At a Glance
| Property | Value |
|---|---|
| Evidence Level | Moderate (observational longevity data strong; RCT for aging indication pending — TAME trial) |
| Primary Use | AMPK activation, mTOR inhibition, potential geroprotection |
| Key Mechanism | AMPK activation, mitochondrial complex I inhibition, improved insulin sensitivity |
Metformin Dosage for Anti-Aging: The Physician’s Perspective
If you are considering metformin for longevity rather than diabetes, the first thing you need to understand is that no regulatory agency has approved metformin for anti-aging. Every prescription for this purpose is off-label. That does not mean it is without evidence — it means the evidence base is different from what you see with a standard drug indication, and the dosing strategy reflects that ambiguity.
Here is what the research actually says about dosing, what longevity physicians are prescribing, and how to think about this critically.
Why Metformin for Non-Diabetics?
The interest in metformin as a geroprotective agent comes from three converging lines of evidence:
Observational data. Bannister et al. published a striking finding in 2014: type 2 diabetics on metformin had lower all-cause mortality than age-matched non-diabetic controls (1). Diabetics — a population with inherently higher mortality risk — were outliving their healthy counterparts. This was not a small study; it included 78,241 metformin-treated patients and 90,463 matched controls over a median 2.8-year follow-up.
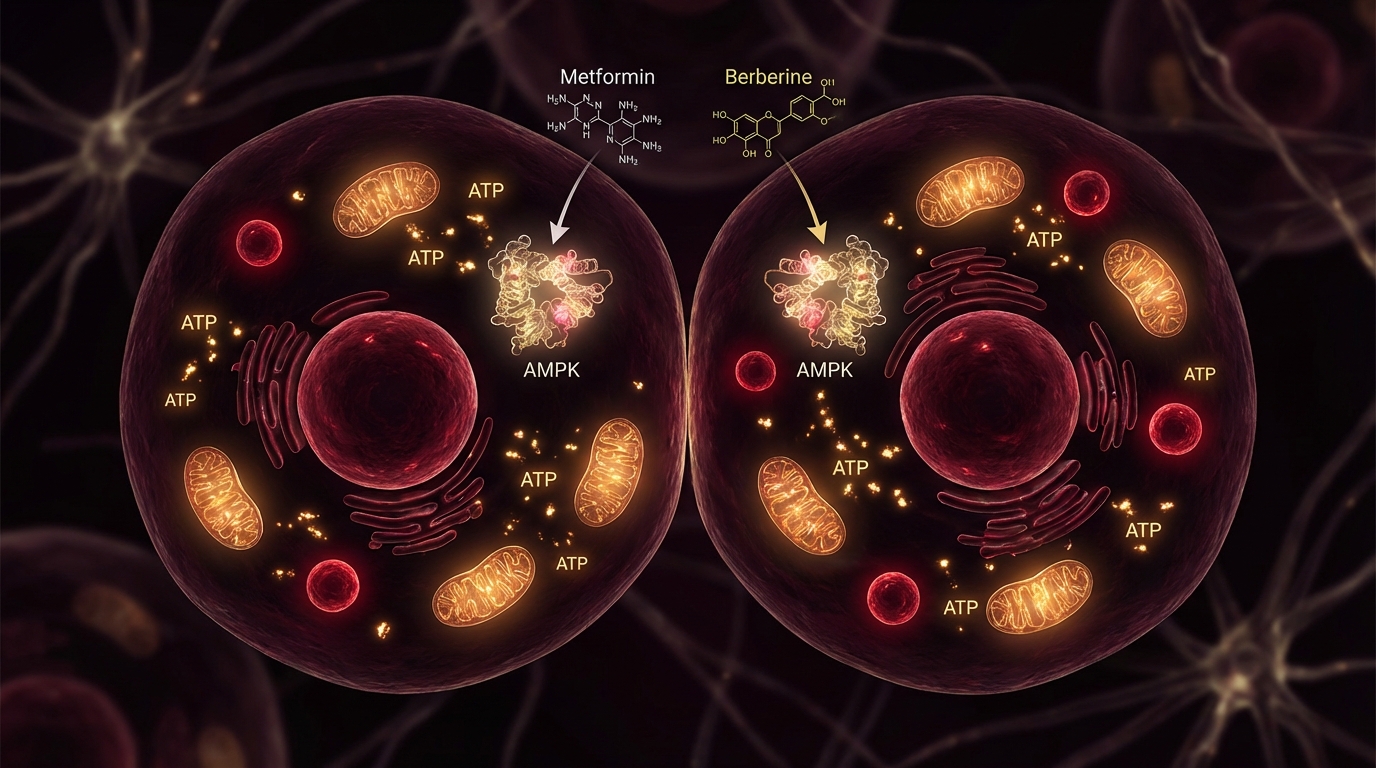
AMPK activation. Metformin activates AMP-activated protein kinase (AMPK), a master metabolic regulator that is also activated by caloric restriction and exercise — two of the most robustly demonstrated longevity interventions. AMPK activation triggers downstream effects including improved insulin signaling, reduced mTOR activity (which slows cellular proliferation and promotes autophagy), and enhanced mitochondrial function.
The TAME trial. The Targeting Aging with Metformin (TAME) trial is the first FDA-approved clinical trial to test a drug with “aging” as the endpoint. It targets 3,000 individuals aged 65-79 and will use 1,500 mg/day of metformin. If positive, it would be the first drug approved for an aging indication. The trial’s design itself tells you something about the strength of the preliminary evidence — the FDA does not approve aging trials lightly.
Current Dosing Protocols
What the Studies Used
| Study / Trial | Population | Daily Dose | Duration | Key Finding |
|---|---|---|---|---|
| MILES (Metformin in Longevity Study) | Non-diabetics, age 40+ | 1,700 mg/day | 6 weeks | Gene expression changes in muscle and adipose tissue consistent with improved metabolic health (2) |
| TAME (Targeting Aging with Metformin) | Non-diabetics, age 65-79 | 1,500 mg/day | 6 years (planned) | Ongoing |
| Bannister et al. (observational) | T2D patients | ~2,000 mg/day (mean) | 2.8 years median | Lower all-cause mortality vs non-diabetic controls |
What Longevity Physicians Prescribe
In clinical practice, the consensus among physicians prescribing metformin off-label for longevity has converged on a range that is lower than the standard diabetic dose:
- Starting dose: 500 mg/day (extended-release), taken with dinner
- Target dose: 1,000-1,500 mg/day (after 4-6 week titration)
- Maximum for longevity use: 1,500 mg/day (based on TAME trial design)
The rationale for lower doses: the goal in non-diabetics is not maximal glucose reduction. It is AMPK activation and mTOR modulation with minimal side effects. The dose-response curve for AMPK activation plateaus below the maximum tolerated dose for glucose control, which means you can achieve the longevity-relevant molecular effects at doses that produce fewer adverse effects.
My Titration Protocol
Here is what I recommend for patients starting metformin for longevity purposes:
Week 1-2: 500 mg extended-release (ER) with dinner Week 3-4: 1,000 mg ER with dinner (two 500 mg tablets or one 1,000 mg tablet) Week 5-6: If tolerating well, 1,500 mg ER with dinner (optional — many patients stay at 1,000 mg)
Key points:
- Always use extended-release (ER/XR). Immediate-release metformin has a 25-30% rate of GI side effects (nausea, diarrhea, bloating). Extended-release cuts this roughly in half.
- Always take with food. This further reduces GI disturbance and improves absorption consistency.
- Evening dosing is preferred for longevity use because metformin’s effects on hepatic glucose output are most relevant during the overnight fasting period.
The Exercise Question
This is where the nuance matters, and where I diverge from some longevity physicians who prescribe metformin to everyone regardless of lifestyle.
Konopka et al. published a concerning finding in 2019: metformin blunted the improvements in aerobic capacity and mitochondrial respiration that normally accompany exercise training in older adults (3). The effect was not trivial — metformin-treated subjects showed significantly less improvement in VO2 max and muscle mitochondrial respiration compared to placebo.
The implication: metformin may partially counteract the benefits of exercise, specifically for mitochondrial adaptation. Since exercise is itself the most robust longevity intervention we know of, this creates a genuine dilemma.
My approach:
- For patients who exercise intensely (>4 hours/week of structured cardio and/or resistance training): I often recommend skipping metformin on training days, or using it only on rest days.
- For patients who are sedentary or exercise minimally: the benefit-risk balance favors metformin more clearly, as they are less likely to be generating the exercise-induced AMPK activation that metformin mimics.
- For patients who exercise moderately (2-4 hours/week): individual assessment. Monitor VO2 max over 3-6 months. If aerobic capacity plateaus or declines, reconsider the metformin dose.
Monitoring and Lab Work
If you are taking metformin for longevity, the following monitoring is essential:
Every 6 Months
- Fasting glucose and HbA1c. You are not diabetic, so these should remain in normal range. If glucose drops below 70 mg/dL, reduce the dose.
- Comprehensive metabolic panel. Monitor kidney function (creatinine, eGFR). Metformin is contraindicated if eGFR falls below 30 mL/min and should be dose-reduced below 45 mL/min.
Annually
- Vitamin B12. Metformin inhibits B12 absorption in the terminal ileum. Deficiency develops in 5-10% of long-term users. Symptoms include fatigue, neuropathy, and cognitive changes — which can be mistaken for aging. Check serum B12 and methylmalonic acid. For more on B12 and the full side effect profile, see metformin side effects for anti-aging users.
- Lactate. Lactic acidosis is the most feared metformin complication, though it is extremely rare at longevity doses in patients with normal kidney function. A baseline and annual lactate level provides reassurance.
Optional but Useful
- VO2 max testing. Baseline and at 6 months to detect any exercise blunting effect.
- Insulin and HOMA-IR. Tracks improvement in insulin sensitivity over time.
- Body composition (DEXA). Monitors for any unintended lean mass changes.
Metformin vs. Natural AMPK Activators
A fair question: if metformin works primarily through AMPK activation, why not use natural AMPK activators instead? Berberine, exercise, caloric restriction, and certain polyphenols all activate AMPK.
This is a legitimate consideration, and I discuss it in detail in my metformin vs. berberine comparison. The short answer: metformin has vastly more clinical data (over 60 years of human use, thousands of studies), a well-characterized safety profile, and the only FDA-approved aging trial in history. Berberine may offer similar mechanistic benefits with potentially less exercise interference, but the evidence base is orders of magnitude smaller.
Safety and Considerations
Common Side Effects (Dose-Dependent)
- GI disturbance. Nausea, diarrhea, bloating, metallic taste. Usually resolves within 2-4 weeks. Extended-release formulations significantly reduce incidence.
- Reduced appetite. Often considered a benefit, but can be problematic for underweight patients or those with high training volumes.
- B12 malabsorption. Develops over months to years. Supplement with methylcobalamin 1,000 mcg/day as a preventive measure.
Rare but Serious
- Lactic acidosis. Extremely rare at longevity doses with normal renal function. Risk increases with renal impairment, alcohol abuse, and acute illness.
- Hypoglycemia. Uncommon as a monotherapy, but possible in combination with fasting protocols or very low carbohydrate diets.
Who Should Not Take Metformin for Longevity
- eGFR below 30 mL/min
- Active liver disease
- History of lactic acidosis
- Heavy alcohol use (>3 drinks/day)
- Planned radiologic contrast procedures (hold 48 hours)
The Bottom Line
Metformin for longevity is a reasonable consideration for non-diabetics, particularly those over 40 with metabolic risk factors who are not already achieving robust AMPK activation through exercise and caloric restriction. The dosing sweet spot for off-label longevity use is 1,000-1,500 mg/day of extended-release, taken with dinner, with careful titration over 4-6 weeks. Monitor B12, kidney function, and — if you exercise seriously — aerobic capacity. The TAME trial will provide the definitive answer. Until then, this is a informed-consent decision between physician and patient, not a universal recommendation.
References
-
Bannister CA, Holden SE, Jenkins-Jones S, et al. Can people with type 2 diabetes live longer than those without? A comparison of mortality in people initiated with metformin or sulphonylurea monotherapy and matched, non-diabetic controls. Diabetes, Obesity and Metabolism. 2014;16(11):1165-1173. doi:10.1111/dom.12354
-
Kulkarni AS, Brutsaert EF, Anghel V, et al. Metformin regulates metabolic and nonmetabolic pathways in skeletal muscle and subcutaneous adipose tissues of older adults. Aging Cell. 2018;17(2):e12723. doi:10.1111/acel.12723
-
Konopka AR, Laurin JL, Schoenberg HM, et al. Metformin inhibits mitochondrial adaptations to aerobic exercise training in older adults. Aging Cell. 2019;18(1):e12880. doi:10.1111/acel.12880
-
Barzilai N, Crandall JP, Kritchevsky SB, Espeland MA. Metformin as a tool to target aging. Cell Metabolism. 2016;23(6):1060-1065. doi:10.1016/j.cmet.2016.05.011