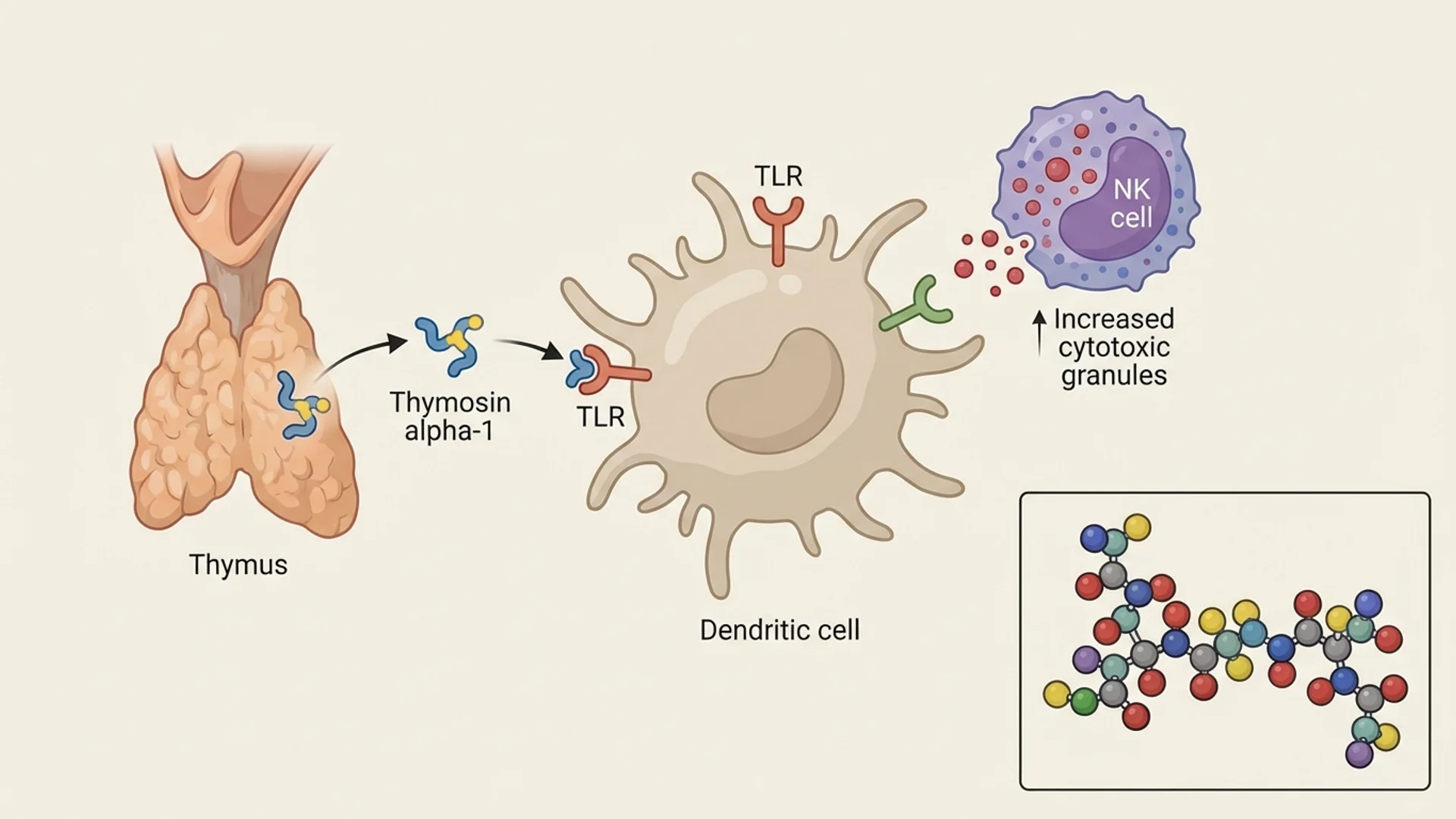
In a field where much of the evidence base consists of animal studies and clinical observation, thymosin alpha-1 stands apart. It is an approved pharmaceutical in over 35 countries. It has been the subject of numerous randomized controlled trials. It has a well-characterized safety profile built on decades of clinical use. When patients ask me which peptide has the strongest evidence behind it, thymosin alpha-1 is my answer.
At a Glance
Property Detail
Type Synthetic analog of endogenous thymic peptide
Amino acid count 28
Primary mechanism Dendritic cell maturation, T-cell differentiation, Th1/Th2 balance
Evidence level Multiple RCTs, meta-analyses, approved drug in 35+ countries
Regulatory status Approved as Zadaxin in 35+ countries (hepatitis B/C, immune adjuvant); not FDA-approved in the US
Route of administration SC injection
What Is Thymosin Alpha-1?
Thymosin alpha-1 (Ta1) is a 28-amino-acid peptide originally isolated from thymic tissue — specifically from thymosin fraction 5, a partially purified extract of calf thymus. It was first characterized by Allan Goldstein and colleagues at George Washington University in 1977.
The thymus gland is the organ primarily responsible for T-cell maturation during development. It is well established that thymic function declines with age — a process called thymic involution — and that this decline contributes to the immune senescence observed in elderly populations. Ta1 is one of several thymic peptides that play a role in T-cell education and immune regulation.
The synthetic version of Ta1, marketed under the brand name Zadaxin (developed by SciClone Pharmaceuticals, now Immunomedics), has been approved as a prescription medication in countries across Asia, South America, and parts of Europe. Its approved indications include chronic hepatitis B, chronic hepatitis C (as an adjunct to interferon), and as an immune adjuvant in immunocompromised patients.
Mechanism of Action
Primary Pathways
- Dendritic Cell Maturation: Ta1 promotes the maturation and functional activation of dendritic cells — the antigen-presenting cells that serve as the bridge between innate and adaptive immunity. Mature dendritic cells are more effective at processing and presenting antigens to T-cells, initiating targeted immune responses.
- T-Cell Differentiation: Ta1 stimulates the differentiation of immature T-cell precursors (thymocytes) into mature, functional T-cells. This includes promotion of CD4+ helper T-cell and CD8+ cytotoxic T-cell maturation. In immunocompromised patients, this effect can help restore depleted T-cell populations.
- Th1/Th2 Balance: Ta1 appears to modulate the balance between Th1 (cell-mediated) and Th2 (humoral) immune responses, favoring Th1 immunity. This is clinically relevant because many chronic infections and cancers are associated with an inappropriate shift toward Th2-dominant responses. Restoring Th1 function can enhance the immune system’s ability to clear intracellular pathogens and recognize tumor cells.
- Natural Killer Cell Activation: Ta1 enhances NK cell activity, which is relevant for both antiviral and antitumor immunity. NK cells are the innate immune system’s primary defense against virus-infected and malignant cells.
- Toll-Like Receptor Modulation: Ta1 acts through TLR-2, TLR-5, TLR-8, and TLR-9, amplifying the innate immune response to pathogen-associated molecular patterns. This positions Ta1 as a bridge between innate and adaptive immunity.
Research Assessment
What the Evidence Shows
Claimed Effect Evidence Level Key Studies Assessment
Hepatitis C adjunct therapy RCTs Moscarella 1998, Sherman 2005 Strong
Vaccine adjuvant (elderly) RCTs Gravenstein 1998, Shen 2007 Strong
Cancer immunoadjuvant Controlled studies Garaci 2000, Maio 2010 Promising
Sepsis survival RCTs Wu 2013 Promising
COVID-19 support Observational studies Liu 2020 Preliminary
chronic Lyme disease treatment immune support Clinical observation
Preliminary
The evidence for Ta1 in hepatitis B is particularly robust. A meta-analysis by You and colleagues (2006) pooling data from multiple RCTs demonstrated that Ta1 monotherapy achieved sustained virological response rates comparable to interferon-alpha, with significantly fewer side effects. In combination with interferon or nucleoside analogs, response rates were further improved.
As a vaccine adjuvant in elderly and immunocompromised patients, Ta1 has demonstrated the ability to enhance antibody responses to influenza and hepatitis B vaccines — a clinically meaningful finding given the well-documented decline in vaccine efficacy with age.
In oncology, Ta1 has been studied as an immunoadjuvant alongside chemotherapy and other treatments. Clinical studies have shown improvements in immune parameters and, in some studies, improvements in survival. The evidence is not yet at the level of large Phase III trials for most cancer types, but the mechanistic rationale is strong and the existing clinical data is encouraging.
What the Evidence Does Not Show
Ta1 is not a direct antiviral or antitumor agent. It works by modulating immune function, which means its efficacy depends on the patient’s underlying immune capacity and the specific pathology being treated. In patients with profoundly compromised immune systems (such as late-stage AIDS or post-transplant immunosuppression), the immune substrate that Ta1 acts upon may be insufficient for a meaningful response.
The evidence for Ta1 in chronic Lyme disease and post-COVID syndrome, while mechanistically rational, is based on clinical observation and case series rather than controlled trials.
Dosing
Route Dose Range Frequency Duration Source of Data
Subcutaneous 0.8-3.2 mg 2-3 times weekly Variable Clinical practice for off-label indications
Note: The 1.6 mg twice-weekly dose is the approved pharmaceutical dose. This dosing is based on human clinical trial data, which places Ta1 on firmer ground than most therapeutic peptides.
Safety and Side Effects
Known
Ta1 has an exceptionally well-documented safety profile. Across clinical trials involving thousands of patients, the adverse event rate has been remarkably low. The most commonly reported side effects are mild injection site reactions. No serious drug-related adverse events have been consistently reported. In the hepatitis trials, Ta1 was noted for its favorable side effect profile compared to interferon-alpha.
Theoretical Concerns
Because Ta1 enhances immune function, there is a theoretical concern about exacerbating autoimmune mechanismse conditions. In patients with active autoimmune disease, immune stimulation could potentially worsen symptoms. This concern is mechanistic rather than observed — Ta1’s immunomodulatory (as opposed to purely immunostimulatory) mechanism may actually mitigate this risk, but caution is warranted.
Contraindications
Active autoimmune disease (relative contraindication — requires careful risk-benefit assessment). Post-transplant immunosuppression (immune enhancement could theoretically antagonize immunosuppressive therapy). Pregnancy and lactation (insufficient data).
Drug and Supplement Interactions
Ta1 has been studied in combination with interferon-alpha, nucleoside analogs, and various chemotherapy regimens with no significant adverse interactions identified. Its combination with interferon-alpha for hepatitis treatment is well-documented and appears to be synergistic. Theoretical caution is warranted with immunosuppressive medications, where Ta1’s immune-enhancing effects could antagonize the intended immunosuppression.
Clinical Perspective
Thymosin alpha-1 is the peptide I have the most confidence in, and the reason is straightforward: it has real clinical data behind it. When I discuss Ta1 with patients, I am not extrapolating from rat studies or relying solely on clinical observation. I am drawing on decades of human clinical trial data and post-market surveillance from millions of administered doses worldwide — a distinction explored further in our physician’s overview of peptide therapy benefits, which covers the full range of domains from tissue repair to neuroprotection.
In my practice, I find Ta1 most valuable in three clinical contexts:
First, chronic infections with documented immune exhaustion. Patients with chronic Lyme disease, reactivated EBV, or chronic viral hepatitis often present with depleted or dysfunctional T-cell populations. Ta1 provides a targeted intervention to restore immune competence. I typically monitor lymphocyte subsets (CD4, CD8, NK cell counts and function) before and during treatment to assess response.
Second, post-COVID immune dysregulation. Many patients presenting with persistent post-COVID symptoms show evidence of immune dysregulation — often a combination of T-cell exhaustion and residual inflammation. Ta1’s ability to promote T-cell maturation and restore Th1/Th2 balance makes it mechanistically appropriate for this population. In these cases I frequently pair Ta1 with low-dose naltrexone (LDN), which addresses the microglial neuroinflammation component via TLR4 inhibition — a pathway Ta1 does not directly target, creating a complementary dual-mechanism approach.
Third, oncological immune support. In coordination with the oncology team at Klinik St. Georg, we use Ta1 as an immunoadjuvant in select cancer patients, particularly those undergoing chemotherapy who show evidence of treatment-related immunosuppression.
What I tell my patients is this: Ta1 is not going to cure their infection or their cancer. It is a tool for restoring and optimizing the immune system’s ability to do its job. That distinction matters.
References
- Goldstein AL, et al. “Thymosin alpha 1: isolation and sequence analysis of an immunologically active thymic polypeptide.” Proc Natl Acad Sci USA. 1977;74(2):725-729.
- Garaci E, et al. “Thymosin alpha 1 in the treatment of cancer.” Int J Immunopharmacol. 2000;22(12):1067-1076.
- You J, et al. “Thymosin alpha-1 for chronic hepatitis B: a meta-analysis.” World J Gastroenterol. 2006;12(46):7503-7506.
- Andreone P, et al. “Thymosin-alpha 1 plus interferon-alpha for naive patients with chronic hepatitis C.” J Viral Hepat. 2001;8(6):382-388.
- Gravenstein S, et al. “Effect of thymosin alpha 1 on the immune response to influenza vaccine in elderly males.” J Am Geriatr Soc. 1998;46(7):900-903.
- Wu J, et al. “Thymosin alpha 1 treatment in sepsis.” Crit Care. 2013;17(1):R8.
- Liu Y, et al. “Thymosin alpha 1 reduces mortality of severe COVID-19.” J Int Med Res. 2020;48(11):300060520966539.
- Maio M, et al. “Thymosin alpha 1 in melanoma: from bench to bedside.” Ann N Y Acad Sci. 2010;1194:138-143.
Disclaimer: This peptide profile is intended for educational purposes. Thymosin alpha-1 (Zadaxin) is approved in 35+ countries but is not FDA-approved in the United States. Consult a qualified physician before pursuing any peptide therapy.