KPV is one of the smallest peptides used in clinical practice — just three amino acids — yet its anti-inflammatory effects in preclinical models of intestinal inflammation are among the most impressive in the peptide literature. This article examines what the research shows, where the evidence stands, and how KPV fits into clinical practice for gut-related conditions.
At a Glance
Property Detail
Type Synthetic fragment of endogenous peptide
Amino acid count 3
Primary mechanism NF-kB inhibition, anti-inflammatory cytokine modulation
Evidence level Animal studies; limited clinical observation
Regulatory status Not approved; available through compounding
Route of administration Oral, SC injection, rectal (compounded)
What Is KPV?
KPV is the C-terminal tripeptide fragment of alpha-melanocyte-stimulating hormone (alpha-MSH), consisting of the amino acids lysine-proline-valine. Alpha-MSH is a 13-amino-acid neuropeptide produced by the pituitary gland and by various immune cells. It is best known for its role in skin pigmentation, but alpha-MSH is also a potent endogenous anti-inflammatory molecule.
Research in the 1990s and 2000s established that the anti-inflammatory activity of alpha-MSH resides primarily in its C-terminal tripeptide — KPV. This was a significant finding because it demonstrated that the melanogenic (pigmentation) effects and the anti-inflammatory effects of alpha-MSH are mediated by different regions of the molecule. KPV retains the anti-inflammatory activity without the pigmentation effects.
This separation of activities makes KPV clinically attractive. Alpha-MSH itself would be problematic as a therapeutic agent because of its pigmentation effects. KPV provides the anti-inflammatory benefits without this limitation.
Mechanism of Action
Primary Pathways
- NF-kB Inhibition: The best-characterized mechanism of KPV is its inhibition of nuclear factor kappa B (NF-kB), the master transcription factor that drives inflammatory gene expression. KPV enters cells and directly interferes with the nuclear translocation of NF-kB, reducing the transcription of pro-inflammatory cytokines including TNF-alpha, IL-1beta, IL-6, and IL-8. This is a direct intracellular action, not a receptor-mediated effect.
- Inflammatory Cytokine Suppression: Downstream of NF-kB inhibition, KPV reduces the production of multiple pro-inflammatory mediators. In colitis models, KPV has been shown to reduce mucosal levels of TNF-alpha, IL-1beta, and myeloperoxidase (a marker of neutrophil infiltration) by 50-70%.
- Immune Cell Modulation: KPV modulates the behavior of several immune cell types. It reduces neutrophil infiltration into inflamed tissues, modulates macrophage activation, and appears to influence the balance between pro-inflammatory and regulatory immune responses in the gut mucosa.
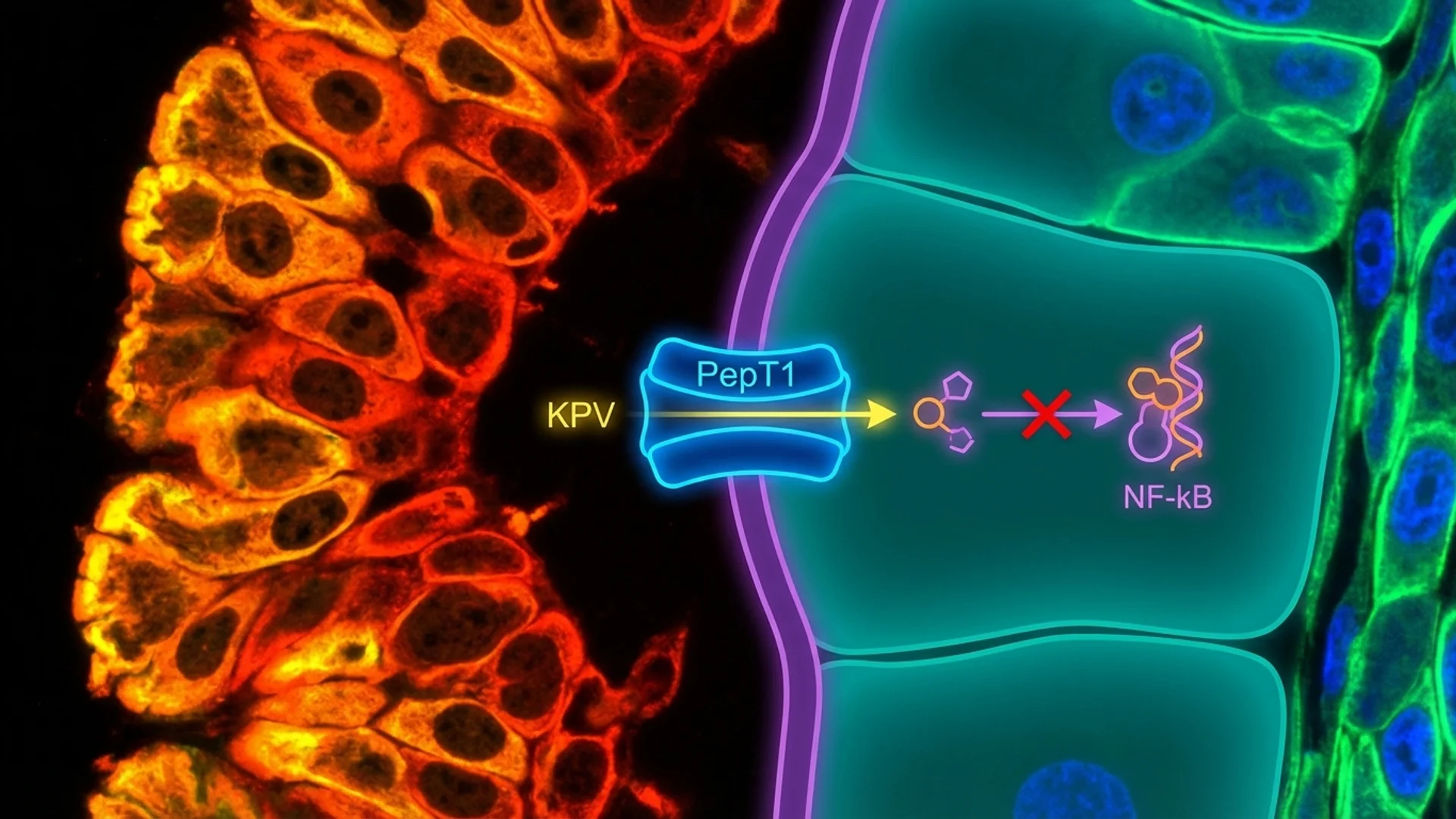
- Intestinal Epithelial Transport: KPV has been shown to be transported across the intestinal epithelium via the peptide transporter PepT1, which is upregulated in inflamed intestinal tissue. This means that KPV may be preferentially absorbed in inflamed gut segments — a form of passive targeting that could enhance its efficacy when administered orally for intestinal inflammation.
Research Assessment
What the Evidence Shows
Claimed Effect Evidence Level Key Studies Assessment
NF-kB inhibition In vitro, animal studies Brzoska 2008 Strong mechanistic data
Mucosal healing Animal studies Dalmasso 2008 Promising
Skin inflammation reduction Animal studies Luger 2003 Preliminary
Wound healing In vitro Cutuli 2000 Preliminary
The preclinical data for KPV in models of inflammatory bowel disease is compelling. Dalmasso and colleagues (2008) demonstrated that oral administration of KPV significantly reduced colitis severity in the dextran sodium sulfate (DSS) mouse model — a standard experimental model for IBD. The peptide reduced clinical disease scores, histological inflammation, and pro-inflammatory cytokine levels.
The finding that KPV is transported by PepT1, and that PepT1 is upregulated in inflamed intestinal tissue, provides an elegant mechanistic explanation for why an orally administered tripeptide might have preferential activity in inflamed gut tissue.
What the Evidence Does Not Show
There are no published human clinical trials of KPV for any indication. The evidence base consists entirely of in vitro studies, animal models, and clinical observation from physicians who have incorporated KPV into their practice.
The translation from DSS-induced colitis in mice to human inflammatory bowel disease is not straightforward. DSS colitis is an acute chemical injury model, while human IBD is a chronic, relapsing condition with complex immunological underpinnings. KPV’s efficacy in one model does not guarantee efficacy in the other.
Dosing
Route Dose Range Frequency Duration Source of Data
Subcutaneous 200-500 mcg Daily 4-8 weeks Clinical practice
Rectal (compounded suppository) 500 mcg-1 mg Once daily 4-8 weeks Clinical practice
Note: All dosing is based on clinical practice. There are no human dose-finding studies for KPV.
Safety and Side Effects
Known
Given the absence of formal human studies, the safety data is limited to clinical observation. No serious adverse events have been reported in clinical use. The parent molecule, alpha-MSH, has a well-characterized safety profile, and KPV as a smaller fragment would be expected to have at least as favorable a safety profile.
Theoretical Concerns
Because KPV suppresses NF-kB signaling and inflammatory responses, there is a theoretical concern about immunosuppression with prolonged use, particularly the potential for reduced defense against gastrointestinal infections. This is a concern that applies to any anti-inflammatory intervention, not uniquely to KPV, but it warrants attention in clinical practice.
Contraindications
Active gastrointestinal infection (theoretical concern — suppressing inflammation during active infection could be counterproductive). Significant immunosuppression. Pregnancy and lactation (no data).
Drug and Supplement Interactions
No formal interaction studies exist. Theoretical caution with other immunosuppressive or anti-inflammatory medications (additive immunosuppressive effects). KPV’s mechanism of action through NF-kB inhibition could theoretically interact with medications that also affect NF-kB signaling (corticosteroids, certain biologics).
Clinical Perspective
KPV is a peptide that I find mechanistically elegant and clinically interesting, but I want to be honest about where the evidence stands. We are working with strong preclinical data and clinical observation. We do not have controlled human trial data.
In my practice, I use KPV primarily in the context of gut healing protocols — particularly for patients with inflammatory bowel disease, post-infectious irritable bowel syndrome, and gut barrier dysfunction. I typically combine it with BPC-157 and address the underlying causes of gut inflammation simultaneously.
What I have observed is that patients with documented intestinal inflammation who receive KPV as part of a comprehensive gut-healing protocol often report improvement in symptoms — reduced abdominal pain, improved stool consistency, reduced bloating — within 2-4 weeks. The challenge, as always with multi-component protocols, is attributing these improvements specifically to KPV.
The PepT1 transport mechanism is what makes KPV particularly interesting to me from a clinical standpoint. The idea that an anti-inflammatory peptide is preferentially absorbed in inflamed intestinal tissue is exactly the kind of targeted delivery that we strive for in medicine. If this mechanism functions as demonstrated in the preclinical studies, oral KPV could deliver its anti-inflammatory effects precisely where they are needed most.
What I tell my patients is that KPV has a strong mechanistic rationale and encouraging preclinical data, but that we are still waiting for the human clinical trials that would move it from “promising” to “established.” In the meantime, for patients who have not responded adequately to conventional approaches, KPV represents a reasonable option with a favorable risk profile — provided they understand the limitations of the current evidence.

References
- Dalmasso G, et al. “PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation.” Gastroenterology. 2008;134(1):166-178.
- Brzoska T, et al. “Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, anti-inflammatory and protective effects.” Endocr Rev. 2008;29(5):581-602.
- Kannengiesser K, et al. “Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease.” Inflamm Bowel Dis. 2008;14(3):324-331.
- Luger TA, et al. “Alpha-melanocyte-stimulating hormone as a mediator of tolerance induction.” Pathobiology. 2003;70(3):156-163.
- Cutuli M, et al. “Antimicrobial effects of alpha-MSH peptides.” J Leukoc Biol. 2000;67(2):233-239.
- Catania A, et al. “The melanocortin system in control of inflammation.” ScientificWorldJournal. 2010;10:1840-1853.
Disclaimer: This peptide profile is intended for educational purposes. KPV is not approved by any regulatory agency for therapeutic use. Consult a qualified physician before pursuing any peptide therapy.