evidence level Russian clinical trials, human studies; limited Western replication
Regulatory status Approved in Russia as anxiolytic/nootropic; not approved in US/EU
Route of administration Intranasal
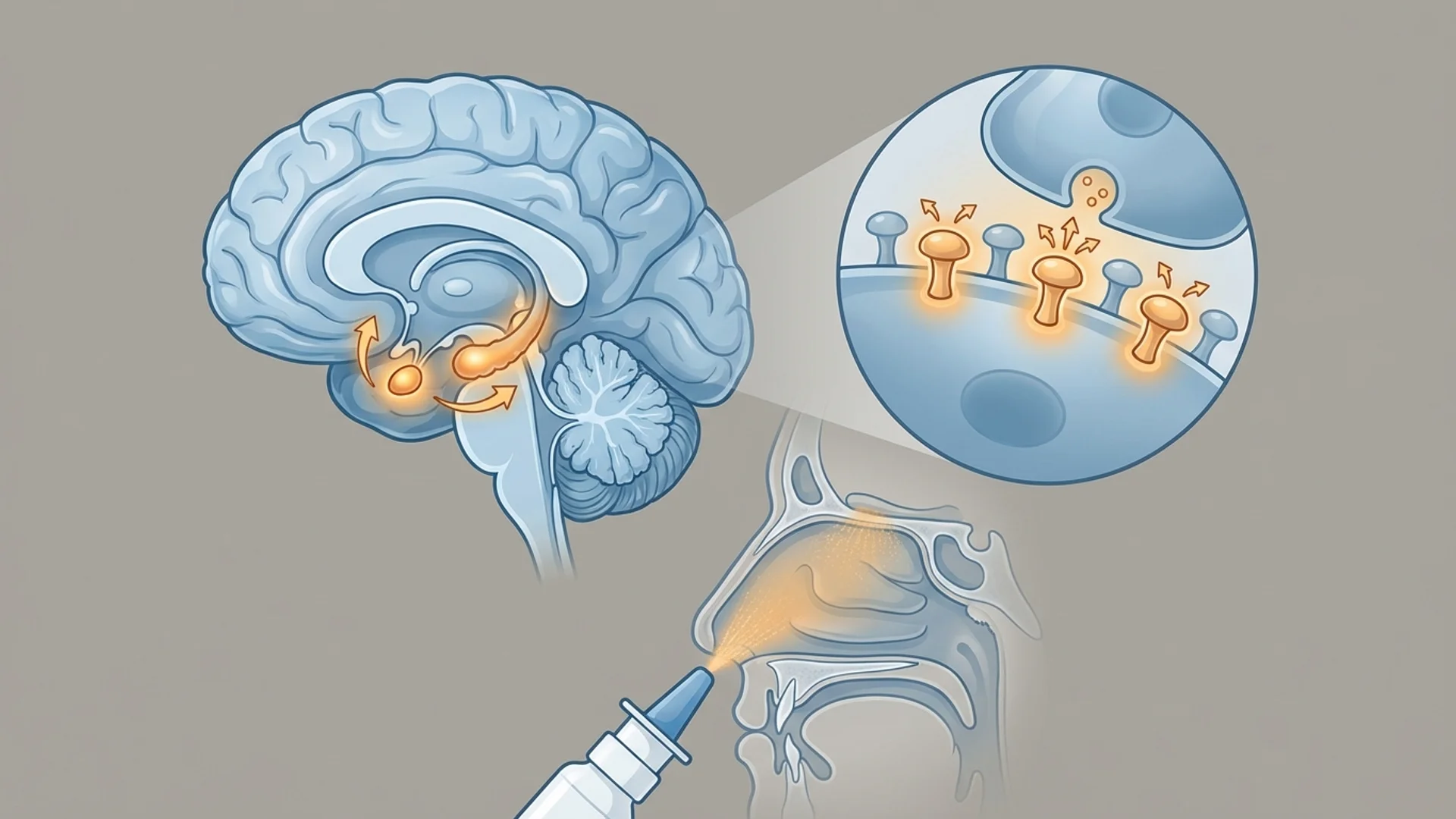
What Is Selank?
Selank is a synthetic heptapeptide developed at the Institute of Molecular Genetics of the Russian Academy of Sciences in the 1990s. It is an analog of tuftsin, a naturally occurring tetrapeptide (Thr-Lys-Pro-Arg) that is produced by enzymatic cleavage of the heavy chain of immunoglobulin G (IgG). The researchers extended the tuftsin sequence with a Pro-Gly-Pro tripeptide to enhance metabolic stability while retaining biological activity.

Tuftsin itself has well-documented immunomodulatory properties — it enhances phagocytosis, activates natural killer cells, and modulates cytokine production. Selank retains some of these immunomodulatory effects while adding anxiolytic and nootropic properties that are not characteristic of tuftsin alone.
Selank is approved in Russia under the name Selanc and is available as a 0.15% nasal spray. Its approved indications include generalized anxiety disorder and as a cognitive enhancer for conditions involving cognitive decline.
Mechanism of Action
Primary Pathways
- BDNF Modulation: Selank has been shown to increase the expression of brain-derived neurotrophic factor (BDNF) in the hippocampus and prefrontal cortex. BDNF is the principal neurotrophin involved in synaptic plasticity, learning, and memory. It also plays a role in mood regulation, and low BDNF levels are associated with depression and anxiety disorders. Selank’s effect on BDNF is thought to underlie both its anxiolytic and nootropic properties.
- Monoamine Neurotransmitter Modulation: Studies have demonstrated that Selank influences the metabolism of serotonin, dopamine, and norepinephrine in the brain. Specifically, it has been shown to stabilize enkephalin levels and modulate the expression of genes involved in serotonin transport and metabolism. This mechanism is conceptually similar to conventional anxiolytic medications, but the peptide appears to achieve these effects without the sedation, dependence, or withdrawal associated with benzodiazepines.
- GABAergic Modulation: Selank appears to enhance GABAergic neurotransmission — the primary inhibitory neurotransmitter system in the brain. Unlike benzodiazepines, which act as positive allosteric modulators at the GABA-A receptor, Selank’s GABAergic effects appear to be indirect, mediated through its effects on neuropeptide signaling and gene expression rather than direct receptor binding.
- Immunomodulatory Effects: Consistent with its tuftsin heritage, Selank retains significant immunomodulatory properties. It has been shown to modulate the expression of inflammatory cytokines, enhance phagocytic activity, and influence the Th1/Th2 balance. This dual anxiolytic-immunomodulatory profile is unusual among therapeutic agents and may be particularly relevant in conditions where anxiety and immune dysregulation co-occur.
Research Assessment
What the Evidence Shows
Claimed Effect Evidence Level Key Studies Assessment
Cognitive enhancement Russian clinical trials Kozlov 2019 Promising (needs Western replication)
BDNF upregulation Animal studies Inozemtseva 2008 Strong mechanistic data
Immunomodulation Animal + human studies Uchakina 2008 Promising
Serotonin metabolism modulation Animal studies Kozlovskii 2001 Preliminary
The Russian clinical data for Selank includes controlled human studies demonstrating anxiolytic efficacy comparable to benzodiazepines in patients with generalized anxiety disorder, but without the sedation, cognitive impairment, or dependence risk associated with benzodiazepines. These studies, reviewed by Kozlov and colleagues (2019), describe a favorable therapeutic profile.
The challenge is that much of this clinical trial data has been published in Russian-language journals and has not been replicated by independent Western research groups. This does not mean the data is invalid. Russian pharmaceutical research has a legitimate scientific tradition, and the Institute of Molecular Genetics is a respected institution. But the absence of independent international replication means we should hold the efficacy claims with appropriate epistemic humility.
What the Evidence Does Not Show
The long-term effects of Selank use have not been established in large-scale studies. The anxiolytic efficacy has not been demonstrated in Western clinical trial frameworks (randomized, double-blind, placebo-controlled trials with sample sizes that would satisfy FDA or EMA standards).
The nootropic effects, while reported in clinical studies and described by users, have not been characterized with the precision or rigor that would establish Selank as a proven cognitive enhancer.
Dosing
Route Dose Range Frequency Duration Source of Data
Subcutaneous 250-500 mcg Daily 10-20 days Clinical practice
Note: The intranasal dosing is based on the approved Russian pharmaceutical formulation. Duration of 10-14 days per course is the standard recommendation, with courses repeated as needed after a break.
Safety and Side Effects
Known
In the Russian clinical experience, Selank has demonstrated a favorable safety profile. No significant adverse events, sedation, or dependence have been reported. Unlike benzodiazepines, Selank does not appear to impair cognitive function or psychomotor performance, and no withdrawal syndrome has been documented upon cessation.
Theoretical Concerns
The long-term neurological effects of exogenous BDNF modulation are not fully understood. While BDNF enhancement is generally considered neuroprotective, sustained upregulation of BDNF has theoretical implications for neural circuit remodeling that have not been studied over extended periods.
Contraindications
No formal contraindications established. Caution in patients with active psychotic disorders (theoretical concern with monoamine modulation). Pregnancy and lactation (no safety data).
Drug and Supplement Interactions
Theoretical interactions with anxiolytic medications (benzodiazepines, SSRIs) due to overlapping mechanisms of action. No formal interaction studies have been conducted. Patients on psychotropic medications should consult their physician before adding Selank.
Clinical Perspective
Selank is a peptide that I discuss with patients who are looking for anxiolytic support without the side effects of conventional anxiolytic medications. The Russian clinical data is encouraging, and the mechanistic profile — BDNF modulation, monoamine stabilization, and GABAergic enhancement without direct receptor agonism — is exactly what you would want in an ideal anxiolytic.
The evidence hierarchy matters here. I am transparent with patients that the clinical trial data comes primarily from Russian research institutions and has not been independently replicated in Western clinical trial frameworks. This is not a reason to dismiss the data, but it is a reason to hold it with appropriate caution.
In my clinical experience, patients who use Selank intranasally typically report a calming effect within 15-30 minutes of administration, without the cognitive dulling associated with benzodiazepines. Several patients have described it as a “quiet clarity” — reduced anxiety without feeling sedated or disconnected. These are clinical observations, not controlled trial outcomes, and I present them as such.
What I find most interesting about Selank is its dual anxiolytic-immunomodulatory profile. Many of the patients I see — particularly those with chronic Lyme disease or post-COVID syndrome — present with both anxiety and immune dysregulation. A single agent that addresses both dimensions, if the clinical data holds, would be clinically valuable.
I do not position Selank as a replacement for established psychiatric treatment. For patients with severe anxiety disorders, evidence-based psychiatric care remains the standard of care. Selank is a tool I consider for patients who prefer to avoid benzodiazepines or who have not responded adequately to conventional approaches.

References
- Kozlov VA, et al. “Selank and its metabolic fragment Thr-Lys-Pro: mechanisms of action and review of preclinical and clinical studies.” Curr Protein Pept Sci. 2019;20(7):615-625.
- Zozulya AA, et al. “Selank: a novel anxiolytic peptide.” Vestn Ross Akad Med Nauk. 2001;(8):17-20.
- Seredenin SB, et al. “Mechanisms of anxiolytic effect of Selank.” Bull Exp Biol Med. 2009;148(4):612-614.
- Inozemtseva LS, et al. “Effect of Selank on BDNF gene expression.” Bull Exp Biol Med. 2008;146(11):583-585.
- Uchakina ON, et al. “Immunomodulatory effects of Selank in patients with anxiety-asthenic disorders.” Zh Nevrol Psikhiatr. 2008;108(5):71-75.
- Kozlovskii II, et al. “Effect of Selank on serotonin metabolism.” Bull Exp Biol Med. 2001;131(2):156-158.
Disclaimer: This peptide profile is intended for educational purposes. Selank is approved in Russia but is not approved by the FDA or EMA for any indication. Consult a qualified physician before pursuing any peptide therapy.