At a Glance
| Property | Value |
|---|---|
| Name | Peptide Therapy for Gut Healing |
| Class | Antimicrobial peptide |
| Route | Subcutaneous injection, Oral, IV |
| Typical Dose | 200-500 mcg |
| Evidence Level | Strong |
| Key Uses | A phased gut healing protocol using BPC-157 (gastric-origin tissue repair), KPV (NF-kB inhibition with preferential uptake in inflamed… |
Key Takeaways:
- The gut is one of the best-supported clinical applications for peptide therapy, with strong preclinical data for multiple peptides
- BPC-157 peptide therapy originated from gastric juice research and has extensive animal data for gastrointestinal protection and healing
- KPV targets intestinal inflammation through NF-kB inhibition with preferential absorption in inflamed tissue
- Peptides work best when the underlying causes of gut dysfunction are addressed simultaneously
- This protocol is a clinical framework based on preclinical data and clinical observation, not RCT-validated guidelines
Gastrointestinal dysfunction is one of the most common presenting complaints in my practice. Whether it manifests as inflammatory bowel disease, post-infectious irritable bowel syndrome, antibiotic-associated gut damage, or the vague but real syndrome of increased intestinal permeability (“leaky gut”), compromised gut function undermines systemic health in ways that extend far beyond digestive symptoms.
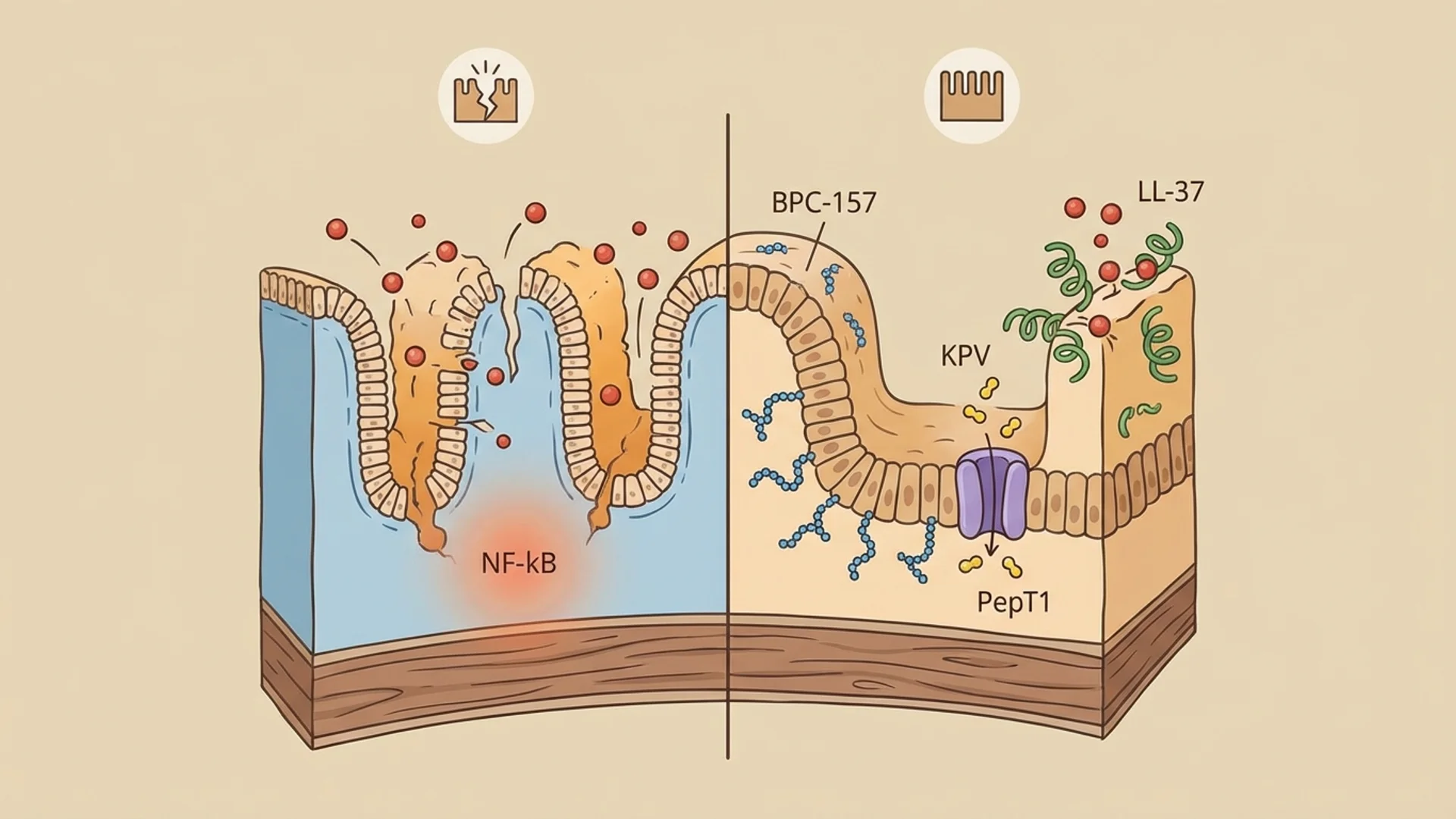
The gut is also, arguably, the organ system for which peptide therapy has the strongest mechanistic rationale. BPC-157 was derived from gastric juice. KPV is preferentially transported into inflamed intestinal tissue. LL-37 is part of the mucosal immune defense. These are not molecules being repurposed for gut applications — they have direct biological connections to gastrointestinal function.
This protocol describes my clinical approach to using these peptides for gut healing. I want to be clear that while the mechanistic rationale is strong and the preclinical data is compelling, we are working primarily with animal study data and clinical observation. Controlled human trials for these specific applications and combinations have not been published.
Who This Protocol Is For
- Patients with documented intestinal inflammation (IBD, microscopic colitis)
- Patients with increased intestinal permeability confirmed by testing (lactulose/mannitol, zonulin)
- Patients with post-infectious irritable bowel syndrome
- Patients with NSAID-induced gastropathy
- Patients recovering from gastrointestinal surgery
- Patients with antibiotic-associated gut damage
Who Should NOT Follow This Protocol Without Medical Supervision
- Patients with undiagnosed gastrointestinal symptoms (rule out serious pathology first)
- Patients with active gastrointestinal malignancy
- Patients with active gastrointestinal infection (address infection before immunomodulation)
- Pregnant or lactating women
- Patients on immunosuppressive therapy (consult prescribing physician)
Phase 1: Remove and Repair Foundations (Weeks 1-2)
Goal
Identify and remove gut-damaging factors. Establish foundational nutritional support for gut healing.
Actions
Diagnostic Assessment:
- Comprehensive stool analysis (microbiome composition, inflammatory markers, digestive function)
- Intestinal permeability testing (lactulose/mannitol ratio or zonulin levels)
- Food sensitivity testing (IgG panel — acknowledging the limitations of these tests)
- Celiac screening if not previously done
- Inflammatory markers (fecal calprotectin, CRP)
Remove Gut-Damaging Factors:
- Discontinue NSAIDs if possible (consult prescribing physician)
- Eliminate identified food triggers
- Reduce or eliminate alcohol
- Assess and address chronic medication effects on gut mucosa
Foundational Supplements:
Supplement Dose Timing Evidence Level Purpose
Zinc carnosine 75 mg twice daily With meals RCTs Mucosal protection, ulcer healing
Vitamin A 10,000 IU/day With meals (fat) Controlled studies Mucosal immune function, epithelial integrity
evidence-based probiotic therapys (multi-strain, 50+ billion CFU) Per label With meals Meta-analyses Microbiome support
Bone broth or collagen peptides 10-20 g/day Any time Traditional/clinical observation Glycine, proline for gut lining repair
Dietary:
- Elimination diet or low-FODMAP diet (depending on presentation)
- Anti-inflammatory whole foods emphasis
- Adequate protein intake (1.2-1.5 g/kg/day)
- Bone broth daily
What to Expect
Some patients notice improvement in digestive symptoms within the first 1-2 weeks from dietary changes and foundational supplements alone. Others may experience temporary worsening as the microbiome adjusts to dietary changes. This phase establishes the substrate for peptide therapy.
Phase 2: Peptide Therapy — Anti-Inflammatory (Weeks 3-10)
Goal
Reduce intestinal inflammation and begin mucosal repair using targeted peptide therapy.
Actions
Peptide Protocol:
Peptide Dose Route Frequency Duration Evidence Level Purpose
KPV 200-500 mcg Oral (capsule) Once daily 8 weeks Animal studies (colitis models) NF-kB inhibition, targeted anti-inflammatory in inflamed mucosa
Notes on Administration:
- BPC-157 oral administration is supported by its stability in gastric juice. For GI applications, oral is the preferred route because it delivers the peptide directly to the intestinal mucosa.
- KPV oral administration exploits the PepT1 transporter, which is upregulated in inflamed intestinal tissue. This provides preferential delivery to the areas that need it most.
- For patients with primarily lower GI involvement (distal colitis, rectal inflammation), compounded KPV suppositories (500 mcg-1 mg) may provide more direct delivery.
Continue:
- All foundational supplements from Phase 1
- Dietary modifications
Monitoring (Week 6):
- Clinical symptom assessment (stool frequency, consistency, pain, bloating)
- Fecal calprotectin (if elevated at baseline)
- Adjust dosing based on response
What to Expect
In my clinical experience, patients who respond to this protocol typically begin noticing improvement in digestive symptoms between weeks 2 and 4 of peptide therapy. Reduced abdominal pain, improved stool consistency, and decreased bloating are the most commonly reported improvements. Some patients describe a progressive “calming” of the gut — less reactivity to foods that previously caused symptoms.
Phase 3: Mucosal Immune Support (Weeks 6-10, overlapping with Phase 2)
Goal
For patients with evidence of mucosal immune dysfunction or recurrent infections, add LL-37 for antimicrobial and immune support.
Actions
Add to existing peptide protocol (if indicated):
Peptide Dose Route Frequency Duration Evidence Level Purpose
Indications for adding LL-37:
- Documented or suspected SIBO (small intestinal bacterial overgrowth)
- Recurrent gastrointestinal infections
- Post-infectious IBS with evidence of persistent mucosal immune activation
- chronic Lyme disease treatment disease with gastrointestinal involvement
Not all patients need LL-37. For straightforward gut barrier dysfunction without infectious or immune components, BPC-157 and KPV are sufficient.
What to Expect
Patients receiving LL-37 may experience mild flu-like symptoms in the first few days as innate immune activation occurs. This is typically self-limiting. Improvements in symptoms related to SIBO or recurrent infections may become apparent within 2-3 weeks.
Phase 4: Consolidation and Microbiome Restoration (Weeks 11-16)
Goal
Consolidate mucosal healing gains and restore a healthy microbiome for long-term gut health.
Actions
Peptide Protocol (tapering):
Peptide Dose Route Frequency Duration Evidence Level Purpose
Microbiome Restoration:
Supplement Dose Timing Evidence Level Purpose
Prebiotic fiber (gradually increase) 5-10 g/day With meals Controlled studies Feed beneficial bacteria
Fermented foods Daily portions With meals Observational Diverse microbial exposure
Short-chain fatty acid support (butyrate) 300-600 mg With meals Controlled studies Colonocyte fuel, anti-inflammatory
Dietary Reintroduction:
- Gradual reintroduction of eliminated foods (one every 3-4 days)
- Monitor for symptom recurrence
- Build toward a diverse, whole-foods anti-inflammatory diet
What to Expect
By this phase, most responding patients have experienced significant improvement in digestive symptoms. The focus shifts from acute healing to establishing a sustainable gut environment. Some patients find that foods they previously could not tolerate can be reintroduced successfully.
When to Seek Medical Help
- Blood in stool (new onset or worsening)
- Severe abdominal pain not explained by the treatment protocol
- Unexplained weight loss exceeding 5% of body weight
- Fever during treatment (could indicate infection)
- Signs of allergic reaction to any peptide (hives, swelling, difficulty breathing)
- Symptoms worsening progressively rather than fluctuating and improving
- New symptoms not previously present
Monitoring and Assessment
- Fecal calprotectin at baseline, week 6, and week 14 (for patients with inflammatory bowel disease)
- Intestinal permeability testing at baseline and week 14 (for patients with confirmed increased permeability)
- Stool analysis at baseline and week 14
- Clinical symptom scoring at each visit
- Dietary diary throughout the protocol
Long-Term Maintenance
After completing the protocol:
- Maintain diverse, anti-inflammatory diet
- Continue probiotics and prebiotic fiber long-term
- Zinc carnosine and L-glutamine can be continued at maintenance doses
- Repeat BPC-157 course (4 weeks) if symptoms recur
- Annual stool analysis to monitor microbiome health
- Address new gut-damaging exposures promptly (stress, medication changes, infections)
Clinical Perspective
The gut is where I have the most confidence in peptide therapy, and BPC-157 is the peptide I have used most extensively in this context. The rationale is straightforward: BPC-157 was derived from gastric juice, has extensive preclinical data for GI protection and healing, and is stable when administered orally — making it one of the few peptides where oral administration is mechanistically appropriate.
The addition of KPV for inflammatory bowel conditions has been a meaningful addition to my practice. The NF-kB inhibition mechanism and the PepT1-mediated preferential absorption in inflamed tissue provide an elegant rationale for targeted anti-inflammatory delivery. What I have observed clinically is that patients with documented intestinal inflammation who receive the BPC-157/KPV combination often show improvements in fecal calprotectin levels alongside symptomatic improvement.
I want to be clear about what this protocol is not: it is not a replacement for appropriate medical management of inflammatory bowel disease. Patients with moderate to severe IBD need gastroenterological care, and peptide therapy is an adjunct to — not a substitute for — evidence-based IBD treatment. For patients with milder gut dysfunction, gut barrier issues, or post-infectious IBS, this protocol may be appropriate as a primary intervention under physician supervision.
The most important insight from my clinical experience is that peptides alone are not sufficient. Patients who address the underlying causes of gut dysfunction — dietary triggers, medication effects, stress, sleep quality — respond significantly better than those who rely on peptides while maintaining the habits that damaged their gut in the first place. The foundation matters more than the peptides.

References
- Sikiric P, et al. “Stable gastric pentadecapeptide BPC 157 in trials for inflammatory bowel disease.” Ann N Y Acad Sci. 2006;1070:324-340.
- Sikiric P, et al. “Toxicity by NSAIDs. Counteraction by stable gastric pentadecapeptide BPC 157.” Curr Pharm Des. 2013;19(1):76-83.
- Dalmasso G, et al. “PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation.” Gastroenterology. 2008;134(1):166-178.
- Kannengiesser K, et al. “Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of IBD.” Inflamm Bowel Dis. 2008;14(3):324-331.
- Vandamme D, et al. “A comprehensive summary of LL-37, the factotum human cathelicidin peptide.” Cell Immunol. 2012;280(1):22-35.
- Rao R, Samak G. “Role of glutamine in protection of intestinal epithelial tight junctions.” J Epithelial Biol Pharmacol. 2012;5(Suppl 1-M7):47-54.
- Mahmood A, et al. “Zinc carnosine, a health food supplement that stabilises small bowel integrity.” Gut. 2007;56(2):168-175.
Disclaimer: This protocol is provided for educational purposes and reflects one physician’s clinical approach. It is not a substitute for individualized medical care. Gastrointestinal conditions require proper diagnosis before treatment. Consult a qualified physician before beginning any supplement or peptide protocol.