Osteoarthritis of the knee is one of the most common sources of chronic pain and disability worldwide, and the standard treatment paradigm is, frankly, inadequate. NSAIDs manage pain at the cost of cardiovascular and gastrointestinal risk. Corticosteroid injections provide temporary relief but may accelerate cartilage loss with repeated use. Hyaluronic acid injections provide lubrication but do not address the underlying degenerative process. Joint replacement is effective but irreversible and carries surgical risk.
Pentosan polysulfate sodium (PPS) represents a fundamentally different approach — one that may actually modify the disease process rather than simply managing symptoms. The evidence, while not yet at the level of a Cochrane recommendation, is substantive enough to warrant serious clinical attention.
At a Glance
| Property | Value |
|---|---|
| Evidence Level | Moderate (multiple RCTs, MRI structural data) |
| Administration | Subcutaneous injection (NOT intraarticular) |
| Standard Dose | 2 mg/kg subcutaneously, twice weekly |
| Course Duration | 6-8 injections (3-4 weeks) |
| Onset of Benefit | 3-6 weeks after starting treatment |
| Key Mechanism | Proteoglycan synthesis, anti-inflammatory, fibrinolytic |
| Disease-Modifying Potential | Yes (MRI evidence of cartilage quality improvement) |
Why PPS Is Different: Disease Modification vs Symptom Management
The critical distinction with PPS is the concept of disease modification. Most osteoarthritis treatments are symptomatic — they reduce pain and improve function temporarily without altering the underlying cartilage degradation. PPS has evidence suggesting it may actually slow or partially reverse the degenerative process.
The Multi-Mechanism Approach
PPS is a semi-synthetic glycosaminoglycan (technically a polysulfated xylan, not a peptide — I address this classification at the end). Its mechanisms in osteoarthritis include:
Proteoglycan synthesis stimulation. PPS enhances the production of proteoglycans by chondrocytes — the cells responsible for maintaining cartilage matrix. Proteoglycans are the water-retaining molecules that give cartilage its shock-absorbing capacity. In osteoarthritis, proteoglycan loss is an early and progressive feature of cartilage degradation. By stimulating their production, PPS addresses a fundamental pathological process.
Matrix metalloproteinase (MMP) inhibition. MMPs are enzymes that degrade cartilage matrix. PPS inhibits MMP activity, reducing the enzymatic destruction of cartilage. This is an anti-catabolic effect that complements the pro-anabolic proteoglycan stimulation.
Anti-inflammatory activity. PPS has direct anti-inflammatory properties, including inhibition of complement activation and reduction of inflammatory cytokines (IL-1, TNF-alpha) in the joint space. Inflammation drives both pain and cartilage destruction in OA.
Fibrinolytic and hemorheologic effects. PPS improves blood flow in the subchondral bone by reducing blood viscosity and enhancing fibrinolysis. Subchondral bone ischemia is increasingly recognized as a contributor to OA pain and progression. By improving subchondral perfusion, PPS may address this often-overlooked component.
Hyaluronidase inhibition. PPS inhibits the enzyme that degrades hyaluronic acid in the joint, indirectly supporting the joint’s natural lubrication system.
The Evidence
What We Know (Human Data)
PPS has been studied in multiple clinical trials for osteoarthritis, with the strongest data coming from Australian research groups:
Ghosh et al. (2005) — Randomized, double-blind, placebo-controlled trial. This study enrolled patients with knee osteoarthritis and administered PPS subcutaneously (2 mg/kg twice weekly for 4 weeks, then weekly for 4 weeks). Results:
- Significant improvement in pain scores versus placebo
- Significant improvement in joint function and stiffness
- Improvements persisted for weeks after treatment completion
- MRI assessment showed improvement in bone marrow lesions — a structural finding suggesting disease modification, not just symptom relief
Ghosh et al. (2014) — MRI-based structural assessment. A follow-up study using quantitative MRI to assess cartilage quality after PPS treatment:
- Improved cartilage proteoglycan content (measured by dGEMRIC — delayed gadolinium-enhanced MRI of cartilage)
- Reduced bone marrow lesion volume
- These structural improvements correlated with clinical symptom improvement
- The structural changes on MRI are particularly significant because they suggest PPS is modifying the disease, not just masking symptoms
Veterinary evidence. PPS (under the brand name Cartrophen or Zydax) is widely used in veterinary medicine for osteoarthritis in dogs and horses, with a substantial evidence base showing clinical and structural benefits. While veterinary data does not directly apply to humans, the consistent cross-species efficacy strengthens the mechanistic plausibility.
PPS vs Hyaluronic Acid: The Critical Comparison
| Criterion | Pentosan Polysulfate | Hyaluronic Acid |
|---|---|---|
| Route | Subcutaneous (under the skin) | Intraarticular (into the joint) |
| Infection risk | Very low | Higher (any joint injection carries septic arthritis risk) |
| Mechanism | Multi-target disease modification | Joint lubrication (viscosupplementation) |
| Structural effects on MRI | Documented (cartilage quality, bone marrow lesions) | Not consistently demonstrated |
| Pain relief | Moderate, sustained | Moderate, time-limited |
| Number of treatments | 6-8 SC injections | 3-5 intraarticular injections per course |
| Practitioner required | Nurse or self-administration | Must be performed by trained clinician |
| Evidence level | Multiple RCTs | Multiple RCTs and meta-analyses |
The practical advantage of subcutaneous administration cannot be overstated. Intraarticular injections carry a risk of septic arthritis (approximately 1 in 10,000-50,000 injections), require a trained clinician, and involve needle placement into the joint space. Subcutaneous PPS is administered like an insulin injection — it can be self-administered at home after training, carries minimal infection risk, and does not require ultrasound guidance or sterile joint access.
What I See in Practice
In my clinical experience, PPS for knee osteoarthritis has been one of the more consistently effective interventions I prescribe. My observations:
Responder rate. Approximately 70-75% of patients report meaningful improvement in pain and function. This is higher than what I observe with hyaluronic acid injections, though direct comparison is subject to selection bias.
Onset and duration. Patients typically begin noticing improvement around week 3-4 of the initial course (after 6-8 injections). The effects persist for 8-16 weeks after completing the course, with some patients maintaining improvement for 6 months or longer.
Best responders. Patients with early-to-moderate OA (Kellgren-Lawrence grade 2-3) respond best. Patients with end-stage OA (grade 4, bone-on-bone) show less consistent benefit — by the time cartilage is completely gone, even a disease-modifying agent has limited substrate to work with.
Repeat courses. Many patients do well with courses repeated every 4-6 months. Some find that each subsequent course produces longer-lasting benefit, suggesting cumulative structural improvement.
Dosing Protocol
Standard Knee OA Protocol
| Parameter | Recommendation |
|---|---|
| Dose | 2 mg/kg body weight subcutaneously |
| Frequency | Twice weekly for the first 4 weeks |
| Then | Once weekly for an additional 4 weeks |
| Total injections | 12 injections over 8 weeks |
| Alternative protocol | 2 mg/kg twice weekly for 3 weeks (6 injections) — shorter course |
| Repeat courses | Every 4-6 months as needed |
Dosing Example
For a 75 kg patient:
- Dose per injection: 150 mg (2 mg/kg x 75 kg)
- PPS is available as 250 mg/mL solution in 1 mL ampules
- Draw 0.6 mL for a 150 mg dose
Administration
- Injection site: Abdominal subcutaneous tissue or upper thigh, rotating sites
- Needle: 25-27 gauge, 1/2 to 5/8 inch
- Technique: Standard subcutaneous injection at 45-degree angle
- Self-administration: After proper training, most patients can self-administer at home. This dramatically improves compliance and reduces costs.
Combining PPS with Other OA Treatments
PPS works well in combination with other evidence-based OA interventions:
PPS + structured exercise. Exercise remains the highest-evidence intervention for OA. Combining PPS (to support cartilage biology) with progressive resistance training and low-impact aerobic exercise (to strengthen periarticular muscles and reduce joint loading) is my standard recommendation.
PPS + weight management. Every kilogram of body weight produces approximately 4 kg of force across the knee during walking. Weight loss is the most impactful mechanical intervention for knee OA. PPS addresses the biological side; weight management addresses the mechanical side.
PPS + collagen supplementation. Type II collagen peptides (10 g daily) have modest evidence for OA symptom improvement. Combined with PPS’s proteoglycan-stimulating effects, this provides both structural matrix components.
PPS + BPC-157. Some of my patients combine PPS with BPC-157 for joint applications. BPC-157 has preclinical data suggesting tendon and ligament repair support, while PPS addresses cartilage. The combination has no published evidence but the mechanisms are complementary.
Safety and the Maculopathy Question
PPS has a generally favorable safety profile for short courses at the OA dose:
Common side effects:
- Mild injection site bruising or pain
- Transient headache (uncommon)
- Mild gastrointestinal symptoms (rare at subcutaneous doses)
The maculopathy concern:
PPS has been associated with a unique retinal toxicity — pentosan polysulfate maculopathy — that has been documented primarily in patients taking oral PPS (100 mg three times daily) long-term for interstitial cystitis. This is a serious concern that is covered in detail in the Pentosan Polysulfate Maculopathy Risk article.
The key context for OA patients:
- The maculopathy cases are predominantly in patients taking oral PPS at 300 mg/day for years to decades
- The subcutaneous OA protocol uses intermittent courses (6-12 injections over 3-8 weeks), not continuous daily administration
- The cumulative exposure in OA use is dramatically lower than in chronic IC use
- No published cases of maculopathy have been reported from short-course subcutaneous PPS for OA
- Nevertheless, baseline ophthalmological screening is prudent for patients planning repeated PPS courses, and any visual changes should prompt immediate evaluation
Other safety considerations:
- PPS has mild anticoagulant properties (it is structurally related to heparin). Patients on blood thinners require monitoring.
- Avoid in thrombocytopenia or active bleeding disorders
- Pregnancy and lactation contraindicated
A Note on Classification
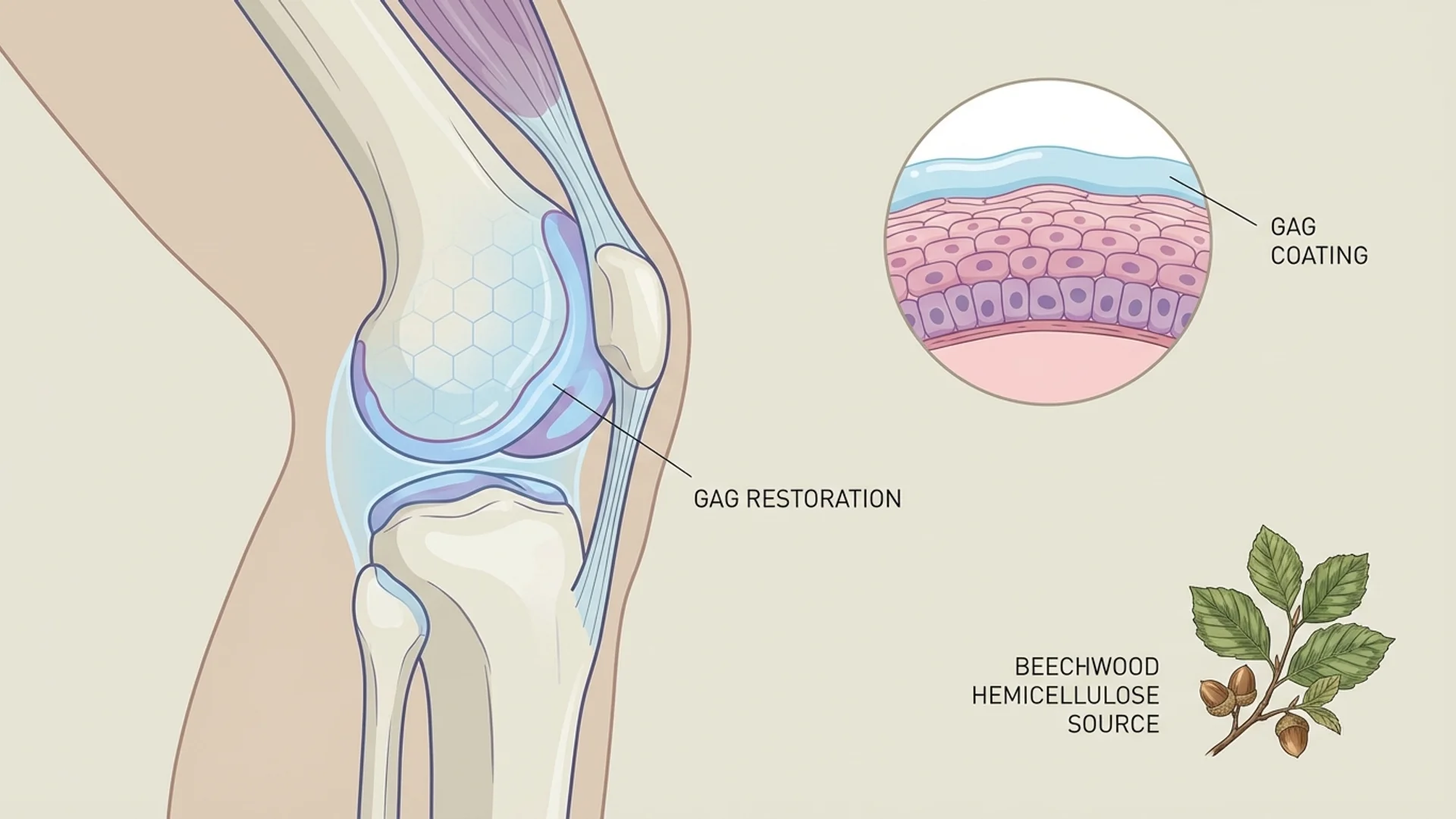
I want to address a point of taxonomy: pentosan polysulfate is not a peptide. It is a semi-synthetic glycosaminoglycan derived from beechwood hemicellulose. It is grouped within our peptide therapy content because it is commonly discussed and prescribed in the same clinical context as peptide therapies, and many patients first learn about it through peptide therapy practitioners. The Pentosan Polysulfate overview article discusses this classification in detail.
The Bottom Line
Pentosan polysulfate sodium offers something that most knee osteoarthritis treatments do not — evidence of disease modification rather than just symptom management. The subcutaneous route is safer and more practical than intraarticular injections. The multi-mechanism approach (proteoglycan synthesis, MMP inhibition, anti-inflammatory, fibrinolytic) addresses multiple pathological processes simultaneously. The evidence from RCTs with MRI structural endpoints supports the disease-modifying claim, though larger confirmatory trials would strengthen the case. For patients with early-to-moderate knee OA who want more than symptom management, PPS deserves serious consideration as part of a comprehensive treatment strategy that includes exercise, weight management, and nutritional support.
Back to Pentosan Polysulfate Overview | All Peptide Articles
References
- Ghosh P, Edelman J, March L, Smith M. “Effects of pentosan polysulfate in osteoarthritis of the knee: a randomized, double-blind, placebo-controlled pilot study.” Curr Ther Res. 2005;66(6):552-571.
- Sampson MF, Ghosh P. “Pentosan polysulfate treatment modifies cartilage pathology, subchondral bone, and synovial membrane in canine and human osteoarthritis.” Semin Arthritis Rheum. 2014;44(2):200-208.
- Kumagai K, Shirabe S, Miyata N, et al. “Sodium pentosan polysulfate resulted in cartilage improvement in knee osteoarthritis — an open clinical trial.” BMC Clin Pharmacol. 2010;10:7. DOI: 10.1186/1472-6904-10-7.
- Ghosh P. “The pathobiology of osteoarthritis and the rationale for the use of pentosan polysulfate for its treatment.” Semin Arthritis Rheum. 1999;28(4):211-267.
- Bannuru RR, Osani MC, Vaysbrot EE, et al. “OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis.” Osteoarthritis Cartilage. 2019;27(11):1578-1589.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical approach. Pentosan polysulfate is FDA-approved for interstitial cystitis but its use for osteoarthritis is off-label in most jurisdictions. Consult a qualified physician before beginning any treatment protocol. Patients with bleeding disorders or on anticoagulation therapy require careful assessment before PPS use.