Featured
Low NK Cell Function: What It Means and What to Do
Understanding low natural killer cell function in chronic illness. What NK cells do, why function drops, how to test, and evidence-based strategies to restore immune surveillance.
Condition
Hyperthermia, mistletoe therapy, immune support, and integrative approaches alongside conventional cancer treatment. Complementary, not alternative — always evidence-based.
Hyperthermia cancer treatment evidence from RCTs and 35 years of clinical use. How moderate and extreme WBH synergize with chemo and radiation therapy.

Why St. George Hospital uses 2 extreme hyperthermia sessions at 41.6-41.8°C for Lyme eradication, not 5-6 moderate sessions. The science behind the protocol.
Honest guide to whole-body hyperthermia side effects. What happens during treatment, common reactions, Herxheimer responses, recovery timeline, and contraindications.
Whole body hyperthermia treatment explained by a physician. How controlled fever therapy works for Lyme, cancer, and post-COVID at St. George Hospital.
Mistletoe extract (Iscador, Helixor) for cancer: mechanism, NK cell activation, quality of life evidence, injection protocols, and the German clinical tradition.
The immune suppression cascade: how Lyme disease triggers EBV, HHV-6, and CMV reactivation, and how these viruses further suppress immunity in a vicious cycle.
A clinician's guide to integrative oncology. What complementary therapies have evidence, how they work alongside conventional treatment, and what 35 years of clinical experience at Klinik St. Georg have shown.
Dihexa safety analysis by Dr. Julian Douwes. The HGF/c-Met oncogenic pathway concern, retracted research, failed clinical trials, and what patients must understand.
How Thymosin Alpha-1 supports cancer immunotherapy. T-cell activation, dendritic cell maturation, clinical trial data, and practical protocols.
How physicians enhance weak immune responses and calm overactive ones. Evidence-based immune modulation explained by Dr. Julian Douwes.
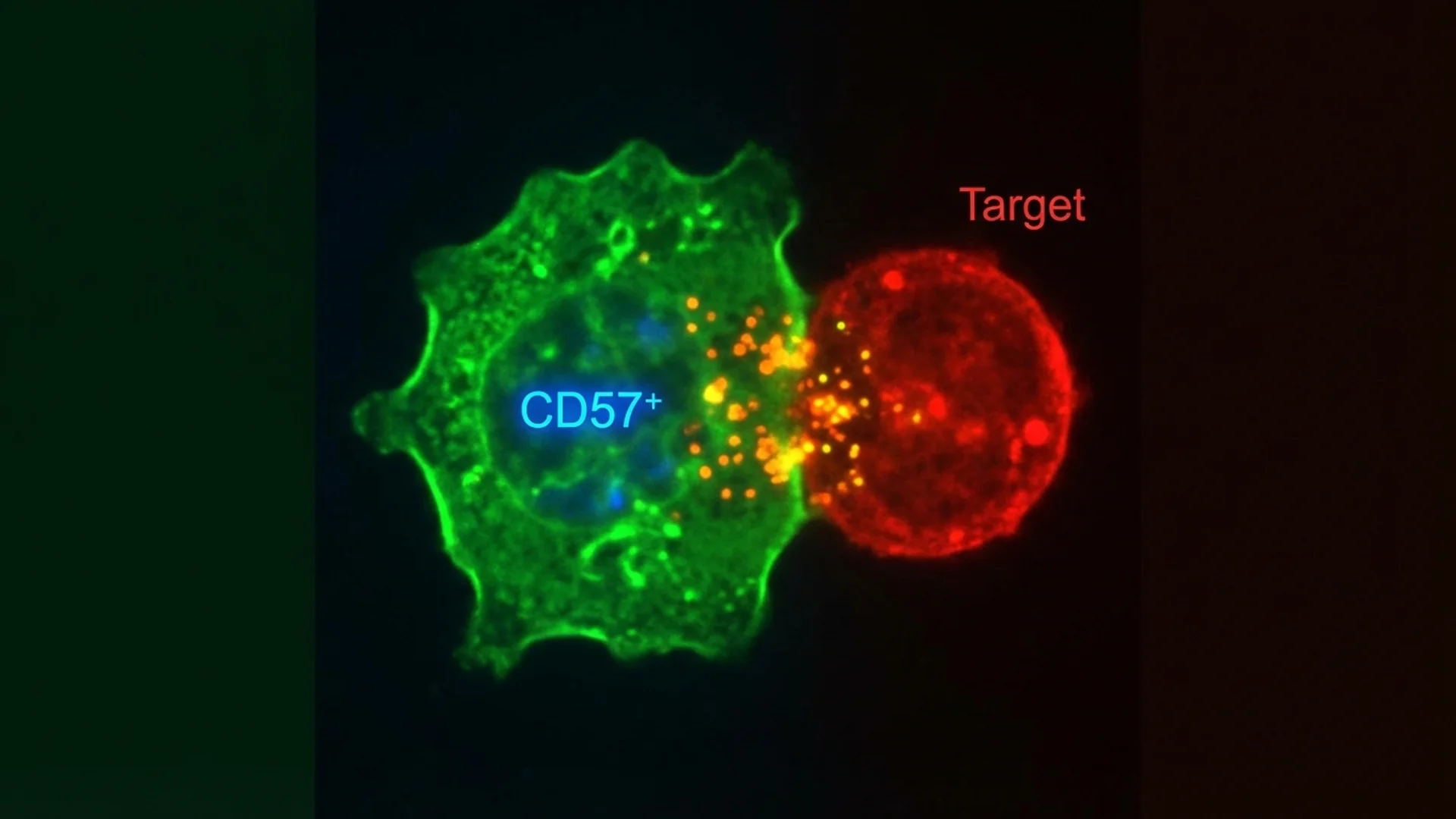
What NK cells do, why CD57 matters in Lyme disease, and how to support natural killer cell function. By Dr. Julian Douwes.
Schedule a consultation to discuss your case with Dr. Julian Douwes and the clinical team at St. George Hospital.
Complete Guide
In-depth reference by Dr. Julian Douwes
I want to state this clearly at the outset, because it is the single most important principle guiding our work: integrative oncology is complementary to conventional cancer treatment. It is not a replacement for surgery, chemotherapy, radiation, or immunotherapy. We work alongside your existing oncologist, not instead of them.
At St. George Hospital, we have treated cancer patients for over 35 years. In that time, we have never advised a patient to abandon standard oncological care. What we offer are evidence-based supportive and complementary therapies that can enhance the effectiveness of conventional treatment, reduce its side effects, strengthen the immune system, and improve quality of life during and after cancer treatment.
This distinction matters enormously. The landscape of cancer care is unfortunately populated with practitioners who make extraordinary claims about alternative cures. We are not among them. Our approach is grounded in published clinical evidence, and where evidence is preliminary, we say so.
St. George Hospital’s commitment to integrative oncology began with my father, Dr. Friedrich Douwes, who founded our integrative approach in the late 1980s. He recognized early — at a time when the concept was considered fringe — that cancer patients benefited from a broader therapeutic framework than conventional oncology alone could provide.
Over the decades, our hospital became one of the leading centers in Europe for combining conventional oncology with hyperthermia, immune support, high-dose intravenous vitamin C, and mistletoe therapy. We have treated patients from over 60 countries, and our protocols have been refined through tens of thousands of treatment cycles.
My father passed away in 2022, but the medical philosophy he established continues to guide our work. Today, under the clinical leadership of our oncology team — including Dr. Martin Rossner, Dr. Frederick Gotte, and Dr. Daniela Hudi — we continue to advance integrative oncology with the same commitment to evidence and patient welfare that defined his career.
Whole-body hyperthermia involves carefully raising the patient’s core body temperature to 39.5-42 degrees Celsius (approximately 103-107.6 degrees Fahrenheit) over a controlled period. This controlled fever state exploits a fundamental vulnerability of tumor cells: their impaired ability to dissipate heat compared to healthy tissue.
The therapeutic effects of hyperthermia operate through multiple pathways:
Direct cytotoxicity. Tumor cells are more susceptible to heat damage than normal cells due to their disorganized vasculature and altered membrane properties. At temperatures above 40 degrees Celsius, tumor cells experience protein denaturation, membrane disruption, and DNA repair inhibition that healthy cells better tolerate.
Heat shock protein activation. Elevated temperature induces the expression of heat shock proteins (HSPs) on the surface of tumor cells. These proteins act as “danger signals” that make tumor cells more visible to the immune system. HSP70 and HSP90, in particular, serve as chaperones that present tumor antigens to dendritic cells, promoting a specific anti-tumor immune response.
Enhanced drug delivery and chemosensitization. Hyperthermia increases blood flow and vascular permeability in tumor tissue, improving the delivery of chemotherapeutic agents. Multiple studies have demonstrated that hyperthermia can increase the effectiveness of certain chemotherapy drugs by a factor of 2-5. This is not theoretical — it has been demonstrated in randomized controlled trials for cervical cancer, soft tissue sarcoma, and bladder cancer, among others.
Immune activation. The controlled fever state triggers a broad immune response, including increased NK cell activity, enhanced dendritic cell maturation, and elevated pro-inflammatory cytokine production. This systemic immune activation can complement the localized anti-tumor effects.
Whole-body hyperthermia has been studied in clinical trials for over four decades. The European Hyperthermia Society (now the European Society for Hyperthermic Oncology) has published guidelines supporting its use as an adjunct to chemotherapy and radiation for specific tumor types.
Landmark trials include the Dutch Deep Hyperthermia Trial, which demonstrated significant survival benefit when hyperthermia was added to radiation for cervical cancer, and multiple trials in soft tissue sarcoma showing improved local control rates. A 2018 meta-analysis published in the International Journal of Hyperthermia confirmed that adding hyperthermia to standard treatment improved outcomes across multiple cancer types.
At St. George Hospital, we perform whole-body hyperthermia using infrared-A irradiation systems under continuous monitoring of core temperature, heart rate, oxygen saturation, and blood pressure. Sessions typically last 2-4 hours including the warming and cooling phases. Treatment is administered by experienced physicians and specialized nursing staff, with patients monitored throughout.
We typically integrate hyperthermia with chemotherapy cycles, timing the heat treatment to coincide with peak drug concentrations for maximum synergy. Treatment plans are individualized based on tumor type, stage, and the patient’s overall condition.
For specific tumor sites, we also offer local and regional hyperthermia using radiofrequency or microwave applicators. These devices focus heat energy directly on the tumor and surrounding tissue, achieving local temperatures of 42-45 degrees Celsius while sparing distant organs.
Local hyperthermia is particularly valuable for superficial tumors, recurrent breast cancer, head and neck cancers, and pelvic tumors. It can be combined with radiation therapy (thermoradiotherapy) or chemotherapy, and the evidence base for these combinations is well established in European oncology guidelines.
The advantage of local over whole-body hyperthermia is the ability to achieve higher temperatures at the tumor site with less systemic stress. The appropriate choice depends on the clinical situation — localized disease may benefit from targeted heat, while disseminated disease or the need for systemic immune activation may favor whole-body approaches.
Mistletoe extract (Viscum album) is the most widely studied complementary cancer therapy in Europe. It has been used in integrative oncology since the 1920s and is an approved pharmaceutical product in Germany, available by prescription under brand names including Iscador, Helixor, and abnobaVISCUM. This is not an herbal supplement sold in health food stores — it is a regulated pharmaceutical manufactured under GMP conditions.
Mistletoe extracts contain biologically active compounds including viscotoxins, mistletoe lectins (ML-I, ML-II, ML-III), and polysaccharides. The therapeutic effects are mediated primarily through:
Lectin-mediated immune activation. Mistletoe lectins bind to specific sugar residues on immune cells, triggering a cascade of immune responses. ML-I in particular stimulates natural killer (NK) cell activity, promotes dendritic cell maturation, and enhances the release of immune-activating cytokines including IL-1, IL-6, and TNF-alpha.
Induction of apoptosis. In laboratory studies, mistletoe lectins induce programmed cell death (apoptosis) in various tumor cell lines through activation of caspase pathways. This effect appears to be selective, with tumor cells being more susceptible than normal cells.
Quality of life improvement. Multiple clinical trials have documented improvements in fatigue, appetite, sleep, emotional well-being, and pain in cancer patients receiving mistletoe therapy.
The evidence base for mistletoe therapy is substantial but heterogeneous. A Cochrane review examined 49 clinical studies and found evidence for improved quality of life and possible survival benefit, while noting the need for more rigorous trials. A Phase I trial conducted at Johns Hopkins University confirmed the safety of intravenous mistletoe extract in cancer patients and documented measurable immune activation.
Observational studies from German cancer registries involving thousands of patients have consistently shown improved quality of life and, in some tumor types, improved survival in patients receiving mistletoe therapy alongside standard care. Randomized trials have shown mixed results depending on tumor type and mistletoe preparation.
I present this evidence accurately because I believe patients deserve honest information. Mistletoe therapy has a reasonable evidence base, particularly for quality of life improvement and immune modulation. The survival data is suggestive but not yet conclusive across all tumor types. We use it as part of a comprehensive integrative approach, not as a standalone treatment.
We administer mistletoe extract by subcutaneous injection, with dosing titrated based on the patient’s immune response (measured by injection site reaction and laboratory markers). Treatment typically begins with low doses and is escalated gradually. In selected cases, we also use intravenous mistletoe administration for stronger immune stimulation. Treatment is individualized based on tumor type, disease stage, and immune status.
Vitamin C at pharmacologic concentrations (achievable only through intravenous administration, not oral supplementation) behaves as a pro-oxidant rather than an antioxidant. At plasma concentrations exceeding 350 micromoles per liter, vitamin C generates hydrogen peroxide selectively in the extracellular space around tumor cells.
Normal cells possess adequate catalase to neutralize this hydrogen peroxide. Tumor cells, which typically have lower catalase activity, are selectively damaged. This mechanism has been elucidated in published research from the National Institutes of Health and confirmed in multiple laboratory studies.
A Phase II randomized controlled trial in patients with stage IV pancreatic cancer, published in Cancer Chemotherapy and Pharmacology, demonstrated that high-dose IV vitamin C combined with gemcitabine and erlotinib approximately doubled median overall survival compared to chemotherapy alone (12 months vs. 6 months). While this was a small trial and requires confirmation in larger studies, the results are notable.
Additional trials have shown that high-dose IV vitamin C reduces chemotherapy side effects — including fatigue, nausea, and neuropathy — without reducing chemotherapy effectiveness. A trial in ovarian cancer patients published in Science Translational Medicine demonstrated that IV vitamin C reduced chemotherapy toxicity while maintaining anti-tumor efficacy.
High-dose IV vitamin C is generally well tolerated, but it requires screening for glucose-6-phosphate dehydrogenase (G6PD) deficiency before initiation. Patients with G6PD deficiency can experience hemolytic anemia with high-dose vitamin C, and this screening is mandatory in our protocols. We also monitor renal function and adjust dosing accordingly.
We administer IV vitamin C in doses of 30-75 grams per infusion, 2-3 times weekly, with pharmacokinetic monitoring to ensure therapeutic plasma concentrations are achieved. Treatment is integrated with, not substituted for, conventional chemotherapy protocols.
Before initiating any immune-supportive therapy, we perform comprehensive immune profiling: lymphocyte subsets (CD3, CD4, CD8, CD19, CD16/56), NK cell activity (functional assay, not just cell count), cytokine profiles, immunoglobulin levels, and tumor markers. This allows us to understand each patient’s specific immune status and target our interventions accordingly.
Thymic peptides support the maturation and differentiation of T cells, which are critical for anti-tumor immune surveillance. In patients with documented T-cell lymphopenia or reduced NK cell function — common findings after chemotherapy — thymic peptide therapy can help restore immune competence. We use pharmaceutical-grade thymic peptide preparations administered by injection over defined treatment cycles.
Dendritic cells are the “directors” of the immune response, responsible for presenting tumor antigens to T cells and initiating targeted immune attacks. We work with specialized laboratories on dendritic cell-based approaches for selected patients, where tumor material is used to prime dendritic cells that are then administered to stimulate a specific anti-tumor immune response.
I must note that dendritic cell therapy for cancer remains an area of active research. While approved dendritic cell therapies exist for specific indications (sipuleucel-T for prostate cancer), the broader application of this approach is still investigational. We offer it to selected patients within an appropriate clinical framework, with full transparency about the evidence level.
A significant portion of our work involves helping patients manage the side effects of conventional cancer treatment. Chemotherapy-induced nausea, fatigue, neuropathy, mucositis, and immunosuppression can be meaningfully reduced through integrative approaches.
We use targeted nutritional support (specific amino acids, antioxidants timed to avoid interference with treatment mechanisms), acupuncture for nausea and pain management, IV nutrient repletion for treatment-depleted minerals and vitamins, and carefully selected botanical medicines.
Cancer and its treatment create significant nutritional demands. We provide individualized nutritional counseling, address cancer-related cachexia and sarcopenia through targeted protein and amino acid supplementation, and optimize micronutrient status. Our approach is evidence-based — we do not advocate extreme elimination diets or unproven nutritional “cures.”
A cancer diagnosis affects the whole person. Our psychosomatic medicine team, led by Dr. Christiane Godau, provides psychological support including individual counseling, mindfulness-based stress reduction, and strategies for managing the anxiety and uncertainty that accompany cancer treatment. This is not optional — it is an integral part of comprehensive cancer care.
Transparency requires me to state clearly what we do not offer:
We do not provide “miracle cures.” We do not claim to cure cancer with vitamins, supplements, or any single therapy. We do not advise patients to refuse or delay proven conventional treatments. We do not use unproven substances with no clinical evidence base. We do not make promises about outcomes that we cannot support with data.
If a therapy we offer has preliminary evidence, we say so. If it is well established, we say that too. Patients deserve accurate information to make informed decisions about their care.
Many of our patients are referred by their oncologists, or come to us while continuing treatment at their home institutions. We welcome and encourage this collaborative model. After evaluation and treatment, we provide your referring physician with detailed reports of our findings and recommendations.
For international patients, our team coordinates with oncologists worldwide, ensuring continuity of care and transparent communication about our complementary interventions. We do not operate in isolation — cancer care is a team effort, and we function as part of your broader treatment team.
If you or someone you care for is dealing with a cancer diagnosis and is interested in evidence-based integrative support alongside conventional treatment, we would be glad to discuss how our approach might complement your existing care.
Our international patient coordination team speaks English, German, Russian, and Arabic, and can arrange preliminary consultations to determine whether our programs are appropriate for your situation. Contact us at info@clinicum-stgeorg.de or +49 (0)8061 398-0.
Cancer treatment is challenging. Our goal is to help you navigate it with every evidence-based tool available — conventional and integrative — while maintaining your quality of life and supporting your body’s own capacity to fight.