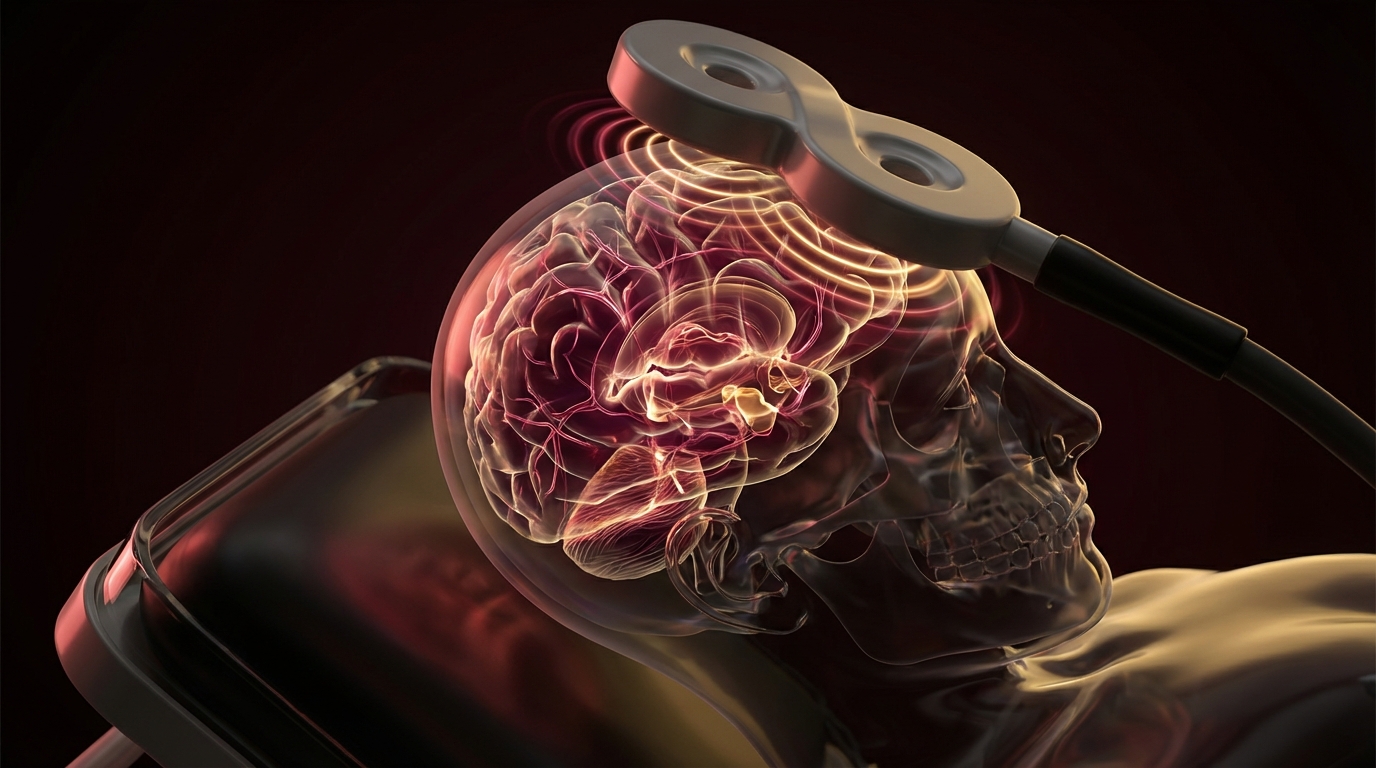
If you have treatment-resistant depression, persistent brain fog, or a neurological condition that has not responded to conventional therapy, you have probably come across the term TMS. The technology has been around for decades, it has strong clinical evidence for specific indications, and it represents one of the most important developments in non-invasive brain stimulation. But there is a newer evolution — Transcranial Pulse Stimulation, or TPS — that I consider the single most important neuromodulation tool in my clinical practice.
Let me walk you through the landscape.
At a Glance
| Property | Detail |
|---|---|
| Evidence Level | Strong (rTMS for depression); Moderate to Emerging (TPS for neurodegeneration, CFS) |
| Primary Use | Treatment-resistant depression, OCD, neurological conditions, chronic fatigue |
| Key Mechanism | Electromagnetic induction generating focal electrical currents in cortical and subcortical tissue |
| FDA Status | rTMS cleared for depression (2008), OCD (2018), smoking cessation (2020); TPS CE-marked in Europe |
| Session Duration | rTMS: 20-40 minutes; TPS: 30 minutes |
What Is Transcranial Magnetic Stimulation?
Transcranial magnetic stimulation is a non-invasive neuromodulation technique that uses rapidly changing magnetic fields to induce small electrical currents in targeted regions of the brain. The principle is electromagnetic induction — the same physics Faraday described in 1831, applied to neural tissue.
A coil placed against the scalp generates a brief, intense magnetic pulse. This pulse passes through the skull unimpeded (bone is transparent to magnetic fields) and induces an electrical current in the underlying cortex. That current depolarizes neurons, either exciting or inhibiting activity depending on the stimulation parameters.
The first modern TMS device was developed by Anthony Barker and colleagues at the University of Sheffield in 1985. Since then, the technology has evolved considerably — from single-pulse diagnostic tools to repetitive TMS (rTMS) protocols that can produce lasting changes in brain circuit activity.
Here is what the evidence shows: rTMS is not experimental for depression. It is an FDA-cleared, guideline-supported treatment with a robust evidence base.
How TMS Works: The Mechanism
Electromagnetic Induction
The TMS coil generates a magnetic field of approximately 1.5-2 Tesla — comparable to an MRI scanner, but focused and pulsed rather than static. When this rapidly changing magnetic field passes through cortical tissue, it induces electrical currents perpendicular to the magnetic field lines.
These induced currents are strong enough to depolarize neurons, triggering action potentials. The key insight is selectivity: by positioning the coil precisely and adjusting stimulation parameters, clinicians can target specific brain regions while leaving adjacent areas relatively unaffected.
Repetitive TMS (rTMS)
Single pulses of TMS are useful for diagnostics — mapping motor cortex, measuring cortical excitability. But the therapeutic applications come from repetitive stimulation.
High-frequency rTMS (typically 10-20 Hz) increases cortical excitability in the stimulated region. Applied to the left dorsolateral prefrontal cortex (DLPFC), this is the standard protocol for depression — the rationale being that depression is associated with hypoactivity of the left prefrontal cortex.
Low-frequency rTMS (1 Hz) decreases cortical excitability. This inhibitory protocol is sometimes applied to the right DLPFC for depression, or to overactive brain regions in other conditions.
The effects of a single rTMS session last minutes to hours. But with repeated daily sessions over weeks, the changes become persistent — a phenomenon attributed to long-term potentiation (LTP) and long-term depression (LTD) of synaptic connections. This is neuroplasticity being directed by an external stimulus.
Deep TMS
Standard figure-8 coils stimulate cortex at a depth of approximately 1.5-2 cm. Deep TMS, using H-coils developed by Brainsway, can reach structures at depths of 3-4 cm, including limbic circuits involved in mood, reward, and compulsive behavior.
Deep TMS received FDA clearance for OCD in 2018, targeting the anterior cingulate cortex and medial prefrontal regions — areas inaccessible to conventional figure-8 coils.
The Evidence
What We Know: FDA-Cleared Indications
Treatment-Resistant Depression. This is where the evidence is strongest. The pivotal trial by O’Reardon et al. (2007) demonstrated significant improvement in depression scores compared to sham stimulation after 4-6 weeks of daily rTMS. Since then, multiple RCTs and meta-analyses have confirmed efficacy. A meta-analysis by Berlim et al. (2014) pooling data from 29 RCTs found that active rTMS was significantly superior to sham, with response rates of approximately 29% and remission rates of approximately 19% — clinically meaningful numbers for a treatment-resistant population.
The Stanford Accelerated Intelligent Neuromodulation Therapy (SAINT) protocol, published by Cole et al. in 2020, demonstrated remission rates of approximately 79% using theta-burst stimulation delivered in an accelerated 5-day format with functional MRI-guided targeting. This was a small open-label study, but it generated significant interest and has been followed by larger controlled trials.
Obsessive-Compulsive Disorder. Deep TMS targeting the anterior cingulate and medial prefrontal cortex showed significant reduction in OCD symptoms compared to sham in the pivotal trial leading to FDA clearance. The evidence is moderate — fewer large RCTs than for depression, but the mechanistic rationale and initial data are solid.
Smoking Cessation. Deep TMS received FDA clearance for smoking cessation in 2020 based on a multicenter RCT demonstrating significantly higher quit rates compared to sham.
What the Evidence Supports But Lacks FDA Clearance For
Chronic pain, migraine prevention, PTSD, tinnitus, and stroke rehabilitation all have varying degrees of evidence from controlled trials. Some of these applications have guideline support in Europe. The evidence ranges from moderate (chronic pain) to emerging (tinnitus).
What I See in Practice: TPS
Here is where I need to be direct about what I consider the most important development in this field.
Transcranial Pulse Stimulation (TPS) is a newer technology that uses short, focused ultrasound-like shockwave pulses rather than magnetic fields. The device (NEUROLITH, developed by Storz Medical) delivers mechanical pressure waves that penetrate to subcortical depths of up to 8 cm — far deeper than any magnetic coil.
In my clinical experience, TPS is the number one treatment I reach for in neurological conditions, chronic fatigue syndrome, and neuropsychiatric disorders. The reasons are practical and clinical:
-
Depth of penetration. TPS reaches structures that rTMS cannot — hippocampus, thalamus, basal ganglia. For neurodegenerative conditions where deep structures are affected, this matters.
-
No seizure risk. TPS does not induce electrical currents in the same manner as TMS. The mechanical stimulation mechanism carries no seizure risk, which simplifies patient selection.
-
Neuroplasticity activation. TPS appears to upregulate BDNF, VEGF, and other neurotrophic factors. In patients with Alzheimer’s disease, the NEUROLITH pivotal study by Beisteiner et al. (2020) demonstrated significant improvement in cognitive function after 6 sessions over 2 weeks, with effects persisting at 3-month follow-up.
-
Chronic fatigue. In patients with CFS and post-COVID fatigue, I observe consistent improvements in cognitive function, energy, and mood after TPS treatment courses. The evidence base for this indication is clinical observation at this stage, not controlled trials — I want to be transparent about that. But the consistency of what I see in practice is compelling.
What I tell my patients: TMS is proven technology with strong evidence for depression. TPS is the evolution — deeper penetration, broader applications, excellent safety profile. For neurological conditions and fatigue syndromes, TPS is where I start.
rTMS vs Deep TMS vs TPS: A Comparison
| Feature | rTMS (Figure-8) | Deep TMS (H-Coil) | TPS (NEUROLITH) |
|---|---|---|---|
| Penetration depth | 1.5-2 cm | 3-4 cm | Up to 8 cm |
| Mechanism | Electromagnetic induction | Electromagnetic induction | Mechanical shockwave |
| Session duration | 20-40 min | 20 min | 30 min |
| Sessions per course | 20-30 (daily) | 20-30 (daily) | 6-12 (2-3x/week) |
| Seizure risk | Low but present | Low but present | None reported |
| FDA/CE status | FDA cleared | FDA cleared | CE marked |
| Best evidence | Depression | Depression, OCD | Alzheimer’s, emerging for CFS |
What a TMS Session Looks Like
For patients considering TMS, here is what to expect:
Before treatment: A psychiatric or neurological evaluation determines whether TMS is appropriate. For rTMS targeting depression, the motor threshold is established — the minimum stimulation intensity needed to produce a visible twitch in the hand. This calibrates the dose to your individual neuroanatomy.
During treatment: You sit in a chair. The coil or device is positioned against your head. For rTMS, you hear clicking sounds and feel tapping on the scalp. For TPS, you feel pressure pulses. Neither requires anesthesia. Most patients describe the sensation as unusual but not painful.
Treatment course: Standard rTMS for depression involves daily sessions, 5 days per week, for 4-6 weeks — approximately 20-30 sessions. The SAINT accelerated protocol compresses this to 5 days. TPS protocols are shorter — typically 6-12 sessions over 2-6 weeks.
After treatment: You walk out and resume normal activities. No recovery period. No cognitive impairment. This is one of the practical advantages over pharmacotherapy — no systemic side effects.
Safety and Considerations
Known Side Effects
The most common side effect of rTMS is scalp discomfort at the stimulation site, reported by approximately 30-40% of patients. This typically diminishes over the course of treatment. Headache occurs in 10-20% of sessions.
The most serious risk of rTMS is seizure, occurring at a rate of approximately 0.1% or less when standard safety guidelines are followed. This risk is managed through careful screening (no uncontrolled epilepsy, no metallic implants near the coil) and adherence to established stimulation parameters.
TPS has an excellent safety profile in published data. No seizures have been reported. Transient mild headache is the most common adverse event.
Who Should Not Receive TMS
- Patients with metallic implants in or near the head (cochlear implants, deep brain stimulators, metallic fragments)
- Patients with uncontrolled epilepsy (for rTMS; TPS does not carry the same risk)
- Patients on medications that significantly lower seizure threshold, unless carefully managed
The Bottom Line
Transcranial magnetic stimulation is a proven, FDA-cleared treatment for depression and OCD with a strong evidence base. It works by using electromagnetic induction to modulate neural circuit activity in targeted brain regions. For patients who have not responded to medication, rTMS offers a non-invasive alternative with a favorable side effect profile.
TPS represents the next evolution. In my clinical practice, it is the neuromodulation tool I reach for most often — particularly for neurological conditions, chronic fatigue, and neuropsychiatric disorders where deeper brain structures need to be engaged. The evidence base is younger than rTMS, but the clinical results I observe are consistent and the safety profile is excellent.
The nuance matters: TMS is not a miracle. It is a tool — a sophisticated, evidence-backed tool that works for the right patients with the right indications.
References
-
O’Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216. PMID: 17573044.
-
Berlim MT, van den Eynde F, Tovar-Perdomo S, Daskalakis ZJ. Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rTMS) for treating major depression: a systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol Med. 2014;44(2):225-239. PMID: 23507264.
-
Beisteiner R, Matt E, Fan C, et al. Transcranial pulse stimulation with ultrasound in Alzheimer’s disease — a new navigated focal brain therapy. Adv Sci. 2020;7(3):1902583. PMID: 32042569.
-
Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford Accelerated Intelligent Neuromodulation Therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716-726. PMID: 32252538.
-
Carmi L, Tendler A, Bystritsky A, et al. Efficacy and safety of deep transcranial magnetic stimulation for obsessive-compulsive disorder: a prospective multicenter randomized double-blind placebo-controlled trial. Am J Psychiatry. 2019;176(11):931-938. PMID: 31109199.
Disclaimer: This article is for educational purposes. Transcranial magnetic stimulation and transcranial pulse stimulation are medical procedures that should only be administered by qualified healthcare professionals. Treatment decisions should be made in consultation with your physician based on your individual clinical situation.