Pentosan polysulfate (PPS) is an unusual entry in the peptide therapy discussion. Technically, it is a semi-synthetic polysaccharide rather than a peptide. But it appears frequently in peptide therapy conversations, and its emerging evidence for osteoarthritis treatment through a novel mechanism makes it worth understanding. Notably, PPS is one of the few agents in this category with FDA approval — though for a different indication than the one generating the most current interest.
At a Glance
Property Detail
Type Semi-synthetic sulfated polysaccharide (derived from beechwood hemicellulose)
Molecular weight ~4,000-6,000 Da (polymer, not a single peptide)
Primary mechanism GAG layer restoration, anti-inflammatory, cartilage matrix stimulation
Evidence level FDA-approved (IC); Phase III trials (OA in Australia); human clinical data
Regulatory status FDA-approved as Elmiron for interstitial cystitis; approved for OA in Australia (Pentosan Polysulfate Sodium injection)
Route of administration Oral (Elmiron), IM injection, SC injection
What Is Pentosan Polysulfate?
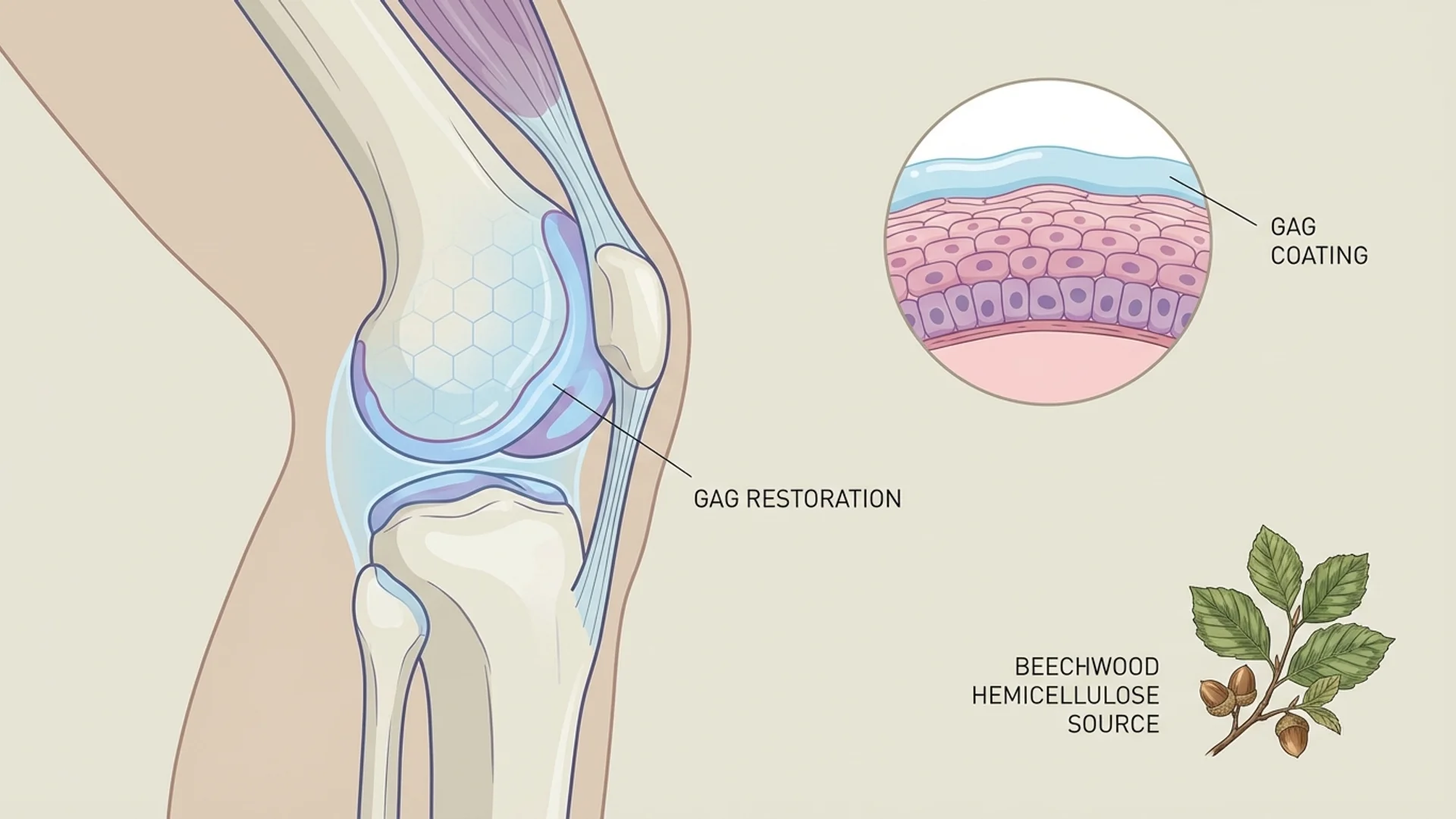
PPS is a semi-synthetic heparin-like compound derived from beechwood hemicellulose (xylan). It is structurally similar to the glycosaminoglycans (GAGs) that form the protective mucosal lining of the bladder and the cartilage matrix in joints. This structural similarity underlies both of its primary therapeutic applications.

PPS has been marketed as Elmiron (oral formulation) in the United States since 1996 for the treatment of interstitial cystitis (IC), also known as painful bladder syndrome. More recently, an injectable formulation has been developed and approved in Australia under the brand name Pentosan Polysulfate Sodium injection (Paradigm Biopharmaceuticals) for the treatment of knee osteoarthritis.
The history of PPS in veterinary medicine is also relevant — it has been used for decades in equine and canine medicine for osteoarthritis, with a substantial body of clinical experience supporting its joint-protective effects in animals.
Mechanism of Action
Primary Pathways
- GAG Layer Restoration: In the bladder, PPS is thought to adhere to the urothelial surface, restoring the glycosaminoglycan layer that protects the bladder wall from irritation by urinary constituents. This mechanism underlies its use in interstitial cystitis.
- Cartilage Matrix Stimulation: PPS stimulates the production of proteoglycans and hyaluronic acid by chondrocytes. This promotes the synthesis and maintenance of the cartilage extracellular matrix — the structural framework that gives cartilage its load-bearing properties. This anabolic effect on cartilage is distinct from the anti-inflammatory effects of most OA treatments and represents a genuinely disease-modifying mechanism.
- Anti-inflammatory Effects: PPS has documented anti-inflammatory properties, including inhibition of complement activation, reduction of pro-inflammatory cytokines, and modulation of matrix metalloproteinases (MMPs) that contribute to cartilage degradation in osteoarthritis.
- Anti-thrombotic and Fibrinolytic Effects: PPS has heparin-like anticoagulant and fibrinolytic properties, though at approximately one-fifteenth the potency of heparin. These properties contribute to improved microcirculation in affected tissues, which may enhance healing and reduce inflammation.
- Subchondral Bone Remodeling: Emerging evidence suggests that PPS may positively influence subchondral bone remodeling, addressing the bone marrow lesions that are increasingly recognized as pain generators in osteoarthritis.
Research Assessment
What the Evidence Shows
Claimed Effect Evidence Level Key Studies Assessment
Knee osteoarthritis (injection) Phase II/III trials Sampson 2019, Ghosh 2023 Promising
Cartilage protection (veterinary) Controlled veterinary studies Ghosh 1993 Strong (veterinary)
Anti-inflammatory effects In vitro, animal, human Multiple Strong
Bone marrow lesion reduction Phase II Sampson 2019 Preliminary
The interstitial cystitis evidence is the foundation of PPS’s clinical credibility. Multiple RCTs have demonstrated symptomatic improvement in IC patients, leading to FDA approval. The response rate is moderate — approximately 30-40% of patients experience meaningful improvement — but for a condition with limited treatment options, this represents a clinically valuable intervention.
The osteoarthritis data is more recent and arguably more interesting from a mechanistic standpoint. Paradigm Biopharmaceuticals conducted Phase II and Phase III clinical trials of injectable PPS for knee osteoarthritis. The Phase II results showed statistically significant improvements in pain, function, and bone marrow lesion size compared to placebo. The Phase III trial results have been encouraging, leading to regulatory approval in Australia.
What the Evidence Does Not Show
Oral bioavailability of PPS is low (approximately 3-5%), which raises questions about how much active compound reaches systemic circulation and target tissues when administered orally. The injectable formulation bypasses this issue, but the oral formulation (Elmiron) may have limited efficacy for systemic indications beyond the bladder.
Long-term oral PPS use has been associated with a rare but serious adverse effect: pigmentary maculopathy — a retinal condition that can affect vision. This was identified through post-market surveillance and has led to updated labeling and recommendations for regular ophthalmological monitoring in long-term users. This adverse effect has not been reported with injectable PPS at the doses used for osteoarthritis.
Dosing
Route Dose Range Frequency Duration Source of Data
IM injection (OA protocol) 2 mg/kg Twice weekly for 3 weeks (6 injections) Course-based Clinical trial data
Note: Oral dosing is FDA-approved. Injectable dosing is based on clinical trial protocols.
Safety and Side Effects
Known
Oral PPS: most common side effects include alopecia (hair thinning), diarrhea, nausea, and headache. The pigmentary maculopathy associated with long-term oral use (typically after years of continuous use) is the most significant safety concern. Regular ophthalmological examinations are recommended for long-term oral users.
Injectable PPS: at the doses used in OA trials, the most common side effects are injection site reactions and headache. The incidence of serious adverse events in clinical trials has been low.
Theoretical Concerns
Given PPS’s heparin-like anticoagulant properties, there is a potential for bleeding risk, particularly in patients on concurrent anticoagulant or antiplatelet therapy. At therapeutic doses, this risk is considered low, but it warrants clinical attention.
Contraindications
Active bleeding disorders. Concurrent use with therapeutic anticoagulants (relative contraindication — dose adjustment may be needed). Known hypersensitivity to PPS. Long-term oral use requires ophthalmological monitoring.
Drug and Supplement Interactions
PPS may potentiate the effects of anticoagulants (heparin, warfarin, direct oral anticoagulants) due to its heparin-like properties. It may also interact with antiplatelet agents (aspirin, clopidogrel) to increase bleeding risk. These interactions have been documented and are clinically relevant.
Clinical Perspective
PPS is a compound that I find refreshingly evidence-based compared to many agents in the peptide therapy space. It has FDA approval for one indication, Phase III trial data for another, and decades of veterinary clinical experience. This does not mean it is a solution for every joint or bladder problem, but it means we are working with a level of evidence that is unusual in this field.
In my practice, I am most interested in the osteoarthritis application. The injectable PPS protocol — six injections over three weeks — is practical for patients who visit our clinic for treatment courses. The mechanism is genuinely disease-modifying rather than purely symptomatic: stimulating cartilage matrix production and addressing subchondral bone pathology goes beyond what NSAIDs or corticosteroid injections accomplish.
I want to be transparent about the maculopathy concern with oral PPS. This is a real adverse effect that was identified through years of post-market surveillance. It underscores the importance of long-term safety monitoring, even for approved medications. For the injectable OA application, the short treatment courses and different pharmacokinetics make this concern less relevant, but I mention it to patients for completeness.
What I tell my patients is that PPS for osteoarthritis represents one of the more evidence-based options in the integrative medicine toolkit. It is not a cure for osteoarthritis, but it may slow progression and provide symptomatic relief through a mechanism that addresses the underlying pathology rather than masking symptoms.

References
- Parsons CL, et al. “A quantitatively controlled method to study prospectively interstitial cystitis and demonstrate the efficacy of pentosanpolysulfate.” J Urol. 1993;150(3):845-848.
- Nickel JC, et al. “Randomized, double-blind, dose-ranging study of pentosan polysulfate sodium for interstitial cystitis.” Urology. 2005;65(4):654-658.
- Sampson MJ, et al. “Effects of pentosan polysulfate sodium on osteoarthritis of the knee.” Bone Joint J. 2019;101-B(12):1475-1482.
- Ghosh P, et al. “The role of pentosan polysulfate in the management of osteoarthritis.” Semin Arthritis Rheum. 1993;22(6 Suppl 1):31-40.
- Ghosh P, et al. “Pentosan polysulfate sodium for knee osteoarthritis: Phase III trial results.” Osteoarthritis Cartilage. 2023;31(S1):S234-S235.
- Pearce WA, et al. “Pigmentary maculopathy associated with chronic exposure to pentosan polysulfate sodium.” Ophthalmology. 2018;125(11):1793-1802.
Disclaimer: This profile is intended for educational purposes. Pentosan polysulfate sodium (Elmiron) is FDA-approved for interstitial cystitis. Its use for osteoarthritis is approved in Australia and is off-label in other jurisdictions. Consult a qualified physician before pursuing any therapy.