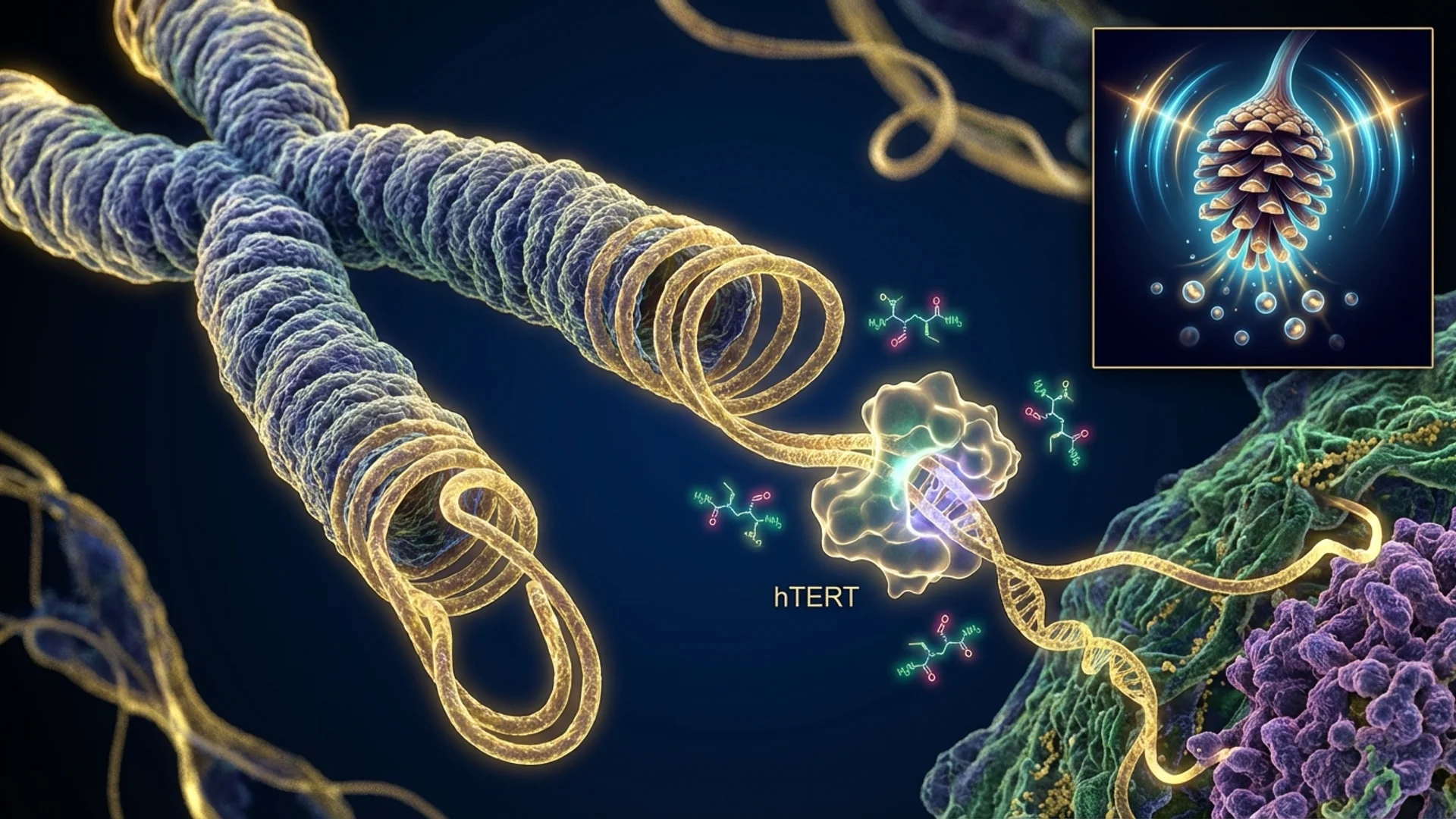
Epithalon may be the most ambitious peptide in the longevity medicine toolkit — it aims to address one of the fundamental mechanisms of cellular aging by activating telomerase, the enzyme that maintains telomere length. The promise is significant. But as with any intervention that touches fundamental cellular biology, the nuance matters enormously, and the gap between what has been demonstrated and what is claimed deserves honest examination.
At a Glance
Property Detail
Type Synthetic analog of endogenous pineal peptide (epithalamin)
Amino acid count 4
Primary mechanism Telomerase activation, melatonin regulation
Evidence level Animal studies, limited human studies (primarily Khavinson group)
Regulatory status Not approved; research compound
Route of administration SC injection, IV
What Is Epithalon?
Epithalon (also spelled Epitalon) is a synthetic tetrapeptide with the sequence Ala-Glu-Asp-Gly. It was developed by Professor Vladimir Khavinson at the St. Petersburg Institute of Bioregulation and Gerontology as a synthetic version of epithalamin, a polypeptide extract derived from the bovine pineal gland.
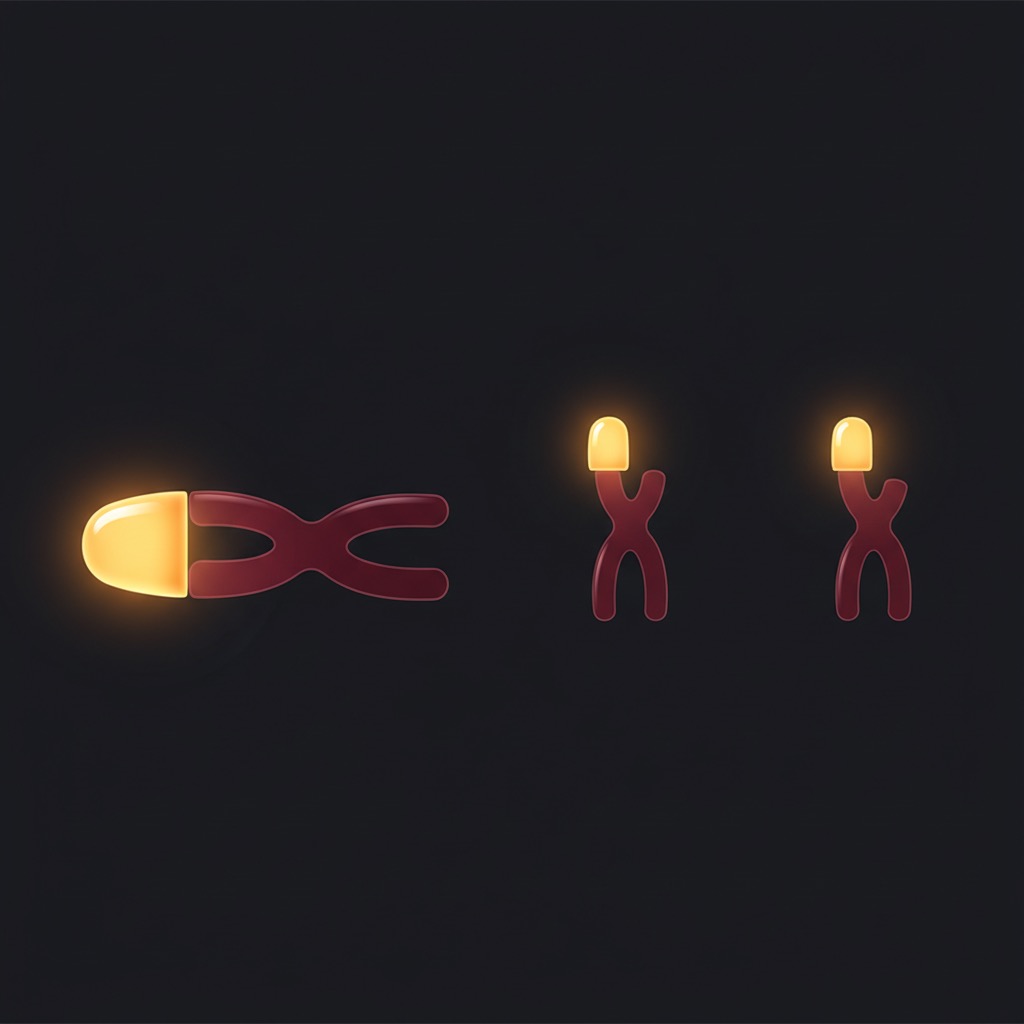
Khavinson’s research program, spanning several decades, has focused on the concept of “bioregulation” — the idea that short peptides can serve as gene regulators, interacting with specific DNA sequences to modulate gene expression. Epithalon is the most prominent product of this research program.
The connection to the pineal gland is relevant because the pineal gland produces melatonin, the hormone that regulates circadian rhythm and has documented antioxidant properties. Pineal function declines with age, and melatonin levels decrease correspondingly. Epithalon appears to support pineal function and melatonin production, which is one of its proposed mechanisms for anti-aging effects.
Mechanism of Action
Primary Pathways
- Telomerase Activation: The primary mechanism attributed to Epithalon is the activation of telomerase — specifically, the upregulation of the telomerase reverse transcriptase (hTERT) gene. Telomerase adds repetitive nucleotide sequences (TTAGGG in humans) to the ends of chromosomes, counteracting the telomere shortening that occurs with each cell division. In cell culture studies, Epithalon has been shown to increase telomerase activity and extend the replicative lifespan of human somatic cells.
- Melatonin Regulation: Epithalon has been shown to restore the normal circadian production of melatonin in elderly subjects, increasing both peak nocturnal melatonin levels and the amplitude of the circadian melatonin cycle. Given melatonin’s role in sleep, antioxidant defense, and immune regulation, this effect may contribute to the observed anti-aging effects independently of telomerase activation.
- Chromatin Remodeling: Research from the Khavinson laboratory suggests that Epithalon interacts with specific regions of DNA, possibly through interaction with histone proteins, to influence chromatin structure and gene accessibility. If confirmed, this would represent a mechanism of epigenetic regulation by a short peptide — a concept that is intriguing but requires independent validation.
- Antioxidant Defense Enhancement: In animal models, Epithalon has been associated with increased activity of superoxide dismutase (SOD) and other antioxidant enzymes, along with reduced lipid peroxidation markers. These effects could be downstream of telomerase activation, melatonin enhancement, or direct gene regulatory effects.
Research Assessment
What the Evidence Shows
Claimed Effect Evidence Level Key Studies Assessment
Lifespan extension (animals) Animal studies Anisimov 2003 Preliminary
Melatonin restoration (humans) Small human studies Korkushko 2006 Preliminary
Immune function improvement (elderly) Small human studies Khavinson 2000 Preliminary
Cancer incidence reduction (animals) Animal studies Anisimov 2002 Preliminary
In cell culture studies, Epithalon has demonstrated the ability to increase telomerase activity and extend the replicative lifespan of human fibroblasts. Khavinson and colleagues reported that treatment with Epithalon increased the number of cell divisions before senescence by approximately 44% compared to untreated controls.
In animal studies, primarily in mice and rats, Anisimov and colleagues demonstrated that chronic administration of Epithalon was associated with modest but statistically significant increases in mean lifespan. Notably, some of these studies also reported reduced incidence of spontaneous tumors in the Epithalon-treated groups — an important finding given the theoretical concern about telomerase activation and cancer.
Limited human studies in elderly subjects have shown improvements in melatonin production, immune parameters (including restoration of T-cell function), and self-reported quality of life measures. These studies are generally small, open-label, and come predominantly from Khavinson’s research group.
What the Evidence Does Not Show
The human evidence for Epithalon is limited in both scale and rigor. The studies that exist are small, often lack proper placebo controls, and come predominantly from a single research group. Independent replication by Western laboratories has been minimal.
The critical question of whether exogenous telomerase activation translates into meaningful clinical anti-aging effects in humans has not been answered. Telomere length is associated with aging and age-related diseases, but the relationship between telomere length and actual health outcomes is complex. Simply lengthening telomeres does not necessarily translate into improved healthspan or lifespan.
The oncological safety question remains insufficiently resolved. While the animal data suggests that Epithalon does not increase cancer incidence — and may actually reduce it — the theoretical concern is real. Telomerase is reactivated in approximately 85% of human cancers. The distinction between telomerase activation in normal somatic cells (potentially beneficial) and in pre-malignant cells (potentially harmful) requires more investigation.
Dosing
Route Dose Range Frequency Duration Source of Data
Note: This dosing is based on the protocols used in the Khavinson research group’s studies and adopted in clinical practice. It is not based on formal dose-finding clinical trials.
Safety and Side Effects
Known
In the published studies and clinical observation, Epithalon has not been associated with serious adverse events. Injection site reactions are the most commonly reported side effect. The animal studies showing reduced rather than increased cancer incidence provide some reassurance, though these were conducted in specific mouse strains and may not be directly generalizable.
Theoretical Concerns
The telomerase-cancer question is the central safety concern with Epithalon. While the available data does not support the concern, the theoretical risk of promoting malignant transformation through telomerase activation has not been definitively excluded. This is particularly relevant in patients with known genetic predisposition to cancer or with a history of malignancy.
Contraindications
Active malignancy (strong precautionary principle). History of malignancy (relative contraindication — requires careful risk-benefit discussion). Pregnancy and lactation (no data). Children and adolescents (no data; also not applicable given the anti-aging indication).
Drug and Supplement Interactions
No drug interactions have been formally studied. Theoretical interactions with melatonin supplements (additive effects on melatonin levels) and with cancer treatments (the telomerase activation mechanism could theoretically interfere with therapies targeting telomerase).
Clinical Perspective
Epithalon occupies a complicated position in my clinical thinking. The mechanistic rationale is compelling — addressing one of the hallmarks of aging at the molecular level is precisely what longevity medicine aspires to do. And the available data, while limited, is more encouraging than I would have expected given the ambition of the claims.
But I want to be transparent about the limitations. The evidence comes primarily from one research group. The human studies are small. The long-term oncological safety has not been established in large human populations. And the fundamental question — whether telomerase activation in adult somatic cells produces meaningful clinical benefits — remains open.
In my practice, I discuss Epithalon with patients who are pursuing comprehensive longevity protocols and who have a sophisticated understanding of the evidence hierarchy. I am clear about what the data shows and what it does not. For patients who choose to proceed, I use it in short cycles (10-20 days, 2-3 times per year), with baseline and follow-up assessment of telomere length, melatonin levels, and comprehensive cancer screening.
I do not use Epithalon in patients with a history of malignancy or with known genetic predisposition to cancer. The theoretical risk, while unconfirmed, warrants this precaution until we have more definitive safety data.
The honest answer is that Epithalon deserves continued research. It addresses a real biological mechanism of aging, and the early data is promising. But “promising” and “proven” are not the same thing, and patients deserve to know the difference.
References
- Khavinson VK, et al. “Peptide Epitalon activates chromatin at the old age.” Neuro Endocrinol Lett. 2003;24(5):329-333.
- Khavinson VK, et al. “Effect of Epithalon on the lifespan increase in Drosophila melanogaster.” Mech Ageing Dev. 2000;120:141-149.
- Anisimov VN, et al. “Effect of Epitalon on biomarkers of aging, life span and spontaneous tumor incidence in female Swiss-derived SHR mice.” Biogerontology. 2003;4(4):193-202.
- Anisimov VN, et al. “Epithalon decelerates aging and suppresses development of breast adenocarcinomas in transgenic her-2/neu mice.” Bull Exp Biol Med. 2002;134(2):187-190.
- Korkushko OV, et al. “Effect of peptide preparation epithalamin on the circadian rhythm of melatonin secretion in elderly people.” Bull Exp Biol Med. 2006;141(6):696-698.
- Khavinson VK. “Peptides and Ageing.” Neuro Endocrinol Lett. 2002;23(Suppl 3):11-144.
Disclaimer: This peptide profile is intended for educational purposes. Epithalon is not approved by any regulatory agency for therapeutic use. The telomerase-cancer relationship requires ongoing investigation. Consult a qualified physician before pursuing any peptide therapy.