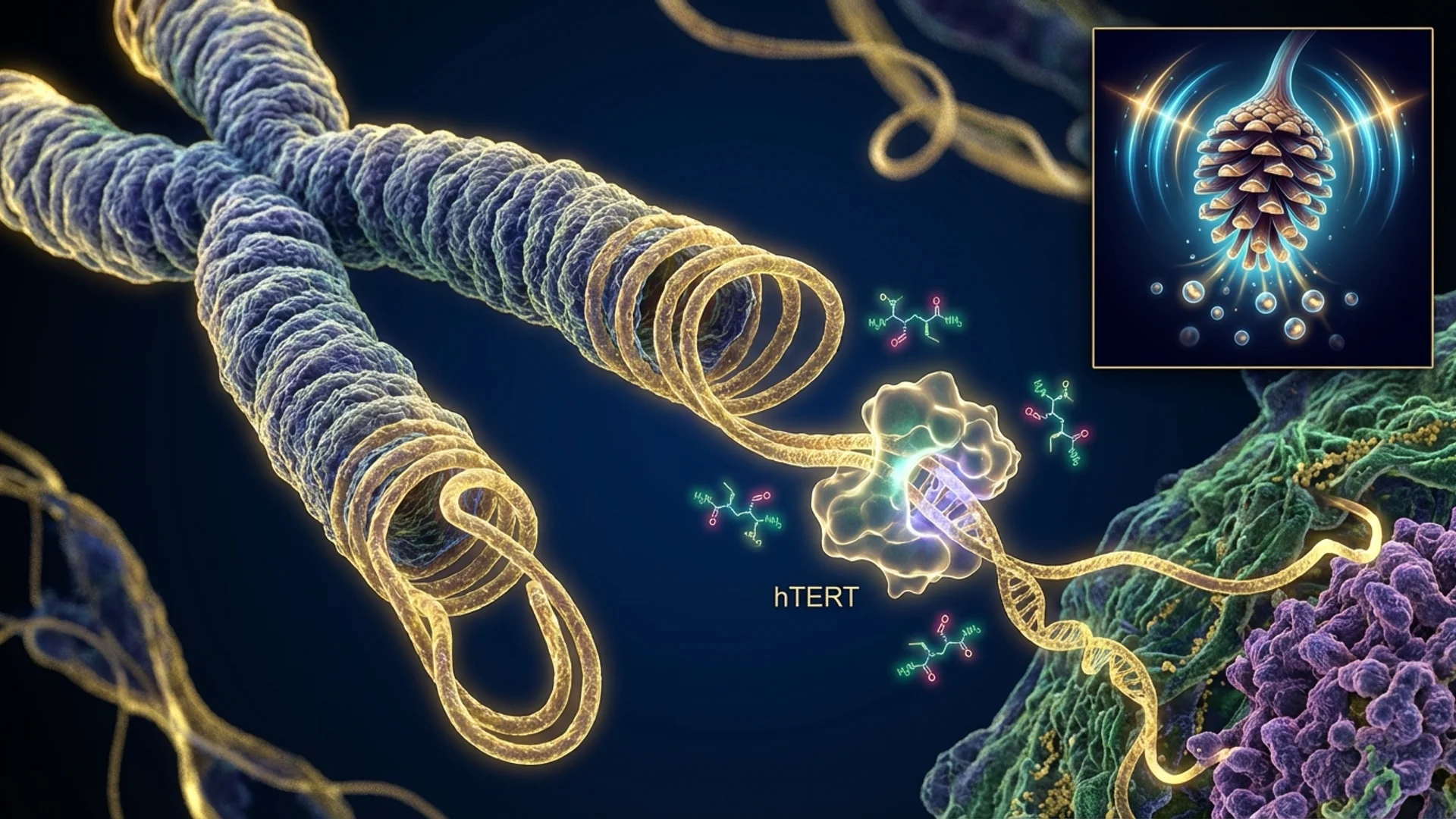
Epithalon (also called Epitalon or Epithalone) occupies a unique position in the peptide landscape: it has the most consistent animal longevity data of any peptide, a plausible mechanism confirmed at the cellular level, and essentially no large-scale human clinical trials. This creates a situation where the dosing protocol is well-established within the longevity medicine community but is derived primarily from the research of one group — Professor Vladimir Khavinson and colleagues at the St. Petersburg Institute of Bioregulation and Gerontology — rather than from internationally replicated dose-finding studies.
Let me be direct about what we know and where the gaps are.
At a Glance
| Property | Value |
|---|---|
| Evidence Level | Emerging (animal lifespan data strong; human data limited to cell lines and case reports) |
| Standard Dose | 5-10 mg subcutaneously, daily |
| Cycle Length | 10-20 days |
| Frequency | 1-2 courses per year |
| Key Mechanism | Telomerase activation, telomere elongation |
| Route | Subcutaneous injection (preferred); IV (clinical research) |
| Monitoring | Telomere length testing before and after |
Epithalon Dosage: The Khavinson Protocol
The dosing protocol for Epithalon comes almost entirely from the work of Vladimir Khavinson, who has published over 200 papers on pineal gland peptides and bioregulation over four decades. Epithalon is a synthetic tetrapeptide (Ala-Glu-Asp-Gly) that replicates the active sequence of epithalamin, a pineal gland extract.
The Standard Protocol
| Parameter | Protocol |
|---|---|
| Dose | 5-10 mg per day |
| Route | Subcutaneous injection |
| Frequency | Once daily, typically evening |
| Duration | 10 days (standard) to 20 days (extended) |
| Courses per year | 1-2, spaced at least 4-6 months apart |
| Best time of day | Evening (aligns with melatonin synthesis cycle) |
Where This Protocol Comes From
Khavinson’s animal studies. The foundational research used epithalamin (the pineal extract) and later Epithalon (the synthetic peptide) in multiple animal models:
- In Drosophila: 10-16% median lifespan extension
- In rats: up to 25% increase in mean lifespan with repeated courses of 10-day administration
- In mice: consistent lifespan extension across multiple studies, with the peptide administered in 10-day courses at intervals
The dosing in these animal studies was translated to human-equivalent doses using standard allometric scaling, arriving at the 5-10 mg daily range.
Khavinson’s human cell studies. The 2003 study in human fetal lung fibroblasts demonstrated that Epithalon activated telomerase and induced telomere elongation, with cells treated with Epithalon dividing significantly more times than untreated controls before reaching the Hayflick limit (Khavinson et al., 2003). The 2025 Springer confirmation further validated telomere lengthening via telomerase upregulation in human cell lines.
Clinical practice protocols. The 10-20 day, 1-2 times per year protocol has become the standard in longevity medicine clinics worldwide, based on Khavinson’s research and the pharmacokinetic rationale that telomerase activation does not require continuous peptide presence — the enzyme, once activated, continues its work during the rest period.
Pharmacokinetic Rationale for Cycling
The cycling schedule is not arbitrary. The logic is:
-
Telomerase activation is a trigger, not a sustained process. Epithalon activates the TERT gene (telomerase reverse transcriptase), which produces telomerase enzyme. Once the gene is expressed, the enzyme operates independently of ongoing Epithalon presence.
-
Telomere elongation is slow. Adding measurable telomere length takes weeks to months. A 10-day course initiates the process; the cellular effects continue long after the peptide is cleared.
-
Annual assessment. By spacing courses 6 months apart, you create a natural assessment window — measure telomere length before each course to evaluate whether the previous course had a measurable effect.
-
Precautionary principle. Continuous telomerase activation raises theoretical concerns about enabling abnormal cell proliferation. Intermittent dosing is considered safer, though this specific risk has not been demonstrated with Epithalon in any published study.
The Evidence for This Protocol
What We Know (Human Cell Data)
The cellular evidence is the strongest mechanistic support for Epithalon:
- Khavinson et al. (2003) demonstrated telomerase activation and telomere elongation in human fetal lung fibroblasts, with treated cells exceeding the Hayflick limit by approximately 10 additional population doublings
- The 2025 Springer study confirmed telomere lengthening via telomerase upregulation in human cell lines, providing independent validation of the core mechanism
- A widely cited case report described a 7.9-year biological age reduction in a patient receiving an Epithalon-inclusive longevity protocol — though this must be interpreted with extreme caution as a single case using multiple interventions simultaneously
What We See in the Lab (Animal Data)
The animal lifespan data is where Epithalon’s evidence is most compelling:
- Rats (Anisimov et al., 2003): Repeated 10-day courses of epithalamin/Epithalon extended mean lifespan by approximately 25% in female rats. Tumor incidence was reduced in treated animals.
- Mice: Consistent lifespan extension across multiple strains and studies.
- Drosophila: 10-16% median lifespan extension.
- Consistency across species: The fact that Epithalon extends lifespan in both invertebrate and vertebrate models, through a conserved mechanism (telomerase), strengthens the translational argument — though the gap between animal lifespan studies and human longevity remains vast.
Importantly, the animal studies also found that Epithalon reduced spontaneous tumor incidence. This counterbalances the theoretical concern about telomerase activation promoting cancer, though the distinction between cancer cells (which typically already have active telomerase) and normal somatic cells (which do not) is important.
What I See in Practice
In my clinical experience, Epithalon is one of the most commonly requested longevity peptides, and I use it within a structured protocol that includes baseline and follow-up telomere testing.
What I observe:
Telomere length changes. In patients who undergo serial telomere testing (we use the validated qPCR method), approximately 60-70% show stabilization or modest increase in telomere length after one year of biannual Epithalon courses. The remaining 30-40% show no measurable change or continued (though potentially slowed) shortening. This is clinical observation, not controlled data — the placebo rate of telomere stabilization is not known in this population.
Subjective reports. Patients commonly report improved sleep quality during and after Epithalon courses (consistent with pineal peptide activity and melatonin production support), improved energy levels, and enhanced skin quality. These are self-reported and subject to placebo effects, but they are consistent enough across patients that I consider them clinically relevant observations.
No adverse effects. I have not observed significant adverse effects from Epithalon at the standard dosing protocol. Mild injection site reactions are the most common complaint.
Practical Dosing Protocol
Beginner Epithalon Protocol (Conservative)
| Parameter | Recommendation |
|---|---|
| Dose | 5 mg subcutaneously, daily |
| Duration | 10 days |
| Timing | Evening (6-9 PM) |
| Courses per year | 1 |
| Monitoring | Telomere length at baseline and 3 months post-course |
Standard Epithalon Protocol
| Parameter | Recommendation |
|---|---|
| Dose | 10 mg subcutaneously, daily |
| Duration | 10 days |
| Timing | Evening (6-9 PM) |
| Courses per year | 2 (spaced 6 months apart) |
| Monitoring | Telomere length at baseline, 3 months, and 6 months |
Extended Protocol (Advanced)
| Parameter | Recommendation |
|---|---|
| Dose | 10 mg subcutaneously, daily |
| Duration | 20 days |
| Timing | Evening (6-9 PM) |
| Courses per year | 1-2 |
| Monitoring | Telomere length, biological age markers, epigenetic clocks |
Administration
- Injection site: Abdominal subcutaneous tissue, rotating sides
- Reconstitution: Add bacteriostatic water to lyophilized powder (typically 2 mL per 10 mg vial)
- Storage: Reconstituted solution refrigerated (2-8 degrees C), use within 21 days
- Evening timing rationale: Epithalon supports melatonin synthesis via pineal gland stimulation. Evening administration aligns with the natural melatonin production cycle.
Monitoring: How to Know If It Is Working
Telomere testing is the only objective way to evaluate Epithalon’s primary mechanism. I recommend:
- Baseline telomere length before the first course (multiple testing methods exist — ensure you use the same method for all measurements)
- 3-month follow-up after completing the first course
- Pre-second-course measurement at 6 months
- Annual tracking to establish a trend line
The nuance matters here: single-point telomere measurements are noisy. Individual readings can vary by 5-10% depending on the method. What you want to see is a trend over 2-3 measurements — stabilization or lengthening rather than the expected age-related shortening of approximately 20-40 base pairs per year.
Combining Epithalon with Other Longevity Interventions
Epithalon is rarely used in isolation in longevity protocols. Common combinations:
Epithalon + NAD+ precursors (NMN/NR). These target different aging mechanisms — telomere lengthening (Epithalon) versus cellular energy and DNA repair (NAD+). The combination addresses two distinct hallmarks of aging. See the Epithalon vs NMN comparison for a detailed mechanistic analysis.
Epithalon + melatonin. Since Epithalon supports endogenous melatonin production via pineal stimulation, some clinicians reduce exogenous melatonin during Epithalon courses to avoid redundancy. Others maintain it. There is no data to guide this decision.
Epithalon + exercise and caloric restriction. Both exercise and caloric restriction independently support telomere maintenance. Combining these with Epithalon is a reasonable multi-mechanism approach.
Epithalon + rapamycin. Some longevity protocols combine telomere support (Epithalon) with mTOR modulation (rapamycin). These target entirely different aging pathways and there is no expected pharmacological interaction.
Safety and Considerations
Epithalon has a clean safety profile across published data:
- No significant adverse effects reported in animal studies, even at high doses and over long durations
- Reduced tumor incidence in multiple animal studies (counterintuitively, given the telomerase mechanism)
- No immunosuppression or immune dysregulation documented
- Mild injection site reactions are the most common human complaint
Theoretical concerns:
- Cancer risk via telomerase activation. This is the most frequently raised concern. The counterargument: cancer cells already express telomerase (it is one of the hallmarks of cancer). Epithalon’s effect on normal somatic cells is to restore a mechanism that was active during development and gradually silenced with aging. The animal data showing reduced tumor incidence with Epithalon supports the safety argument, but it cannot be considered definitive.
- Insufficient human safety data. The absence of large-scale, long-term human trials means we cannot rule out rare or long-latency adverse effects.
Contraindications:
- Active malignancy (theoretical telomerase concern warrants avoidance until more data exists)
- Pregnancy and lactation (no safety data)
- Active autoimmune disease (insufficient data on immune effects)
The Bottom Line
Epithalon dosing follows the Khavinson protocol: 5-10 mg subcutaneously daily for 10-20 day courses, 1-2 times per year. The mechanism — telomerase activation and telomere elongation — is confirmed in human cells and produces consistent lifespan extension in animal models. The gap is the absence of controlled human longevity trials, which means we are extrapolating from strong preclinical data and clinical observation rather than human RCTs. For patients pursuing evidence-informed longevity optimization under physician supervision, Epithalon represents one of the most mechanistically compelling peptides available, with the honest caveat that the human evidence is not yet definitive.
Back to Epithalon Overview | All Peptide Articles
References
- Khavinson VKh, Bondarev IE, Butyugov AA. “Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells.” Bull Exp Biol Med. 2003;135(6):590-592. DOI: 10.1023/A:1025493705728.
- Anisimov VN, Khavinson VKh, Popovich IG, et al. “Effect of Epitalon on biomarkers of aging, life span and spontaneous tumor incidence in female Swiss-derived SHR mice.” Biogerontology. 2003;4(4):193-202. DOI: 10.1023/A:1025114230714.
- Khavinson VKh, Morozov VG. “Peptides of pineal gland and thymus prolong human life.” Neuro Endocrinol Lett. 2003;24(3-4):233-240.
- Anisimov VN, Khavinson VKh. “Peptide bioregulation of aging: results and prospects.” Biogerontology. 2010;11(2):139-149. DOI: 10.1007/s10522-009-9249-8.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical approach. Epithalon is not approved by the FDA or EMA. It is not a substitute for individualized medical care. Consult a qualified physician before beginning any peptide protocol. Telomerase activation in the context of active malignancy is contraindicated.