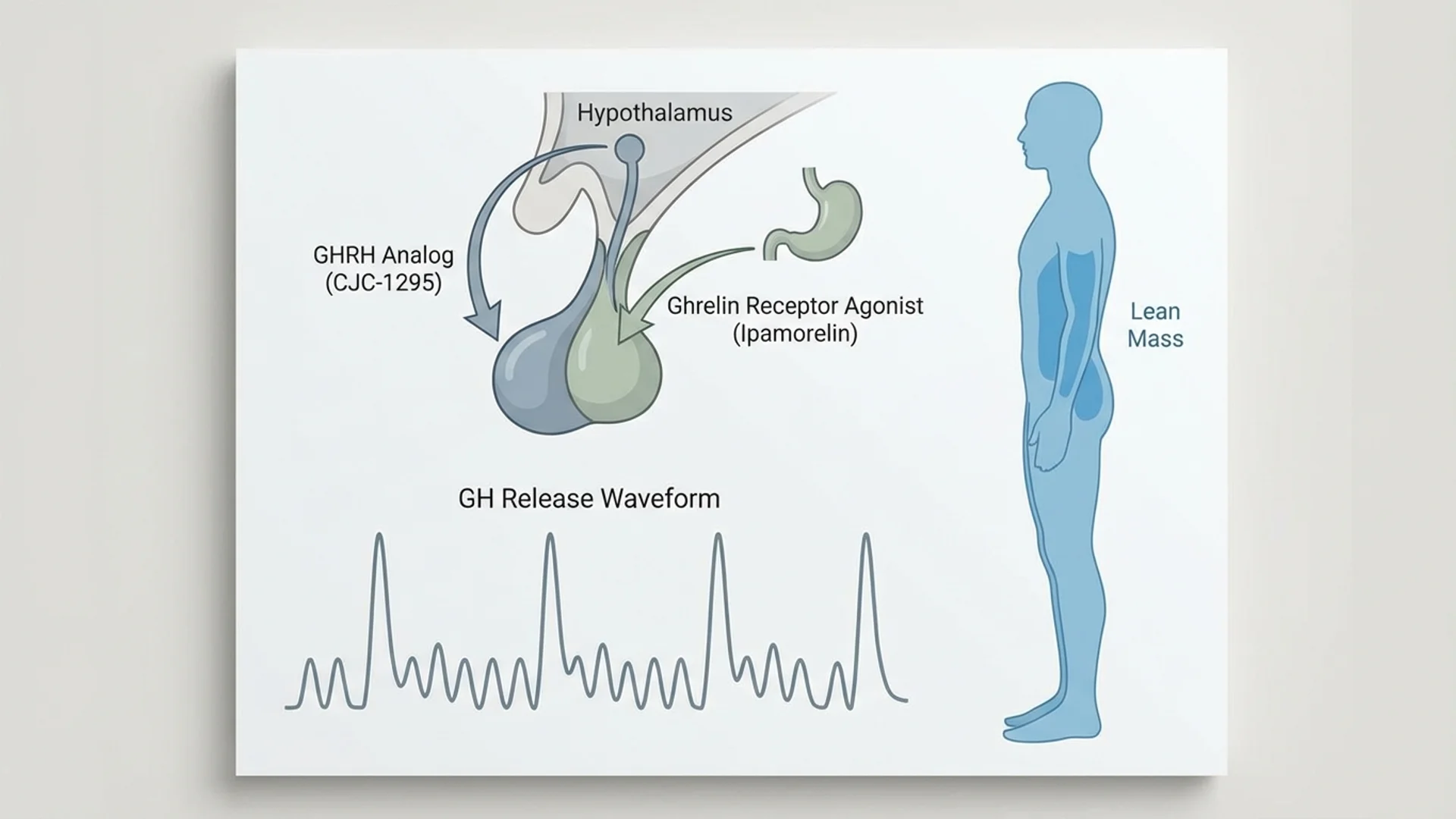
The combination of CJC-1295 and Ipamorelin represents one of the most commonly used growth hormone secretagogue protocols in clinical practice. Unlike direct growth hormone administration, these peptides stimulate the body’s own pulsatile release of growth hormone — a distinction that has both physiological and regulatory implications. Here is what the evidence supports, where it falls short, and how I approach these peptides in clinical practice.
At a Glance
Property Detail
Type CJC-1295: synthetic GHRH analog; Ipamorelin: synthetic GH secretagogue
Amino acid count CJC-1295: 29; Ipamorelin: 5
Primary mechanism Stimulation of pulsatile GH release from the anterior pituitary
Evidence level Controlled human studies (individual peptides); clinical practice (combination)
Regulatory status Not FDA-approved; restricted from US compounding (2023); varies internationally
Route of administration SC injection
What Are CJC-1295 and Ipamorelin?
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), the hypothalamic peptide that stimulates the anterior pituitary to release growth hormone. The “CJC” designation refers to the research group (ConjuChem) that developed it. The version most commonly used in clinical practice is Modified GRF 1-29 (also called CJC-1295 without DAC), which is the first 29 amino acids of GHRH with four amino acid substitutions that improve metabolic stability. The DAC (Drug Affinity Complex) version, which binds to albumin and extends the half-life to days, is less commonly used in clinical practice due to concerns about sustained non-pulsatile GH elevation.

Ipamorelin is a synthetic pentapeptide that acts as a selective growth hormone secretagogue receptor (GHSR) agonist. Unlike other GH secretagogues such as GHRP-2 and GHRP-6, Ipamorelin is notably selective — it stimulates GH release without significantly affecting cortisol, prolactin, or ACTH levels. This selectivity is clinically relevant because it means Ipamorelin can enhance GH release without the side effects associated with cortisol and prolactin elevation.
The Rationale for Combination
The combination of CJC-1295 and Ipamorelin is based on the physiological principle that GH release from the pituitary is regulated by two signals: GHRH (the “accelerator”) and ghrelin/GH secretagogues (acting through a different receptor, also promoting release). By stimulating both pathways simultaneously, the combination produces a more robust and physiological GH pulse than either peptide alone.
This dual-pathway approach also preserves the pulsatile nature of GH release — the body’s normal pattern of releasing GH in bursts, primarily during sleep. This is in contrast to exogenous GH injection, which delivers a bolus of GH that does not replicate the natural pulsatile pattern.
Mechanism of Action
Primary Pathways
- GHRH Receptor Activation (CJC-1295): CJC-1295 binds to GHRH receptors on somatotroph cells in the anterior pituitary, stimulating the synthesis and release of growth hormone. It also promotes GH gene transcription, which means it increases the pituitary’s capacity to produce GH over time — not just its immediate release.
- GHS Receptor Activation (Ipamorelin): Ipamorelin activates the growth hormone secretagogue receptor (GHS-R1a), the same receptor targeted by ghrelin. This provides a complementary stimulus for GH release through a distinct intracellular signaling pathway (primarily involving IP3/DAG and calcium signaling, as opposed to the cAMP pathway used by GHRH).
- IGF-1 Elevation: The downstream effect of increased GH secretion is elevation of insulin-like growth factor 1 (IGF-1), primarily produced by the liver in response to GH stimulation. IGF-1 is the principal mediator of GH’s anabolic effects on muscle, bone, and other tissues.
- Negative Feedback Preservation: Unlike exogenous GH, which can suppress the hypothalamic-pituitary axis, CJC-1295/Ipamorelin works within the body’s feedback system. Somatostatin, the GH-inhibiting hormone, still functions normally, preventing excessive GH elevation and maintaining physiological pulsatility.
Research Assessment
What the Evidence Shows
Claimed Effect Evidence Level Key Studies Assessment
IGF-1 elevation Controlled human studies Teichman 2006 Strong
Body composition improvement Clinical observation, limited studies Various Promising
Improved sleep quality Clinical observation
Preliminary
Enhanced recovery Clinical observation
Preliminary
Selective GH release (Ipamorelin) Controlled studies Raun 1998, Hansen 1999 Strong (selectivity confirmed)
CJC-1295 has been studied in human clinical trials. Teichman and colleagues (2006) demonstrated that a single injection of CJC-1295 produced sustained GH and IGF-1 elevation for 6-8 days (with the DAC version). The modified GRF 1-29 version without DAC produces a shorter, more pulsatile GH elevation lasting several hours.
Ipamorelin has been studied in both animal and human settings. The selectivity data is robust — multiple studies confirm that Ipamorelin stimulates GH release without the cortisol, prolactin, and ACTH elevation seen with GHRP-2 and GHRP-6.
What the Evidence Does Not Show
The combination of CJC-1295 and Ipamorelin has not been studied in a rigorous clinical trial for any specific clinical outcome. The body composition benefits, improved sleep quality, and enhanced recovery that are commonly attributed to this combination are based on clinical observation and physiological reasoning from the known effects of GH optimization.
The long-term safety of chronic GH secretagogue use has not been established. While the physiological pulsatile pattern is presumably safer than supraphysiological exogenous GH, this assumption has not been formally tested over multi-year durations.
Dosing
Route Dose Range Frequency Duration Source of Data
SC (Ipamorelin) 100-300 mcg Once daily (pre-bedtime) or 5 days/week 8-12 week cycles Clinical practice
Note: These peptides are typically administered together as a single injection before bedtime to coincide with the natural nocturnal GH surge. Dosing is based on clinical practice, not formal dose-finding studies for the combination. Cycling (8-12 weeks on, 4 weeks off) is commonly recommended to prevent pituitary desensitization, though the necessity of cycling has not been established in clinical trials.
Safety and Side Effects
Known
Common side effects include injection site reactions, water retention (particularly in the first 1-2 weeks), transient numbness or tingling in the extremities (a recognized effect of GH elevation), and increased appetite (more common with Ipamorelin). These effects are generally mild and dose-dependent.
Theoretical Concerns
The oncological implications of sustained GH/IGF-1 elevation deserve attention. IGF-1 is a growth factor that promotes cell proliferation, and epidemiological data links chronically elevated IGF-1 levels to increased cancer risk. The question is whether the modest, pulsatile GH elevation produced by secretagogues carries the same risk as chronically elevated levels. This question remains unanswered.
Insulin sensitivity may be affected by GH secretagogues. GH is a counter-regulatory hormone that opposes insulin action. Sustained GH elevation can worsen insulin resistance, which is particularly relevant in patients with pre-diabetes or metabolic syndrome.
Contraindications
Active malignancy. Diabetes or significant insulin resistance (relative contraindication — requires careful monitoring). Pituitary disorders. Pregnancy and lactation. Children (risk of premature growth plate closure).
Drug and Supplement Interactions
GH secretagogues should be used with caution alongside insulin or oral hypoglycemic agents due to the counter-regulatory effects of GH on glucose metabolism. Concurrent use with exogenous GH is contraindicated due to risk of supraphysiological GH levels. Glucocorticoids may blunt the GH-releasing effects of these peptides.
Clinical Perspective
CJC-1295/Ipamorelin is a peptide combination that I approach with measured enthusiasm. The pharmacology is sound — stimulating pulsatile, physiological GH release through two complementary pathways is more elegant than direct GH injection. And the selectivity of Ipamorelin, in particular, addresses many of the side effect concerns associated with older GH secretagogues.
In my clinical experience, the patients who benefit most from GH secretagogues are those with documented evidence of GH insufficiency — typically adults over 40 with low IGF-1 levels who present with symptoms consistent with GH decline: increased adiposity (particularly visceral), reduced lean mass, poor recovery from exercise or injury, and diminished sleep quality.
What I tell my patients is that GH secretagogues are not anabolic steroids. The GH elevation produced by CJC-1295/Ipamorelin is within the physiological range. Patients should expect gradual improvements in body composition, recovery, and sleep quality over 8-12 weeks — not dramatic overnight changes. Those who approach these peptides with realistic expectations are consistently the most satisfied.
I monitor IGF-1 levels, fasting glucose, and insulin at baseline and during treatment. If IGF-1 exceeds the upper end of the age-appropriate reference range, or if glucose homeostasis deteriorates, I adjust or discontinue the protocol. This is not a set-and-forget intervention.
The regulatory landscape has changed significantly in the United States, where these peptides have been restricted from compounding. In jurisdictions where they remain available through medical channels, I continue to find them a useful tool for appropriate patients — always within the context of comprehensive metabolic assessment and monitoring.

References
- Teichman SL, et al. “Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults.” J Clin Endocrinol Metab. 2006;91(3):799-805.
- Raun K, et al. “Ipamorelin, the first selective growth hormone secretagogue.” Eur J Endocrinol. 1998;139(5):552-561.
- Hansen BS, et al. “The growth hormone secretagogue ipamorelin: pharmacokinetics and pharmacodynamics in beagles.” J Pharmacol Exp Ther. 1999;288(1):57-63.
- Nass R, et al. “Effects of an oral ghrelin mimetic on body composition and clinical outcomes in healthy older adults.” Ann Intern Med. 2008;149(9):601-611.
- Veldhuis JD, et al. “Differential impact of age, sex steroid hormones, and obesity on basal versus pulsatile growth hormone secretion in men.” J Clin Endocrinol Metab. 1995;80(11):3209-3222.
Disclaimer: This peptide profile is intended for educational purposes. CJC-1295 and Ipamorelin are not FDA-approved and have been restricted from US compounding. Availability varies by jurisdiction. Consult a qualified physician before pursuing any peptide therapy.