Few topics in medicine generate as much hope — and as much misrepresentation — as stem cell therapy. The concept is powerful: use the body’s own regenerative cells to repair damaged tissue, reverse disease, and potentially slow aging. The reality is that stem cell medicine exists on a spectrum from rigorously validated to frankly exploitative, and patients deserve to understand where on that spectrum any given treatment falls.
Let me be direct. Some stem cell therapies are among the most important advances in modern medicine. Others, as currently marketed, are selling a promise the science does not yet support.
What Stem Cells Actually Do
Stem cells are distinguished by two capabilities: self-renewal (they can divide to produce more stem cells) and differentiation (they can become specialized cell types). Different types of stem cells exist:
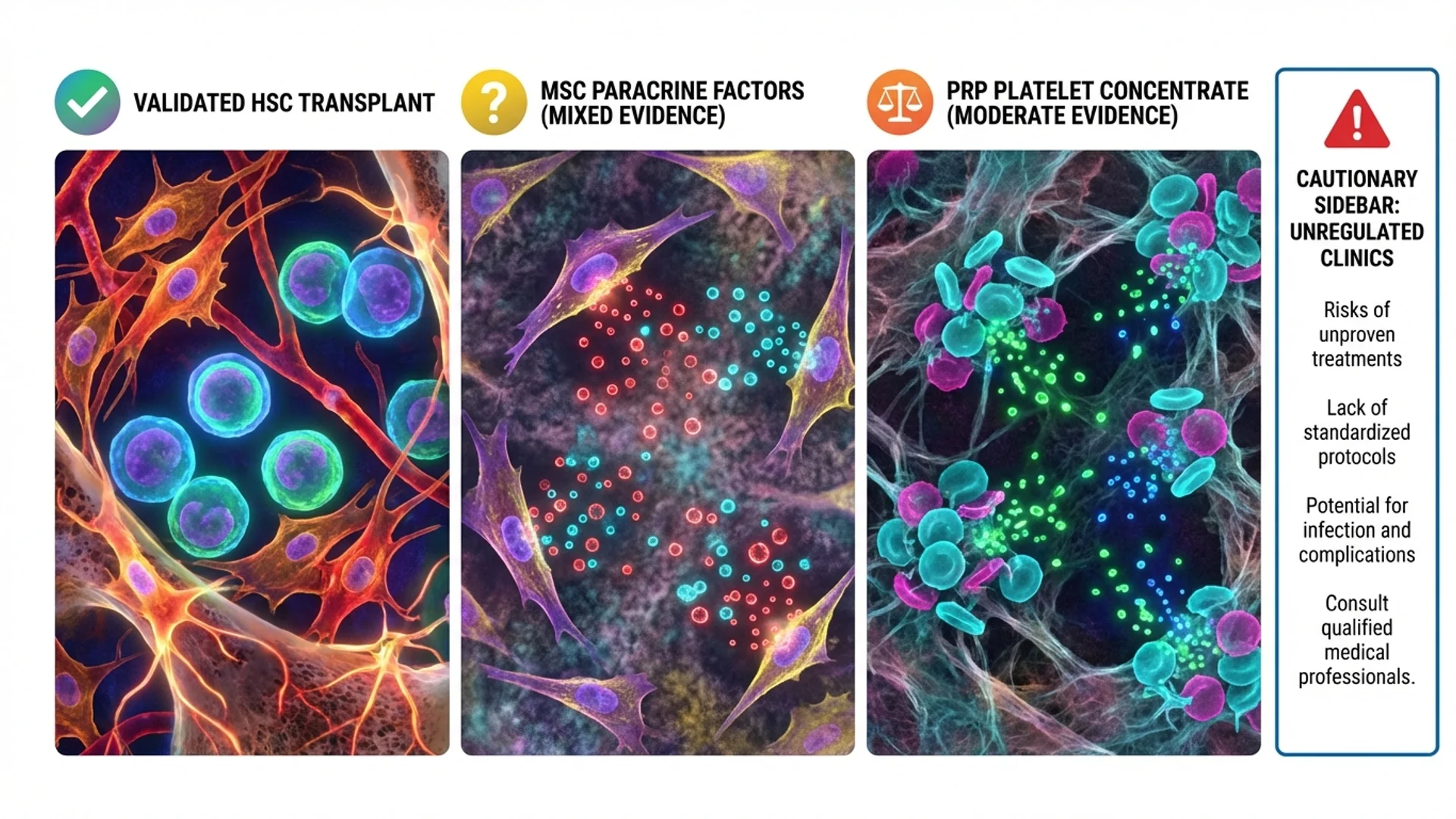
Hematopoietic stem cells (HSCs): Found in bone marrow. Give rise to all blood cell types. HSC transplantation for leukemia and other blood cancers is well-established, FDA-approved, and life-saving. This is not controversial.
Mesenchymal stem/stromal cells (MSCs): Found in bone marrow, adipose tissue, umbilical cord tissue, and other sources. These are the cells used in most of the therapies marketed for longevity, orthopedic conditions, and chronic disease. MSCs appear to work primarily through paracrine signaling — secreting anti-inflammatory and immunomodulatory factors — rather than by replacing damaged cells directly [1].
Induced pluripotent stem cells (iPSCs): Adult cells reprogrammed to an embryonic-like state. Enormous research potential but not yet widely available clinically outside of specific trials.
Embryonic stem cells: The most versatile but ethically complex. Not used in the therapies I discuss here.
Where the Evidence Supports Use
Orthopedic applications. MSC injections for knee osteoarthritis have shown modest benefit in multiple randomized controlled trials — reduced pain, improved function, and some evidence of cartilage preservation [2]. The effect sizes are moderate, and not all patients respond. But this is an area with genuine clinical evidence.
Hematologic malignancies. As noted, hematopoietic stem cell transplantation is established medicine.
Certain autoimmune conditions. Autologous HSC transplantation for severe, treatment-refractory multiple sclerosis has shown striking results in clinical trials, with some patients achieving long-term remission [3]. This is a high-risk procedure reserved for severe cases, but the outcomes have been remarkable.
Wound healing. Stem cell-derived products have shown promise in chronic wound healing and burn treatment.
Where the Evidence Is Preliminary
Systemic IV MSC infusion for “rejuvenation.” This is the treatment most commonly marketed by longevity clinics. The patient receives an IV infusion of MSCs — typically derived from umbilical cord tissue or bone marrow — with claims of systemic anti-inflammatory, immunomodulatory, and regenerative effects.
The mechanistic rationale has some basis. IV-administered MSCs do release immunomodulatory factors. Animal studies have shown that young MSCs can improve various markers in aged animals. Some human studies, particularly in frailty and aging, have shown safety and preliminary signals of benefit [4].
However, I want to be transparent about the limitations:
- Most IV-administered MSCs become trapped in the lungs within hours and do not reach target tissues in meaningful numbers
- The therapeutic effects, to the extent they occur, are likely paracrine (secreted factors) rather than engraftment-based
- Cell viability, potency, and characterization vary enormously between clinics and preparations
- Large, rigorous randomized trials for longevity or anti-aging indications are lacking
- The cost (often $10,000-$50,000 per treatment) is not commensurate with the current evidence level
Exosomes. Exosome therapy — using the extracellular vesicles secreted by stem cells rather than the cells themselves — has generated significant interest. The rationale is that the therapeutic effects of MSCs are mediated by their secreted factors, so why not use the factors directly? This is a reasonable hypothesis under active investigation. However, exosome products currently available at clinics are poorly standardized, poorly regulated, and the clinical evidence base is minimal. The FDA has issued multiple warnings about unapproved exosome products.
What Concerns Me
The stem cell industry has a significant credibility problem. Many clinics market stem cell therapy for conditions where no meaningful evidence exists — autism, Parkinson’s disease, erectile dysfunction, general “anti-aging” — at substantial cost, often to vulnerable patients.
This is not all of stem cell medicine. But it is enough of it that patients should approach any stem cell offering with informed skepticism.
Questions I recommend patients ask before any stem cell procedure:
- What is the source, type, and characterization of the cells?
- What clinical trial data supports this specific application?
- What are the realistic expected outcomes, and what is the expected response rate?
- What are the known risks and adverse events?
- Is this clinic participating in any registered clinical trials?
- What follow-up and monitoring is provided?
My Practice
I am cautious about stem cell therapy in its current form for longevity applications. I use MSC-based treatments in specific orthopedic and musculoskeletal contexts where clinical evidence exists. For systemic longevity applications, I consider it an area of legitimate scientific interest that is not yet ready for routine clinical use.
When patients express interest in stem cell therapy for longevity, I discuss the evidence honestly, outline what we know and what we do not know, and — if they choose to proceed — ensure they do so with appropriate expectations and at reputable facilities that provide adequate characterization and follow-up.
The field is advancing rapidly. Five to ten years from now, stem cell and exosome therapies may be standard components of longevity medicine. But promising early science and validated clinical therapy are not the same thing, and patients deserve to know which one they are receiving.
References
- Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. Journal of Cellular Biochemistry. 2006;98(5):1076-1084.
- Pers YM, et al. Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: a phase I dose-escalation trial. Stem Cells Translational Medicine. 2016;5(7):847-856.
- Burt RK, et al. Effect of nonmyeloablative hematopoietic stem cell transplantation vs continued disease-modifying therapy on disease progression in patients with relapsing-remitting multiple sclerosis. JAMA. 2019;321(2):165-174.
- Tompkins BA, et al. Allogeneic mesenchymal stem cells ameliorate aging frailty: A phase II randomized, double-blind, placebo-controlled clinical trial. Journals of Gerontology Series A. 2017;72(11):1513-1522.
This content is educational and does not constitute medical advice. Stem cell therapy should only be undertaken at reputable medical facilities after thorough discussion of the evidence, expectations, and risks with a qualified physician.