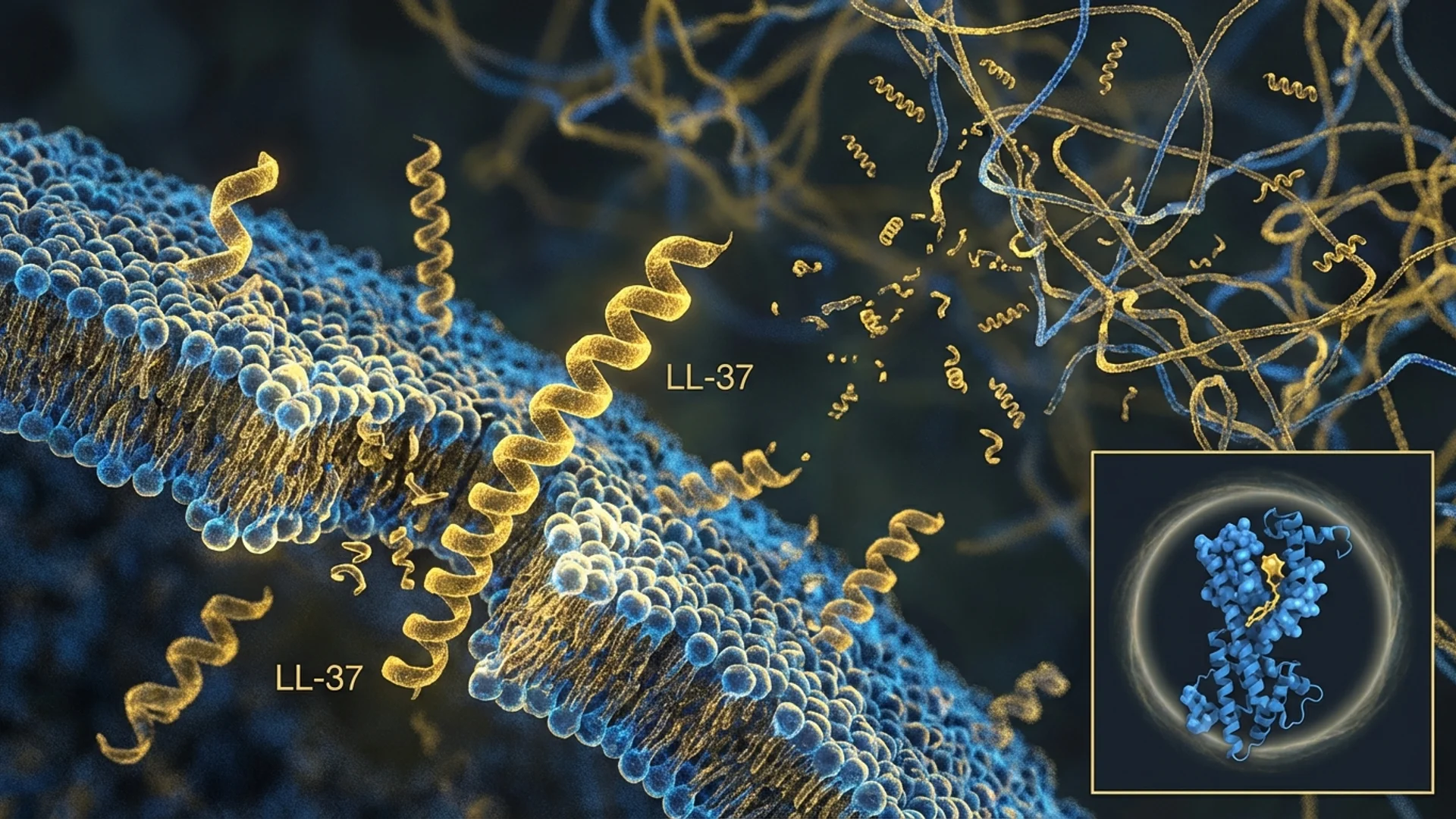
LL-37 is unlike most peptides discussed in the therapeutic peptide space. It is not a signaling molecule that modulates receptors. It is a weapon — the human body’s own antimicrobial peptide, part of the innate immune system’s first line of defense against pathogens. Its potential role in addressing biofilm-associated chronic infections, including chronic Lyme disease, makes it of particular interest in my practice.
At a Glance
Property Detail
Type Endogenous antimicrobial peptide
Amino acid count 37
Primary mechanism Direct antimicrobial activity, biofilm disruption, immune modulation
Evidence level Extensive in vitro and animal data; limited therapeutic human studies
Regulatory status Not approved as therapeutic; research and clinical observation
Route of administration SC injection, IV (in clinical settings)
What Is LL-37?
LL-37 is the only cathelicidin antimicrobial peptide identified in humans. It is derived from the larger precursor protein hCAP18 (human cationic antimicrobial protein 18 kDa), which is cleaved by proteinase 3 to release the active 37-amino-acid LL-37 peptide. The “LL” designation refers to the two leucine residues at the N-terminus.
LL-37 is produced by neutrophils, macrophages, epithelial cells, and other cell types in response to infection and tissue injury. It is found in skin, the respiratory tract, the gastrointestinal tract, and in body fluids including sweat, saliva, and breast milk. Its expression is upregulated by vitamin D — which is why vitamin D status and innate immune defense are linked.
The biological role of LL-37 extends well beyond simple antimicrobial activity. It functions as a multifaceted immune effector molecule with roles in inflammation modulation, wound healing, and chemotaxis of immune cells to sites of infection.
Mechanism of Action
Primary Pathways
- Direct Antimicrobial Activity: LL-37 kills bacteria, fungi, and enveloped viruses through direct membrane disruption. As an amphipathic peptide (containing both hydrophobic and hydrophilic regions), LL-37 interacts with the negatively charged membranes of microorganisms, inserting into the lipid bilayer and forming pores or otherwise disrupting membrane integrity. This mechanism is rapid and difficult for pathogens to develop resistance against, because it targets the fundamental physical structure of the microbial membrane rather than a specific metabolic pathway.
- Biofilm Disruption: LL-37 has demonstrated the ability to prevent biofilm formation and to disrupt established biofilms. This is clinically significant because biofilm-associated infections are notoriously resistant to conventional antibiotics. Biofilms are structured communities of bacteria encased in a self-produced matrix of extracellular polymeric substances, and bacteria within biofilms can be 100-1,000 times more resistant to antibiotics than their planktonic (free-floating) counterparts. LL-37 appears to interfere with biofilm formation by reducing initial bacterial attachment and by destabilizing the biofilm matrix.
- Immune Cell Chemotaxis: LL-37 acts as a chemoattractant for neutrophils, monocytes, and T-cells, recruiting immune cells to sites of infection. This bridges the innate and adaptive immune responses by ensuring that the initial antimicrobial activity is followed by a coordinated immune cell response.
- Inflammation Modulation: LL-37 has both pro-inflammatory and anti-inflammatory effects, depending on the context. It can stimulate pro-inflammatory cytokine production in the presence of active infection (amplifying the immune response) while also modulating excessive inflammation through effects on NF-kB signaling and by promoting the clearance of cellular debris and apoptotic cells.
- Wound Healing: LL-37 promotes wound healing through stimulation of cell migration, proliferation, and angiogenesis at wound sites. It also enhances the production of growth factors and matrix metalloproteinases involved in tissue remodeling.
Research Assessment
What the Evidence Shows
Claimed Effect Evidence Level Key Studies Assessment
Biofilm disruption In vitro, animal models Overhage 2008, Dean 2011 Promising
Anti-Borrelia activity In vitro Combs 2020 Preliminary
Wound healing promotion Animal studies, limited human Heilborn 2003 Promising
Immune cell recruitment In vitro, animal studies Yang 2000 Strong mechanistic data
Antiviral activity In vitro Barlow 2011 Preliminary
The antimicrobial activity of LL-37 has been extensively characterized in vitro against a broad range of pathogens, including Gram-positive and Gram-negative bacteria, fungi (including Candida species), and enveloped viruses. The breadth of antimicrobial activity is consistent with its role as a first-line innate immune defense.
The biofilm data is particularly relevant for clinical practice. Overhage and colleagues (2008) demonstrated that LL-37 could inhibit Pseudomonas aeruginosa biofilm formation at sub-antimicrobial concentrations and could reduce established biofilm biomass. Dean and colleagues (2011) extended these findings to other bacterial species.
Of specific interest for chronic Lyme disease is the in vitro demonstration that LL-37 has activity against Borrelia burgdorferi, including against the stationary-phase persister forms that are hypothesized to contribute to persistent infection.
What the Evidence Does Not Show
The therapeutic use of exogenous LL-37 in humans has not been validated in clinical trials. The in vitro antimicrobial and anti-biofilm data is compelling, but the translation to systemic therapeutic use faces significant challenges, including peptide stability in vivo, potential for host cell toxicity at high concentrations, and the question of whether systemically administered LL-37 can achieve effective concentrations at sites of infection.
The anti-Borrelia data is in vitro only. Whether systemically administered LL-37 can penetrate the tissues where Borrelia persists (joints, connective tissue, nervous system) at concentrations sufficient for antimicrobial or anti-biofilm effects has not been demonstrated.
Dosing
Route Dose Range Frequency Duration Source of Data
IV (clinical setting) Variable Variable Variable Clinical practice
Note: Dosing is based entirely on clinical practice. There are no human dose-finding studies for therapeutic LL-37 administration. The doses used in clinical practice are substantially lower than the concentrations shown to be antimicrobial in vitro, which raises questions about whether the therapeutic mechanism in clinical use is direct antimicrobial activity or immune modulation.
Safety and Side Effects
Known
Clinical observation has not revealed serious adverse events. The most commonly reported side effects are injection site reactions, which can be more pronounced than with other peptides due to LL-37’s pro-inflammatory properties. Transient flu-like symptoms have been reported, possibly reflecting immune activation.
Theoretical Concerns
At high concentrations, LL-37 can be cytotoxic to host cells, not just microorganisms. The therapeutic window — the range between immune-modulating doses and cytotoxic doses — requires characterization in humans. LL-37 has been implicated in the pathogenesis of certain autoimmune conditions (including psoriasis and rosacea, where excessive LL-37 contributes to inflammation), raising questions about whether exogenous administration could exacerbate these conditions.
Contraindications
Psoriasis (LL-37 is implicated in pathogenesis). Rosacea (same concern). Active autoimmune conditions with inflammatory skin involvement. Pregnancy and lactation (no data).
Drug and Supplement Interactions
Vitamin D supplementation increases endogenous LL-37 production, which could be additive with exogenous LL-37 administration. Immunosuppressive medications could theoretically blunt the immune-modulatory effects of LL-37. No formal interaction studies exist.
Clinical Perspective
LL-37 is a peptide that I find genuinely interesting in the context of chronic Lyme disease, which is a significant focus of our practice at Klinik St. Georg. The rationale is straightforward: Borrelia burgdorferi is hypothesized to form biofilms as a persistence mechanism, conventional antibiotics have limited efficacy against biofilm-associated bacteria, and LL-37 has demonstrated anti-biofilm activity in vitro.
I want to be transparent about the evidence gaps. We are taking in vitro observations about biofilm disruption and applying them in a clinical context where we cannot confirm that LL-37 reaches the relevant tissue sites at effective concentrations. This is an acknowledged limitation.
In my clinical experience, I use LL-37 as part of comprehensive Lyme disease treatment protocols — alongside antibiotics, immune modulation (thymosin alpha-1), and other supportive measures. I do not use it as a standalone antimicrobial. What I have observed is that patients who receive LL-37 as part of these protocols sometimes report improvements in symptoms that had been refractory to antibiotics alone. Whether this reflects direct anti-biofilm activity, immune modulation, or something else entirely, I cannot say with certainty.
The connection between vitamin D and LL-37 production is clinically actionable. Many chronic Lyme patients are vitamin D deficient, and optimizing vitamin D status is a simple intervention that supports endogenous LL-37 production. I consider this a foundational step before discussing exogenous LL-37 supplementation.
What I tell patients is that LL-37 has a strong mechanistic rationale for chronic infection support, particularly in the context of biofilm-associated infections. But we are working with in vitro data and clinical observation, not controlled trial results. For patients who have exhausted conventional options and understand the limitations of the current evidence, LL-37 represents a reasonable addition to a comprehensive treatment protocol.
References
- Durr UH, et al. “LL-37, the only human member of the cathelicidin family of antimicrobial peptides.” Biochim Biophys Acta. 2006;1758(9):1408-1425.
- Vandamme D, et al. “A comprehensive summary of LL-37, the factotum human cathelicidin peptide.” Cell Immunol. 2012;280(1):22-35.
- Overhage J, et al. “Human host defense peptide LL-37 prevents bacterial biofilm formation.” Infect Immun. 2008;76(9):4176-4182.
- Dean SN, et al. “Natural and synthetic cathelicidin peptides with anti-microbial and anti-biofilm activity against Staphylococcus aureus.” BMC Microbiol. 2011;11:114.
- Combs JA, et al. “Cathelicidin LL-37 has direct antimicrobial activity against Borrelia.” FASEB J. 2020;34(S1):1.
- Yang D, et al. “LL-37, the neutrophil granule-and epithelial cell-derived cathelicidin, utilizes formyl peptide receptor-like 1 (FPRL1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and T cells.” J Exp Med. 2000;192(7):1069-1074.
- Heilborn JD, et al. “The cathelicidin anti-microbial peptide LL-37 is involved in re-epithelialization of human skin wounds and is lacking in chronic ulcer epithelium.” J Invest Dermatol. 2003;120(3):379-389.
- Barlow PG, et al. “Antiviral activity and increased host defense against influenza infection elicited by the human cathelicidin LL-37.” PLoS One. 2011;6(10):e25333.
Disclaimer: This peptide profile is intended for educational purposes. LL-37 is not approved for therapeutic use by any regulatory agency. Consult a qualified physician before pursuing any peptide therapy.