At a Glance
| Property | Detail |
|---|---|
| Evidence Level | Moderate preclinical; limited human (clinical observation) |
| Structure | Tripeptide: Lys-Pro-Val (from alpha-MSH C-terminus) |
| Primary Mechanism | NF-kB inhibition, anti-inflammatory cytokine modulation |
| Unique Property | PepT1-mediated uptake concentrates peptide in inflamed epithelium |
| Applications | IBD (Crohn’s, ulcerative colitis), intestinal inflammation, mucosal healing |
| Route | Oral (preferred for GI targets), subcutaneous |
| Regulatory Status | Not FDA-approved; investigational |
What Is KPV and Why Does It Matter for Gut Inflammation?
If you are dealing with inflammatory bowel disease, colitis, or chronic gut inflammation, your body is caught in a cycle that conventional treatments often struggle to break. The inflammatory cascade perpetuates itself — damaged tissue triggers more inflammation, which causes more damage, which triggers more inflammation. Breaking this cycle requires an intervention that reaches the inflamed tissue and shuts down the inflammatory signaling.
KPV does something that most anti-inflammatory compounds cannot: it is preferentially absorbed by inflamed intestinal cells. This is not a drug design feature — it is a consequence of the biology. Inflamed intestinal epithelium upregulates a transporter called PepT1, and KPV is a substrate for this transporter. The sicker the tissue, the more it absorbs the peptide. This is an elegant biological mechanism that concentrates the anti-inflammatory effect precisely where it is most needed.
Let me walk you through what the research shows, where the evidence is strong, and where we are still waiting for human confirmation.
Mechanism: How KPV Reduces Gut Inflammation
Origin: Alpha-MSH
KPV (Lys-Pro-Val) is the C-terminal tripeptide fragment of alpha-melanocyte-stimulating hormone (alpha-MSH). Alpha-MSH is a 13-amino-acid neuropeptide with well-established anti-inflammatory properties. It was discovered that the anti-inflammatory activity of alpha-MSH is concentrated in its three C-terminal amino acids — KPV. This means KPV retains the anti-inflammatory potency of the parent hormone in a smaller, more practical package.
NF-kB Inhibition
NF-kB (nuclear factor kappa-light-chain-enhancer of activated B cells) is the master transcription factor for inflammatory gene expression. When activated, NF-kB translocates to the nucleus and drives the production of inflammatory cytokines (TNF-alpha, IL-1beta, IL-6, IL-8), adhesion molecules, and other mediators that perpetuate the inflammatory response.
KPV inhibits NF-kB activation by preventing the translocation of the p65 subunit to the nucleus. This is a upstream intervention — rather than blocking a single cytokine, KPV shuts down the transcriptional machinery that produces multiple inflammatory mediators simultaneously. This makes it mechanistically broader than biologics that target a single cytokine (like anti-TNF agents), though the clinical implications of this broader targeting are still being characterized.
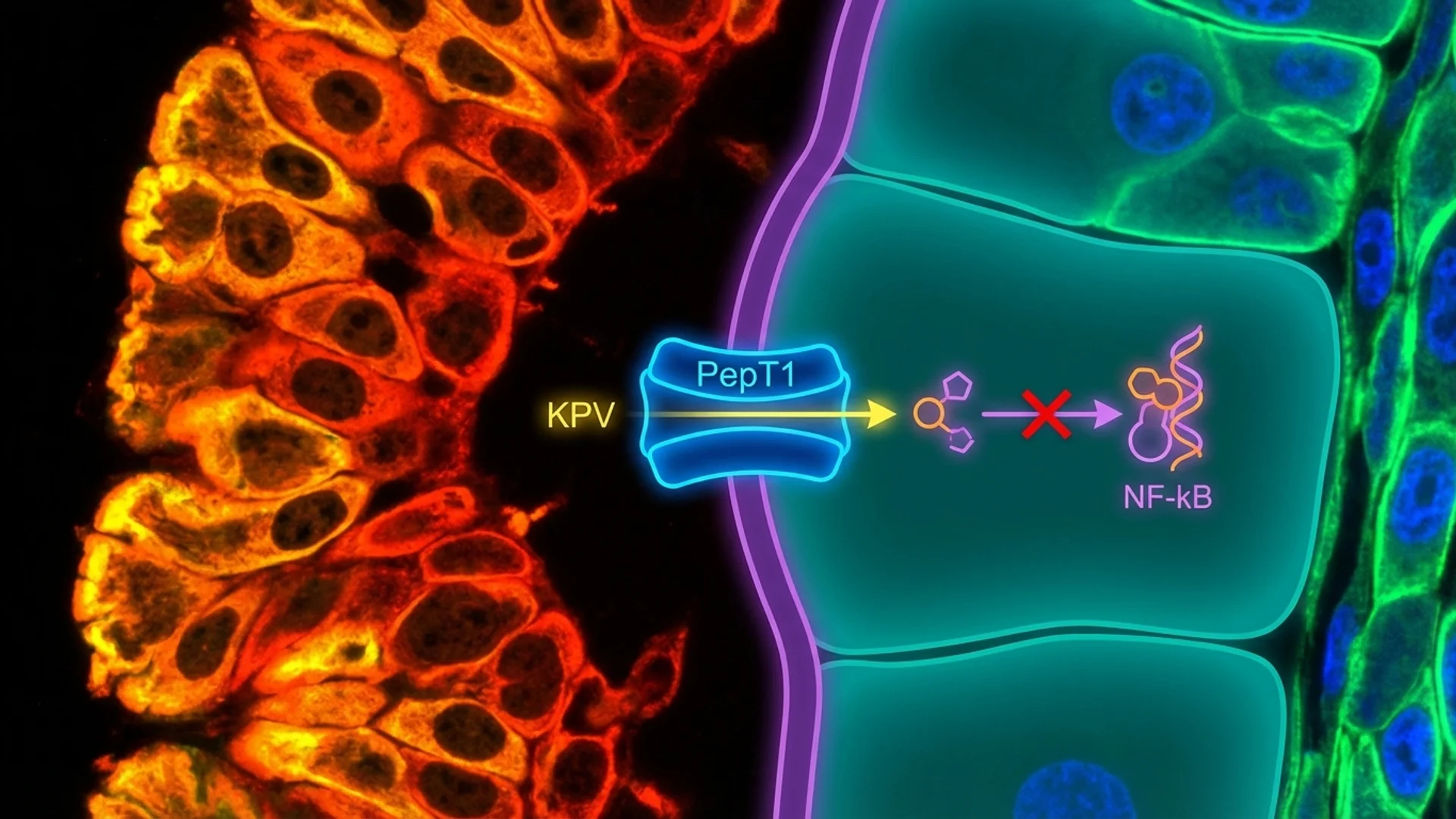
The PepT1 Transporter: Targeted Delivery to Inflamed Tissue
This is KPV’s most distinctive feature for gut applications. PepT1 (peptide transporter 1) is a proton-coupled oligopeptide transporter expressed on intestinal epithelial cells. Under normal conditions, PepT1 is expressed at moderate levels and functions primarily in dietary peptide absorption.
In inflamed intestinal tissue — as seen in IBD, colitis, and other inflammatory conditions — PepT1 expression is significantly upregulated. Dalmasso et al. (2008) demonstrated that this upregulation creates a mechanism for preferential absorption of KPV into inflamed epithelial cells. The more inflamed the tissue, the more PepT1 is expressed, and the more KPV is absorbed.
Once inside the epithelial cell, KPV inhibits NF-kB from within. The result is intracellular anti-inflammatory activity delivered specifically to the cells that need it most.
Anti-Inflammatory Cytokine Modulation
Beyond NF-kB inhibition, KPV modulates the production of specific inflammatory mediators:
| Mediator | Effect of KPV | Significance |
|---|---|---|
| TNF-alpha | Reduced production | Key driver of IBD inflammation |
| IL-1beta | Reduced production | Amplifies inflammatory cascade |
| IL-6 | Reduced production | Systemic inflammation marker |
| IL-8 | Reduced production | Neutrophil recruitment chemokine |
| IL-10 | May increase | Anti-inflammatory cytokine |
| IFN-gamma | Reduced production | Th1-mediated inflammation |
What the Evidence Shows
Animal Studies
The preclinical evidence for KPV in gut inflammation is robust and consistent:
Dalmasso et al. (2008): Demonstrated that KPV is transported into colonocytes via PepT1, with uptake significantly increased in inflamed tissue. Oral KPV significantly reduced colitis severity in a murine model, with improvements in histological scores, colon weight, and inflammatory markers.
Laroui et al. (2010): Showed that KPV loaded into nanoparticles and delivered orally reduced colitis in mice more effectively than free KPV, suggesting that formulation can further enhance efficacy. The nanoparticle formulation protected KPV from degradation and increased its delivery to the colon.
Brzoska et al. (2008): Reviewed the anti-inflammatory properties of alpha-MSH and its fragments, confirming that the KPV tripeptide retains the anti-inflammatory activity of the full-length peptide in multiple inflammatory models, including intestinal inflammation.
Evidence Summary Table
| Study | Model | Route | Finding |
|---|---|---|---|
| Dalmasso et al., 2008 | Murine colitis (DSS) | Oral | Significant reduction in colitis severity via PepT1-mediated uptake |
| Laroui et al., 2010 | Murine colitis (DSS) | Oral (nanoparticle) | Enhanced efficacy with nanoparticle delivery |
| Brzoska et al., 2008 | Multiple inflammatory models | Various | Confirmed KPV anti-inflammatory activity across models |
| Kannengiesser et al., 2008 | Murine colitis | IP injection | Alpha-MSH fragment reduced intestinal inflammation |
Human Evidence
Let me be direct: there are no published randomized controlled trials of KPV for inflammatory bowel disease or any other human condition. The human evidence consists of clinical observation from practitioners who use KPV in patients with gut inflammation.
What practitioners report — and what I observe in my own clinical experience — is consistent with the preclinical data: patients with active gut inflammation who receive oral KPV frequently report reduction in symptoms (abdominal pain, bloating, diarrhea frequency) and, in cases where follow-up endoscopy is performed, improvement in mucosal appearance. These observations are encouraging but do not constitute proof of efficacy.
Clinical Applications
Inflammatory Bowel Disease (Crohn’s Disease and Ulcerative Colitis)
KPV is most commonly used as an adjunct in IBD, not a replacement for established therapies. The rationale:
- Standard IBD therapies (5-ASA, corticosteroids, immunomodulators, biologics) have significant side effect profiles
- Many patients have partial responses or lose response over time
- KPV addresses inflammation through a distinct mechanism (NF-kB inhibition via intracellular delivery) that may complement existing therapies
- The favorable side effect profile makes it a low-risk addition
Intestinal Permeability (“Leaky Gut”)
Increased intestinal permeability is a feature of many inflammatory and autoimmune conditions. The tight junctions between intestinal epithelial cells are disrupted by chronic inflammation, allowing bacterial products and food antigens to cross the intestinal barrier and trigger systemic immune activation.
KPV, by reducing intestinal inflammation, may support restoration of tight junction integrity. This is a secondary effect — KPV does not directly repair tight junctions, but by reducing the inflammatory signaling that disrupts them, it may create the conditions for barrier restoration.
Post-Infectious Gut Inflammation
Following gastrointestinal infections (viral, bacterial, or parasitic), some patients develop persistent gut inflammation that does not resolve with antimicrobial treatment. KPV’s anti-inflammatory mechanism may be useful in this context, particularly when the underlying infection has been cleared but the inflammatory cascade continues.
Dosing Protocols
Oral KPV for Gut Inflammation
| Parameter | Detail |
|---|---|
| Dose | 250-500 mcg |
| Frequency | Once or twice daily |
| Timing | On an empty stomach, 30 minutes before food |
| Duration | 4-8 weeks for initial course |
| Reassessment | Clinical symptoms and, if applicable, inflammatory markers at 4-8 weeks |
Why Oral Is Preferred for Gut Targets
Oral administration delivers KPV directly to the intestinal epithelium — the target tissue. The PepT1 uptake mechanism then concentrates it in inflamed cells. Subcutaneous injection delivers KPV systemically, which is appropriate for systemic anti-inflammatory goals but less efficient for gut-specific targets.
The oral route also means KPV encounters the inflamed mucosa before systemic absorption, maximizing local tissue exposure.
Subcutaneous KPV for Systemic Inflammation
| Parameter | Detail |
|---|---|
| Dose | 200-500 mcg |
| Frequency | Once daily |
| Route | Subcutaneous |
| Duration | 4-8 weeks |
Subcutaneous KPV is used when the goal is systemic anti-inflammatory effect rather than gut-specific targeting. This may be appropriate for patients with skin inflammation, joint inflammation, or other non-GI inflammatory conditions.
KPV vs. BPC-157 for Gut Healing: Different Tools for Different Problems
Patients frequently ask how KPV compares to BPC-157 for gut conditions. The answer is that they address different aspects of gut pathology:
| Property | KPV | BPC-157 |
|---|---|---|
| Primary mechanism | Anti-inflammatory (NF-kB inhibition) | Tissue repair (angiogenesis, growth factors) |
| Best for | Active inflammation, flares, cytokine-driven pathology | Structural damage, ulcers, mucosal defects |
| PepT1 uptake | Yes — preferentially targets inflamed cells | No |
| Timing | During active inflammation | During and after inflammation for tissue repair |
In practice, some clinicians use both: KPV to calm the inflammatory storm, and BPC-157 to support structural repair once the acute inflammation is controlled. The mechanistic rationale for this combination is sound, though it has not been formally studied.
Safety Considerations
KPV has a generally favorable safety profile based on preclinical data and clinical observation:
Reported Side Effects
| Side Effect | Frequency | Severity |
|---|---|---|
| Mild GI discomfort | Uncommon | Mild, transient |
| Nausea (oral route) | Uncommon | Mild |
| Injection site reaction (SC) | Common | Mild |
Theoretical Concerns
Melanocyte stimulation. As a fragment of alpha-MSH, there is a theoretical concern that KPV could stimulate melanocyte activity and alter pigmentation. In practice, the KPV tripeptide has significantly less melanotropic activity than full-length alpha-MSH, and pigmentation changes have not been reported at therapeutic doses.
Immune modulation. NF-kB inhibition suppresses part of the immune response. Chronic or excessive NF-kB suppression could theoretically impair host defense. This concern is mitigated by the relatively low doses used, the short cycle durations, and the absence of reported immune compromise in clinical use.
Contraindications
- Active systemic infection (NF-kB inhibition could impair antimicrobial defense)
- Pregnancy and lactation (no safety data)
- Known hypersensitivity to alpha-MSH or its fragments
- Children (no pediatric data)
What I Tell My Patients
When a patient comes to me with inflammatory bowel disease or chronic gut inflammation, KPV is one tool in the toolbox — not the only tool. I explain the mechanism clearly: it works by shutting down the inflammatory master switch in the cells that are most inflamed, and it gets there through a biological delivery system that targets the sickest tissue.
I set expectations honestly. The animal data is compelling. The clinical observations are encouraging. But we do not have the kind of large human trials that would allow me to promise specific outcomes. What I can say is that the risk profile is favorable and the mechanistic rationale is among the most elegant in the peptide space.
The patients who do best are those who use KPV as part of a comprehensive approach — dietary modification, stress management, microbiome support, and addressing underlying triggers — rather than expecting a single peptide to solve a complex, multifactorial condition.
The Bottom Line
KPV is a tripeptide with a compelling mechanism for gut inflammation: it inhibits NF-kB (the master inflammatory switch) and is preferentially absorbed by inflamed intestinal cells via the PepT1 transporter. Animal studies consistently show significant reduction in colitis severity. Human evidence is limited to clinical observation. Oral dosing at 250-500 mcg on an empty stomach is the preferred route for gut targets. KPV is best used as an adjunct to comprehensive gut management, not as a standalone therapy.
For the full KPV overview, see KPV: Anti-Inflammatory Tripeptide. For the full peptide pillar, see All Peptide Articles.
References
- Dalmasso G, et al. “PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation.” Gastroenterology. 2008;134(1):166-178. PMID: 18061177.
- Laroui H, et al. “Functional TNFalpha gene silencing mediated by polyethyleneimine/TNFalpha siRNA nanocomplexes in inflamed colon.” Biomaterials. 2011;32(4):1218-1228. PMID: 20950853.
- Brzoska T, et al. “Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases.” Endocr Rev. 2008;29(5):581-602. PMID: 18612058.
- Kannengiesser K, et al. “Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease.” Inflamm Bowel Dis. 2008;14(3):324-331. PMID: 18092346.
- Getting SJ, et al. “Molecular determinants of the anti-inflammatory function of the neuropeptide alpha-MSH.” Ann N Y Acad Sci. 2003;994:133-140. PMID: 12851308.
Disclaimer: This article is for educational purposes and reflects current published research and clinical observation. It is not medical advice. KPV is not FDA-approved for any therapeutic indication. Inflammatory bowel disease requires medical management — do not discontinue prescribed medications without consulting your gastroenterologist. Consult a qualified physician before pursuing any peptide therapy.