Babesia stands apart from other tick-borne co-infections for a fundamental reason: it is not a bacterium. It is a protozoan parasite — a relative of the Plasmodium species that causes malaria. It infects and destroys red blood cells. And because it is a parasite, not a bacterium, the antibiotics used to treat Lyme disease and other bacterial co-infections have no effect on it.
This distinction has enormous clinical implications. A patient with undiagnosed Babesia who receives even the most comprehensive antibiotic protocol for Lyme disease will not improve fully — because the parasite requires entirely different treatment.
Species and Distribution
Several Babesia species infect humans:

- Babesia microti: The most common species in the northeastern and upper midwestern United States
- Babesia divergens and Babesia venatorum: More common in Europe
- Babesia duncani: Found in the western United States
- Other species continue to be identified
In my practice in Germany, B. divergens and related European species are the primary concern, though I see patients from around the world carrying various species.
Clinical Presentation
Babesia produces a symptom pattern that overlaps with other tick-borne infections but has several distinguishing features:
Air hunger is the most characteristic symptom. Patients describe a sensation of not being able to get a satisfying breath — sighing respirations, the feeling of needing to yawn or take a deep breath constantly. When a chronically ill patient with tick exposure describes air hunger, Babesia should be at the top of the differential [1].
Night sweats — drenching, not mild perspiration — are common and often cyclical, reflecting the parasite’s replication cycle in red blood cells.
Cyclical fevers or chills — again reflecting the parasitic lifecycle. Some patients notice symptom patterns on roughly 48-72 hour cycles.
Severe fatigue disproportionate to other findings.
Headache — often severe and resistant to standard analgesics.
Hemolytic anemia in more severe cases, with elevated LDH, low haptoglobin, and elevated indirect bilirubin.
Diagnostic Challenges
Standard testing for Babesia has significant limitations:
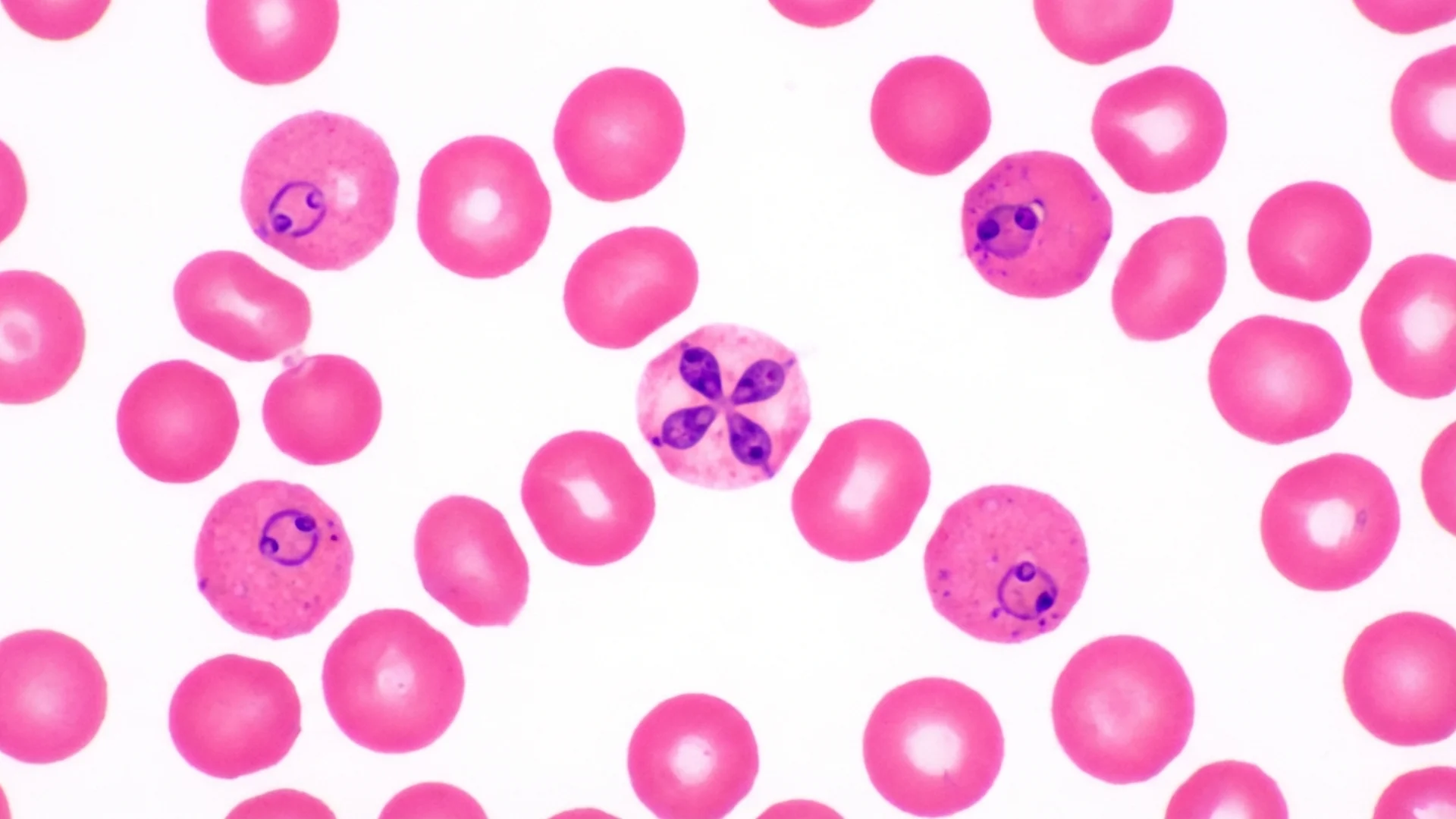
Blood smear: The classic diagnostic method. A skilled technician examining a Giemsa-stained thin smear can identify the characteristic ring forms or “Maltese cross” (tetrad) formations within red blood cells. However, in chronic, low-level infection, parasitemia may be too low to detect on routine smear.
Serology (IFA): Detects antibodies against specific Babesia species. Limitations include species-specificity (testing for B. microti will miss B. duncani) and false negatives in immunocompromised patients.
PCR: More sensitive than smear for low-level parasitemia. Species-specific, so the correct PCR must be ordered.
I recommend testing for multiple Babesia species and using more than one modality when clinical suspicion is high. A negative blood smear does not exclude Babesia.
Treatment
First-Line
The standard treatment for Babesia is:
- Atovaquone plus azithromycin — the most commonly used combination, generally well-tolerated
- Quinine plus clindamycin — more potent but associated with significant side effects (tinnitus, GI disturbance); reserved for severe cases or atovaquone failures
Treatment Duration
Acute babesiosis in immunocompetent patients may respond to 7-10 days of treatment. Chronic babesiosis, particularly in immunocompromised patients or those with co-infections, often requires extended treatment — weeks to months.
In my experience, relapse is common if treatment is too short. I monitor with serial PCR and/or blood smears when available, and I continue treatment until clinical improvement is sustained and laboratory markers normalize.
Supportive Care
Because Babesia destroys red blood cells, patients may develop significant anemia. Supporting red blood cell production (iron, B12, folate as indicated), managing the inflammatory burden from hemolysis, and monitoring for complications (splenic enlargement, renal impairment from hemoglobin deposition) are important components of care.
In severe, refractory cases — particularly in immunocompromised patients — red blood cell exchange transfusion or apheresis may be considered.
The Interaction with Lyme Disease
Babesia and Borrelia co-infection is worse than either alone. Studies have shown that co-infected patients have more severe symptoms, longer duration of illness, and more complications than patients with either infection alone [2].
The mechanism is partially understood: Babesia’s immune-modulating effects impair the host’s ability to control Borrelia, and vice versa. Each pathogen benefits from the immune suppression caused by the other.
This is why comprehensive co-infection testing at the outset of treatment is essential, not optional. Identifying and treating Babesia early in the course of a Lyme treatment program significantly improves outcomes.
The Bottom Line
Babesia is a parasite, not a bacterium. It requires antiparasitic treatment, not antibiotics. Its hallmark symptoms — air hunger, drenching night sweats, cyclical fevers, and severe fatigue — should prompt specific testing even if other tick-borne infections have already been identified. Missing Babesia is one of the most common reasons that tick-borne disease treatment programs fail.
References
- Krause PJ. Human babesiosis. International Journal for Parasitology. 2019;49(2):165-174.
- Diuk-Wasser MA, et al. Coinfection by Ixodes Tick-Borne Pathogens: Ecological, Epidemiological, and Clinical Consequences. Trends in Parasitology. 2016;32(1):30-42.
This content is educational and does not constitute medical advice. Babesia testing and treatment require medical supervision by a physician experienced in tick-borne parasitic infections.