At a Glance
| Property | Value |
|---|---|
| Evidence Level | Moderate |
| Primary Use | Differential diagnosis and clinical comparison |
| Key Mechanism | Both parasites invade and destroy red blood cells, but differ in species biology, vector, and treatment |
Two Parasites, One Target: Your Red Blood Cells
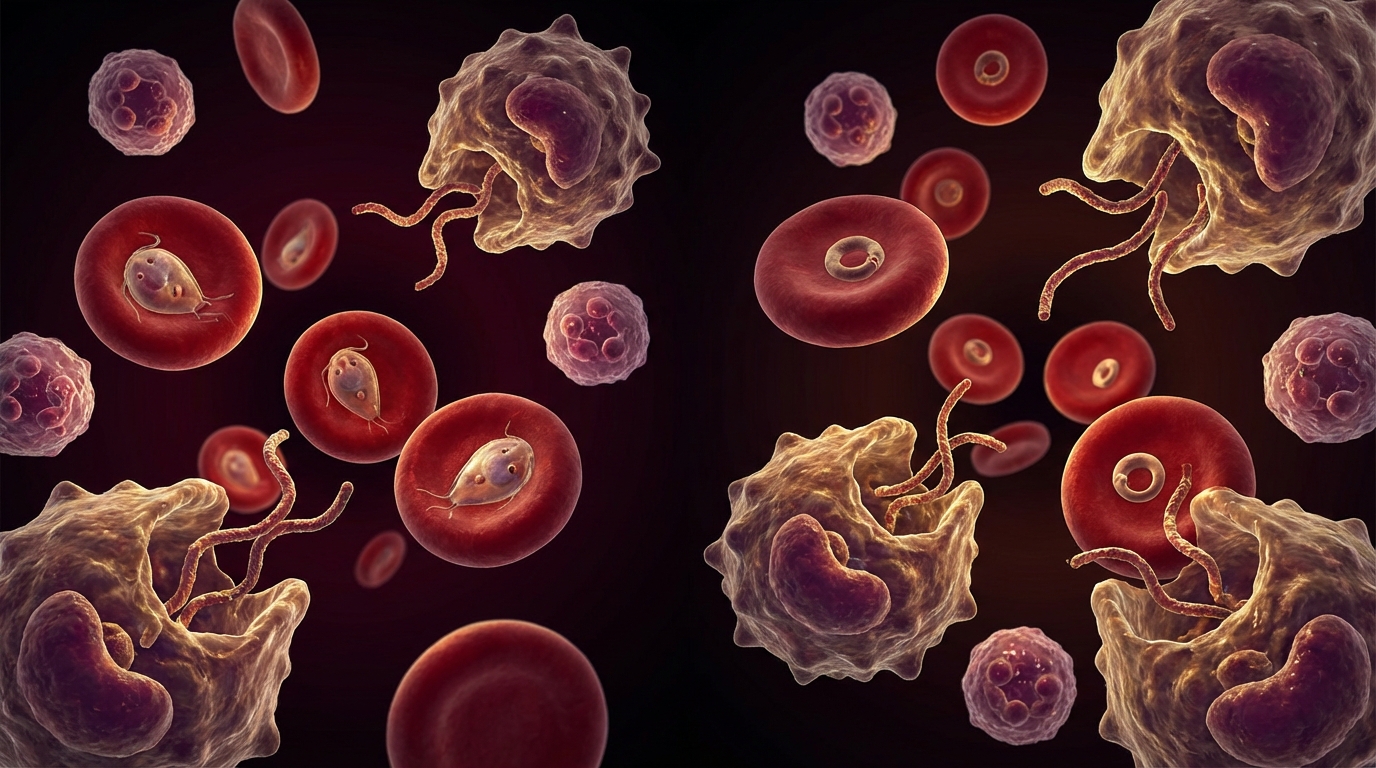
If you have been diagnosed with Babesia — or suspect you have it — you have probably noticed the comparison to malaria. Physicians call babesiosis “the malaria of the North,” and for good reason: both parasites invade red blood cells, both cause hemolytic anemia, both produce cyclical fevers, and both can be fatal in vulnerable populations.
But the similarities end there. Understanding where these two diseases diverge is critical for accurate diagnosis and effective treatment. This is what the research actually says about how these parasites compare.
The Biology: Same Strategy, Different Parasites
Both Babesia and Plasmodium (the genus that causes malaria) are apicomplexan protozoan parasites. They share an evolutionary strategy — invading erythrocytes and hijacking them as reproductive nurseries. But they diverged hundreds of millions of years ago, and the differences are clinically significant.
Babesia Species
- Primary human pathogens: Babesia microti (Northeast/Midwest US), Babesia duncani (West Coast US), Babesia divergens (Europe)
- Vector: Ixodes ticks (the same ticks that transmit Lyme disease)
- Reservoir: White-footed mice, voles, and other small mammals
- Lifecycle in humans: Merozoites invade red blood cells, replicate by budding into 2-4 daughter cells (forming the characteristic tetrad or “Maltese cross”), and rupture the cell to release new merozoites [1]
Plasmodium Species (Malaria)
- Primary human pathogens: P. falciparum (most lethal), P. vivax, P. ovale, P. malariae, P. knowlesi
- Vector: Anopheles mosquitoes
- Reservoir: Humans (for most species)
- Lifecycle in humans: Sporozoites first infect liver cells (hepatic stage), then release merozoites into the bloodstream to invade red blood cells. P. vivax and P. ovale can form dormant liver stages (hypnozoites) causing relapse months to years later [2]
The critical difference: Babesia has no liver stage. It goes directly from the tick bite into the bloodstream and into red blood cells. This means there are no dormant liver forms to worry about — but it also means that Babesia can be transmitted through blood transfusion, which is why it is now the most common transfusion-transmitted parasitic infection in the United States.
Symptom Comparison
| Feature | Babesia | Malaria |
|---|---|---|
| Fever pattern | Irregular, sometimes cyclical every 2-4 days | Classic cyclical: 48h (vivax, ovale, falciparum), 72h (malariae) |
| Air hunger | Hallmark symptom, very common | Less commonly reported |
| Night sweats | Drenching, very characteristic | Common (during fever resolution) |
| Hemolytic anemia | Mild to severe depending on parasitemia | Mild to severe; falciparum can exceed 20% parasitemia |
| Jaundice | Moderate cases | Common in severe falciparum |
| Splenomegaly | Common | Very common |
| Cerebral involvement | Rare | P. falciparum causes cerebral malaria |
| Renal failure | In severe cases | P. falciparum causes blackwater fever |
| Fatigue | Profound, often persists beyond parasite clearance | Resolves with parasite clearance typically |
| Co-infections | Frequently co-occurs with Lyme and Bartonella | Tropical co-infections (dengue, typhoid) |
The Air Hunger Distinction
Here is what I tell my patients: air hunger is Babesia’s calling card. While malaria patients certainly experience respiratory distress — particularly in severe P. falciparum with ARDS or severe anemia — the specific sensation of unsatisfied breathing at rest with normal oxygen saturation is far more characteristic of Babesia. In our clinical experience, this symptom pattern is one of the most reliable clinical indicators distinguishing the two.

Diagnostic Differences
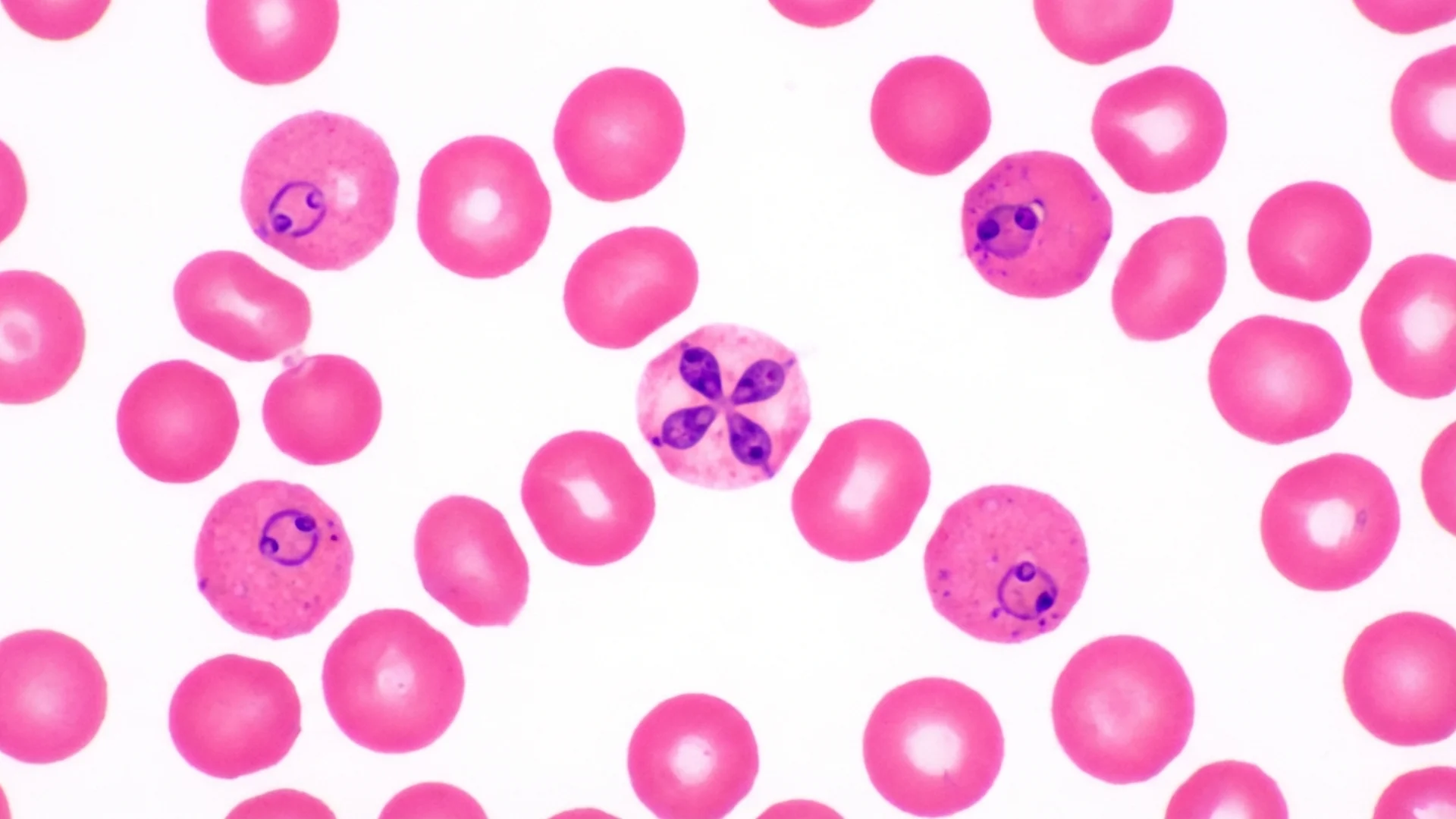
Microscopy
Under the microscope, both parasites appear as ring-form trophozoites inside red blood cells — which is exactly why Babesia is sometimes misidentified as malaria in laboratories not experienced with tick-borne infections.
The distinguishing features:
- Babesia: Tetrad forms (“Maltese cross”) are pathognomonic but not always present. Multiple parasites per red blood cell are common. No hemozoin (malaria pigment) is produced.
- Malaria: Banana-shaped gametocytes are pathognomonic for P. falciparum. Schuffner’s dots appear with P. vivax. Hemozoin pigment is present.
The key laboratory clue: Babesia does not produce hemozoin pigment. If your blood smear shows intraerythrocytic ring forms but no brown-black pigment granules, Babesia should be suspected over malaria [1].
Advanced Testing
| Test | Babesia | Malaria |
|---|---|---|
| Blood smear | Ring forms, tetrads, no pigment | Ring forms, gametocytes, pigment present |
| PCR | Species-specific PCR available | Gold standard for species identification |
| Rapid antigen test | Not widely available | HRP-2 and pLDH rapid tests available |
| Serology | IgM/IgG (4-fold rise or single high titer) | Limited utility |
| FISH | Highly sensitive for B. microti | Not standard |
Geographic Consideration
This is where clinical context matters enormously. If your patient has never traveled to a malaria-endemic region but lives in or has visited the northeastern United States, upper Midwest, or northern Europe — and presents with hemolytic symptoms — Babesia is far more likely than malaria. Conversely, a traveler returning from sub-Saharan Africa with cyclical fevers has malaria until proven otherwise.
In my clinical practice, I see patients who have been worked up for malaria based on their blood smear findings, only to have the diagnosis corrected to Babesia when geographic and clinical context is properly considered.
The Evidence
What We Know (Human Data)
The epidemiological divergence between these parasites is stark. Malaria infects approximately 250 million people annually worldwide (WHO 2023), while Babesia accounts for roughly 2,000-3,000 reported cases per year in the United States — though the actual number is almost certainly much higher due to underdiagnosis and underreporting [3].
A landmark study by Vannier and Krause in the New England Journal of Medicine established that babesiosis case fatality rates in hospitalized patients range from 6-9%, rising to 20% or higher in immunocompromised individuals — comparable to severe malaria in non-endemic settings [1].
Co-infection data from endemic areas shows that up to 40% of patients with Lyme disease in the northeastern US may have concurrent Babesia infection (Krause et al., 1996), and these co-infected patients experience more severe symptoms and longer illness duration than those with either infection alone.
What I See in Practice
In our clinical experience, the patients we see with Babesia almost always have it as a co-infection rather than a standalone disease. The typical presentation is a chronic Lyme patient who has been partially treated but continues to have fatigue, air hunger, and night sweats that do not resolve with anti-Borrelia therapy alone.
What I observe in practice is that Babesia requires its own specific treatment — antibiotics targeting Borrelia do not treat Babesia. This is perhaps the most important clinical distinction: if you are treating a tick-borne infection and respiratory symptoms persist, you must consider whether a concurrent parasite is being missed.

Treatment: Completely Different Protocols
This is where the comparison matters most. Malaria drugs do not treat Babesia, and Babesia drugs do not treat malaria (with some overlap in artemisinin compounds).
Babesia Treatment
- First-line: Atovaquone + azithromycin (7-10 days for acute; longer for chronic/relapsing)
- Alternative: Clindamycin + quinine (more side effects, reserved for severe cases)
- Severe cases: Exchange transfusion when parasitemia exceeds 10% or in asplenic patients
- Herbal adjuncts: Cryptolepis sanguinolenta, Artemisia annua — emerging research on botanical options
- Relapsing cases: Extended combination therapy, sometimes months
Malaria Treatment
- P. falciparum: Artemisinin-based combination therapy (ACT), IV artesunate for severe cases
- P. vivax/ovale: Chloroquine + primaquine (for liver hypnozoites; requires G6PD testing)
- Severe malaria: IV artesunate, exchange transfusion in extreme cases
The Artemisinin Overlap
One interesting area of overlap is Artemisia annua (sweet wormwood), the source of artemisinin — the Nobel Prize-winning antimalarial. Research suggests that artemisinin-derived compounds also have activity against Babesia species. A Johns Hopkins-supported study found that Cryptolepis sanguinolenta demonstrated significant in vitro activity against Babesia duncani, with Artemisia annua showing additive effects in combination [4].
This is an area where integrative medicine and conventional pharmacology converge. The data is promising but preliminary — large human trials are still needed.
Practical Application
When to Think Babesia Instead of Malaria
- Patient has no travel history to malaria-endemic regions
- Lives in or has visited tick-endemic areas (Northeast US, upper Midwest, northern Europe)
- Blood smear shows ring forms without hemozoin pigment
- Air hunger is a prominent symptom
- Co-infection with Lyme disease is known or suspected
- Night sweats are drenching and persistent
When to Test for Both
- Travel history includes both tick-endemic and malaria-endemic regions
- Blood smear is ambiguous
- Patient is immunocompromised (both can be severe)
- Symptoms do not respond to initial targeted therapy
Safety and Considerations
- Both infections can be fatal. Babesia in asplenic or immunocompromised patients and malaria (particularly P. falciparum) require urgent management.
- Transfusion-transmitted babesiosis is a recognized risk. Blood donors in endemic areas should be screened.
- Herxheimer reactions can occur during Babesia treatment, as with other antimicrobial therapies.
- Self-diagnosis based on symptom comparison is not recommended. Definitive diagnosis requires laboratory testing.
The Bottom Line
Babesia and malaria are evolutionary cousins that share a strategy of red blood cell parasitism, but they are clinically, geographically, and therapeutically distinct. If you are in a temperate tick-endemic region with hemolytic symptoms, unexplained air hunger, and a normal malaria workup — Babesia deserves a place on your differential. Here is what the evidence shows: these are different diseases that require different treatments, and confusing them leads to treatment failure.
References
- Vannier E, Krause PJ. Human babesiosis. N Engl J Med. 2012;366(25):2397-2407. PMID: 22716978
- White NJ. Malaria pathophysiology. In: Manson’s Tropical Diseases. 24th ed. Elsevier; 2024.
- Centers for Disease Control and Prevention. Babesiosis surveillance — United States, 2011-2019. MMWR Surveill Summ. 2023;72(2):1-12.
- Zhang Y, Alvarez-Manzo HS, Leone J, et al. Botanical medicines Cryptolepis sanguinolenta, Artemisia annua, Scutellaria baicalensis, Polygonum cuspidatum, and Alchornea cordifolia demonstrate inhibitory activity against Babesia duncani. Front Cell Infect Microbiol. 2021;11:624745. PMID: 33816335