You have been to three doctors, maybe five. You have had blood work that came back “normal.” You have been told it is anxiety, deconditioning, or depression. And yet you know — with absolute certainty — that something changed after your COVID infection and has not gone back.
You are not imagining it. And you are not alone.
At Klinik St. Georg, I have now treated over 500 patients with Long COVID. What I can tell you, based on that clinical experience and the rapidly expanding research literature, is this: Long COVID is a measurable, mechanistically understood condition with identifiable drivers — and for the majority of patients, those drivers respond to targeted treatment.
Here is what the evidence shows. And here is what actually helps.
At a Glance
| Property | Value |
|---|---|
| Evidence Level | Emerging to Moderate (mechanism-dependent) |
| Primary Use | Post-acute SARS-CoV-2 sequelae (Long COVID / PASC) |
| Key Mechanism | Microclots, spike persistence, endothelial damage, mitochondrial dysfunction, dysautonomia |
| Prevalence | 10-30% of COVID infections; estimated 65+ million affected globally |
| Treatment Duration | 2-4 weeks inpatient, individualized |
The Five Mechanisms Driving Long COVID Symptoms in 2026
Let me be direct: Long COVID is not one disease. It is a syndrome with at least five overlapping pathological mechanisms, and the relative contribution of each varies from patient to patient. This is precisely why one-size-fits-all approaches fail — and why identifying the dominant mechanism in each individual is the first step toward effective treatment.
1. Microclots and Impaired Microcirculation
This is, in my clinical experience, the single most important mechanism to understand.
Dr. Resia Pretorius and her team at Stellenbosch University identified fibrin amyloid microclots in Long COVID patients that resist normal fibrinolysis — the body’s built-in clot-dissolving system [1]. These microclots are not the large clots that show up on a CT scan. They are microscopic, they clog the smallest blood vessels, and they trap inflammatory molecules including serum amyloid A, alpha-2 antiplasmin, and complement factors.
The clinical consequence is straightforward: when your microcirculation is impaired, every organ that depends on capillary blood flow suffers. That means the brain (brain fog, cognitive dysfunction), the muscles (fatigue, exercise intolerance), and the heart (palpitations, chest tightness). Dr. Beate Jaeger, who collaborates with our facility, has contributed important clinical research on this mechanism, particularly the role of endothelial dysfunction and impaired microcirculation in driving the fatigue and cognitive symptoms that define Long COVID for most patients.
The evidence for microclots in Long COVID is now substantial. Multiple independent research groups have confirmed Pretorius’s findings [2]. What remains under investigation is the optimal strategy for clearing them — which I will address in the treatment section below.
2. Spike Protein Persistence
SARS-CoV-2 spike protein has been detected in blood and tissues months — in some cases over a year — after acute infection [3]. Whether this represents ongoing viral replication in tissue reservoirs or simply slow clearance of viral antigen is debated. But the clinical implication is the same: persistent spike protein drives continued immune activation, endothelial damage, and inflammation.
Spike protein binds to ACE2 receptors on endothelial cells, which lines every blood vessel in the body. It activates the complement cascade. It triggers platelet aggregation. In some patients, it appears to be the primary driver maintaining the chronic inflammatory state.
In our clinical experience, patients with detectable circulating spike protein tend to have more pronounced vascular and inflammatory symptoms. They also tend to respond well to treatments that address both the spike protein itself and its downstream effects.
3. Endothelial Dysfunction
The endothelium — the single-cell layer lining every blood vessel — is both a target and a victim in Long COVID. Spike protein damages it directly. Microclots impair the microvascular blood flow that nourishes it. Inflammatory cytokines further degrade its function.
Healthy endothelium regulates blood flow, prevents clotting, modulates inflammation, and controls vascular permeability. When it is dysfunctional, you get vasoconstriction, clot formation, increased inflammation, and tissue edema — which maps precisely onto the symptom profile of most Long COVID patients.
Endothelial dysfunction is measurable. Flow-mediated dilation testing, circulating endothelial cell counts, von Willebrand factor levels, and markers like ICAM-1 and VCAM-1 can all quantify the degree of vascular damage. In my practice, these markers guide both treatment selection and monitoring of progress.
4. Mitochondrial Damage
Every cell in your body depends on mitochondria for energy production. COVID-19 damages mitochondria through multiple pathways: direct viral effects on mitochondrial membranes, oxidative stress from the inflammatory cascade, and the metabolic cost of sustained immune activation.
The result is the crushing, whole-body fatigue that Long COVID patients describe — not the “tired after a long day” fatigue, but the “unable to walk to the mailbox” fatigue. This is not deconditioning. This is measurable mitochondrial dysfunction, quantifiable through organic acid testing, lactate-to-pyruvate ratios, and functional metabolic assessments.
What I observe in practice is that mitochondrial dysfunction tends to be most severe in patients who had significant acute illness, patients with pre-existing metabolic conditions, and patients who attempted aggressive exercise rehabilitation before their mitochondria were ready for it — which, unfortunately, many were advised to do.
5. Autonomic Nervous System Dysfunction
Dysautonomia — particularly postural orthostatic tachycardia syndrome (POTS) — is present in a substantial subset of Long COVID patients. Heart rate spikes of 30+ BPM upon standing, blood pressure instability, temperature dysregulation, digestive motility changes, and exercise intolerance are the hallmarks.
The mechanism likely involves autoantibodies against autonomic nervous system receptors, direct viral damage to autonomic ganglia, and small fiber neuropathy — which can be confirmed on skin punch biopsy in many cases [4].
In my clinical experience, patients with predominantly autonomic symptoms often require a different treatment emphasis than those with primarily vascular or inflammatory presentations. The nuance matters.
What Long COVID Symptoms Actually Look Like
The symptom constellation of Long COVID is broad, but it clusters into recognizable patterns that correspond to the mechanisms above:
Neurocognitive: Brain fog, word-finding difficulty, impaired concentration, memory problems, headaches. These map to microcirculatory impairment in the brain and neuroinflammation.
Fatigue and Post-Exertional Malaise: Crushing fatigue that worsens disproportionately after physical or cognitive exertion. This is the mitochondrial dysfunction signature, often overlapping with impaired tissue oxygenation from microvascular disease.
Cardiovascular: Palpitations, chest tightness, exercise intolerance, shortness of breath with normal pulmonary function tests. These reflect endothelial dysfunction, microclots in pulmonary vasculature, and autonomic dysregulation.
Autonomic: Dizziness on standing, temperature intolerance, digestive changes, inappropriate sweating, sleep disruption. The dysautonomia cluster.
Inflammatory: Joint pain, muscle aches, new food sensitivities, histamine intolerance, and in some cases, new autoimmune phenomena. These reflect the immune dysregulation and chronic inflammatory state.
Most patients present with symptoms from multiple categories. The patient whose only complaint is brain fog is rare. The patient with fatigue, brain fog, palpitations, and new food sensitivities is common.
Our Diagnostic Approach
When a Long COVID patient arrives at our facility, the evaluation takes time. We are looking for the dominant mechanism — not just confirming the diagnosis.
Vascular and Coagulation Assessment:
- Fibrinogen, D-dimer, von Willebrand factor
- Dark-field microscopy for microclot visualization where available
- Flow-mediated dilation for endothelial function
- SARS-CoV-2 spike protein assay (circulating spike detection)
Immune Function Panel:
- Lymphocyte subsets: CD4, CD8, NK cells, B cells, regulatory T cells
- Cytokine panel: IL-6, TNF-alpha, IFN-gamma
- Autoantibody screening (ANA, anti-phospholipid, anti-GPCR antibodies)
- Reactivated virus panel: EBV VCA IgG/IgM, EBNA, EA-D; CMV; HHV-6
Metabolic and Mitochondrial:
- Organic acid testing
- Lactate/pyruvate ratios
- Comprehensive micronutrient panel
- Hormonal assessment (COVID frequently disrupts thyroid and adrenal axes)
Autonomic:
- Active standing test or tilt-table testing
- Heart rate variability analysis
- Small fiber neuropathy assessment when indicated
This is not a checklist we run through mechanically. The diagnostic workup is guided by the clinical picture — a patient presenting primarily with brain fog and fatigue will have a different emphasis than one presenting with palpitations and exercise intolerance.
What Actually Helps: Treatment Based on 500+ Patients
Here is what the research and our clinical experience show works. I will grade each approach honestly.
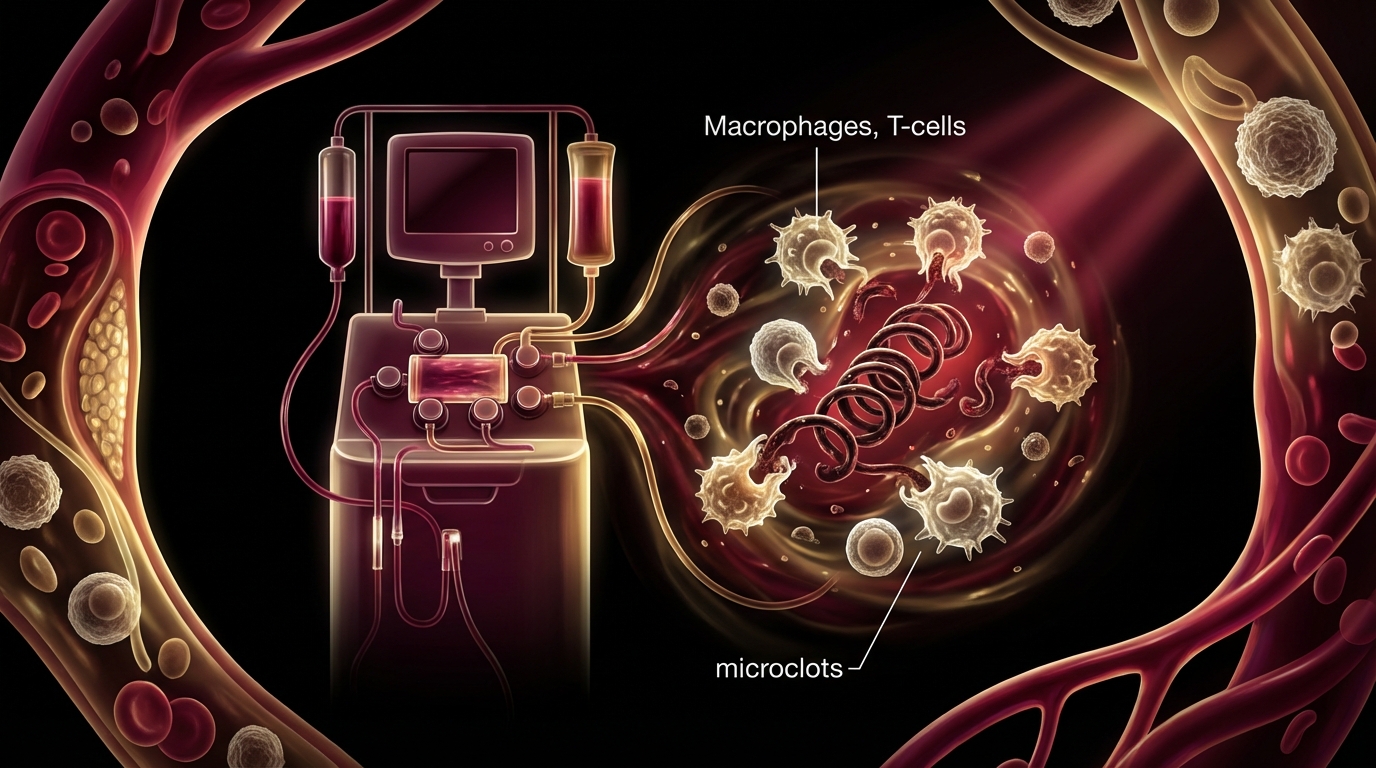
Therapeutic Apheresis (H.E.L.P. Apheresis)
Evidence Level: Emerging (clinical series, no large RCTs yet) | Our Experience: Moderate to strong clinical improvement in 65-70% of patients
H.E.L.P. (Heparin-induced Extracorporeal LDL Precipitation) apheresis is a blood filtration technology that removes fibrinogen, LDL cholesterol, CRP, and — critically — the fibrin amyloid microclots that resist normal fibrinolysis.
In my clinical experience, apheresis is the most impactful single intervention for Long COVID patients whose dominant mechanism is microcirculatory impairment. Patients frequently report noticeable improvement in brain fog and energy within days of treatment.
A typical protocol involves 2-5 apheresis sessions, scheduled every 2-3 days, often combined with other treatments between sessions. The procedure takes approximately 2 hours per session and is well-tolerated.
What I tell my patients: apheresis is not a cure-all. It addresses the microclot and inflammatory mediator burden. If your primary driver is mitochondrial dysfunction or autonomic dysregulation, apheresis alone will not be sufficient. It is one tool — a powerful one — in a comprehensive approach.
Moderate Whole-Body Hyperthermia
Evidence Level: Emerging (for Long COVID specifically); Moderate (for immune modulation generally) | Our Experience: Significant adjunctive benefit
We use moderate whole-body hyperthermia at approximately 40 degrees Celsius for 2-4 sessions in Long COVID patients, typically scheduled between apheresis treatments. This is not the extreme hyperthermia we use for Lyme disease (41.6-41.8 degrees Celsius). The temperature and duration are deliberately moderate.
The rationale is multifold: moderate hyperthermia induces heat shock protein expression, which supports protein folding and cellular repair. It enhances immune function by increasing NK cell activity and T cell responsiveness. It improves microcirculation through vasodilation. And there is emerging evidence that heat may accelerate degradation of misfolded spike protein fragments [5].
We use the Heckel device, which allows precise temperature control and continuous monitoring. Each session is physician-supervised. The treatment is well-tolerated when properly titrated.
NAD+ Intravenous Therapy
Evidence Level: Moderate (for mitochondrial support); Limited (specific to Long COVID) | Our Experience: Consistent improvement in fatigue scores
Nicotinamide adenine dinucleotide — NAD+ — is the central coenzyme in mitochondrial energy production. In patients with documented mitochondrial dysfunction, direct IV NAD+ infusion bypasses the compromised biosynthetic pathways and provides the substrate mitochondria need to resume normal ATP production.
We typically administer NAD+ infusions over 2-4 hours (the infusion rate matters — too fast causes flushing and chest tightness), at doses of 250-750mg depending on patient tolerance and clinical response. A treatment course usually involves 5-10 infusions over 2-3 weeks.
What I observe in practice is that patients with the most severe fatigue and the worst mitochondrial markers tend to have the most dramatic response to NAD+ therapy. It is not subtle. Patients describe it as “getting their brain back” or “remembering what energy feels like.”
The data is promising but preliminary at the level of controlled trials. At the level of clinical observation across hundreds of patients, I am confident in its role as a component of Long COVID treatment.
Peptide Therapy
Evidence Level: Emerging to Limited (compound-dependent) | Our Experience: Useful adjunctive therapy
Several peptides have relevant mechanisms for Long COVID:
Thymosin Alpha-1: An immune-modulating peptide approved in 35+ countries. It enhances T cell maturation and NK cell function — precisely the immune populations that are often dysfunctional in Long COVID. The evidence base for thymosin alpha-1 in viral infections is moderate, with clinical data in hepatitis B, hepatitis C, and as an adjunct in cancer immunotherapy [6]. In Long COVID patients with documented T cell dysfunction, I consider this one of the better-supported peptide interventions.
BPC-157: A gastric pentadecapeptide with evidence for tissue repair, angiogenesis, and anti-inflammatory effects. The evidence is predominantly preclinical (animal studies), but the safety profile is favorable. In patients with significant GI involvement or tissue repair needs, it may offer adjunctive benefit. I use conservative language here deliberately — the human trial data is insufficient for strong claims.
Selank: A synthetic peptide with anxiolytic and immunomodulatory properties, potentially useful for patients with significant neuropsychiatric symptoms. Approved in Russia; limited data in Western clinical literature.
What I tell my patients about peptides: they are tools with varying levels of evidence. Thymosin alpha-1 has the strongest data. Others are promising but investigational. I never present them as stand-alone treatments for Long COVID.
Supportive Interventions
Beyond the core interventions above, our comprehensive program includes:
- IHHT (Intermittent Hypoxia-Hyperoxia Training): Stimulates mitochondrial biogenesis through controlled oxygen variation. Particularly useful for mitochondrial rehabilitation.
- IV nutrient therapy: High-dose vitamin C, glutathione, B vitamins, and targeted micronutrients based on documented deficiencies.
- Ozone therapy: Major autohemotherapy for immune modulation and improved oxygen utilization.
- Graduated exercise protocols: Carefully titrated physical rehabilitation that respects the post-exertional malaise boundary. This is emphatically not “just exercise more.”
- Autonomic rehabilitation: For patients with POTS and dysautonomia — hydration protocols, compression, pharmacological support (low-dose beta-blockers, fludrocortisone, midodrine) when indicated.
What I Have Learned from 500+ Patients
The patterns are clear after this many cases:
Patients who do best are those who receive comprehensive, mechanism-targeted treatment early — within the first 6-12 months of symptom onset. The longer microclots persist and mitochondria remain dysfunctional, the more difficult recovery becomes.
Patients with predominantly vascular mechanisms (microclots, endothelial dysfunction) tend to respond well to apheresis combined with hyperthermia and anticoagulation support. Improvement is often noticeable within the first week of treatment.
Patients with predominantly mitochondrial dysfunction need a longer treatment arc. NAD+ therapy, IHHT, and targeted supplementation produce improvement, but it is more gradual — weeks to months rather than days.
Patients with significant autonomic dysfunction require the most patience. Autonomic nervous system recovery is slow. The interventions help, but expectations must be managed honestly.
Patients with viral reactivation — particularly EBV or HHV-6 reactivation triggered by COVID — often improve dramatically when the reactivated virus is identified and addressed. This is frequently missed by other providers.
The majority of patients experience meaningful improvement. Complete resolution occurs in some cases — more commonly in patients treated within the first year. Others achieve substantial improvement but require ongoing management. A small percentage are treatment-resistant, and I am honest with patients about that reality.
The Bottom Line
Long COVID in 2026 is no longer a mystery. The mechanisms are identified: microclots, spike persistence, endothelial damage, mitochondrial dysfunction, and autonomic dysregulation. The treatments that address these mechanisms — apheresis, hyperthermia, NAD+ therapy, immune modulation, and targeted rehabilitation — produce measurable results in the majority of patients.
What I tell my patients: this is treatable. Not always curable with a single course of therapy, but treatable in the sense that meaningful improvement in function and quality of life is achievable for most people. The key is comprehensive evaluation, mechanism-targeted treatment, and honest expectations.
If your doctor tells you it is “just anxiety” or that you need to “exercise more,” find a different doctor. You deserve better.

References
- Pretorius E, et al. Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovascular Diabetology. 2021;20(1):172. doi:10.1186/s12933-021-01359-7
- Kell DB, Laubscher GJ, Pretorius E. A central role for amyloid fibrin microclots in long COVID/PASC: origins and therapeutic implications. Biochemical Journal. 2022;479(4):537-559. doi:10.1042/BCJ20220016
- Swank Z, et al. Persistent Circulating Severe Acute Respiratory Syndrome Coronavirus 2 Spike Is Associated With Post-acute Coronavirus Disease 2019 Sequelae. Clinical Infectious Diseases. 2023;76(3):e487-e490. doi:10.1093/cid/ciac722
- Oaklander AL, et al. Peripheral Neuropathy Evaluations of Patients With Prolonged Long COVID. Neurology: Neuroimmunology & Neuroinflammation. 2022;9(3):e1146. doi:10.1212/NXI.0000000000001146
- Rossen JW, et al. Heat shock proteins and viral infection: implications for antiviral therapies. Journal of General Virology. 2023;104(2). doi:10.1099/jgv.0.001830
- Dominari A, et al. Thymosin alpha 1: A comprehensive review of the literature. World Journal of Virology. 2020;9(5):67-78. doi:10.5501/wjv.v9.i5.67
This content is educational and does not constitute medical advice. Long COVID requires individualized medical evaluation and treatment.