The thymus is perhaps the most underappreciated organ in medicine. Located behind the sternum, this small gland is responsible for one of the most critical processes in immunology: the maturation and selection of T cells. Without a functioning thymus, the adaptive immune system cannot develop properly. And yet, the thymus begins shrinking after puberty and is largely replaced by fatty tissue by middle age.
This involution is not without consequence. It is one of the primary mechanisms behind the age-related decline in immune function known as immunosenescence — and it is an area where integrative medicine may have something meaningful to offer.
What the Thymus Does
The thymus receives immature T cell precursors from the bone marrow and subjects them to a rigorous selection process that takes approximately three weeks.
Positive selection occurs in the thymic cortex. T cells that can recognize self-MHC molecules (the proteins that present antigens on cell surfaces) are selected to survive. Those that cannot recognize MHC molecules are eliminated — they would be useless, unable to interact with the antigen presentation system.
Negative selection occurs in the thymic medulla. T cells that react too strongly to self-antigens presented by medullary epithelial cells and dendritic cells are eliminated through apoptosis. These are the cells that, if released, would attack the body’s own tissues and cause autoimmune disease.
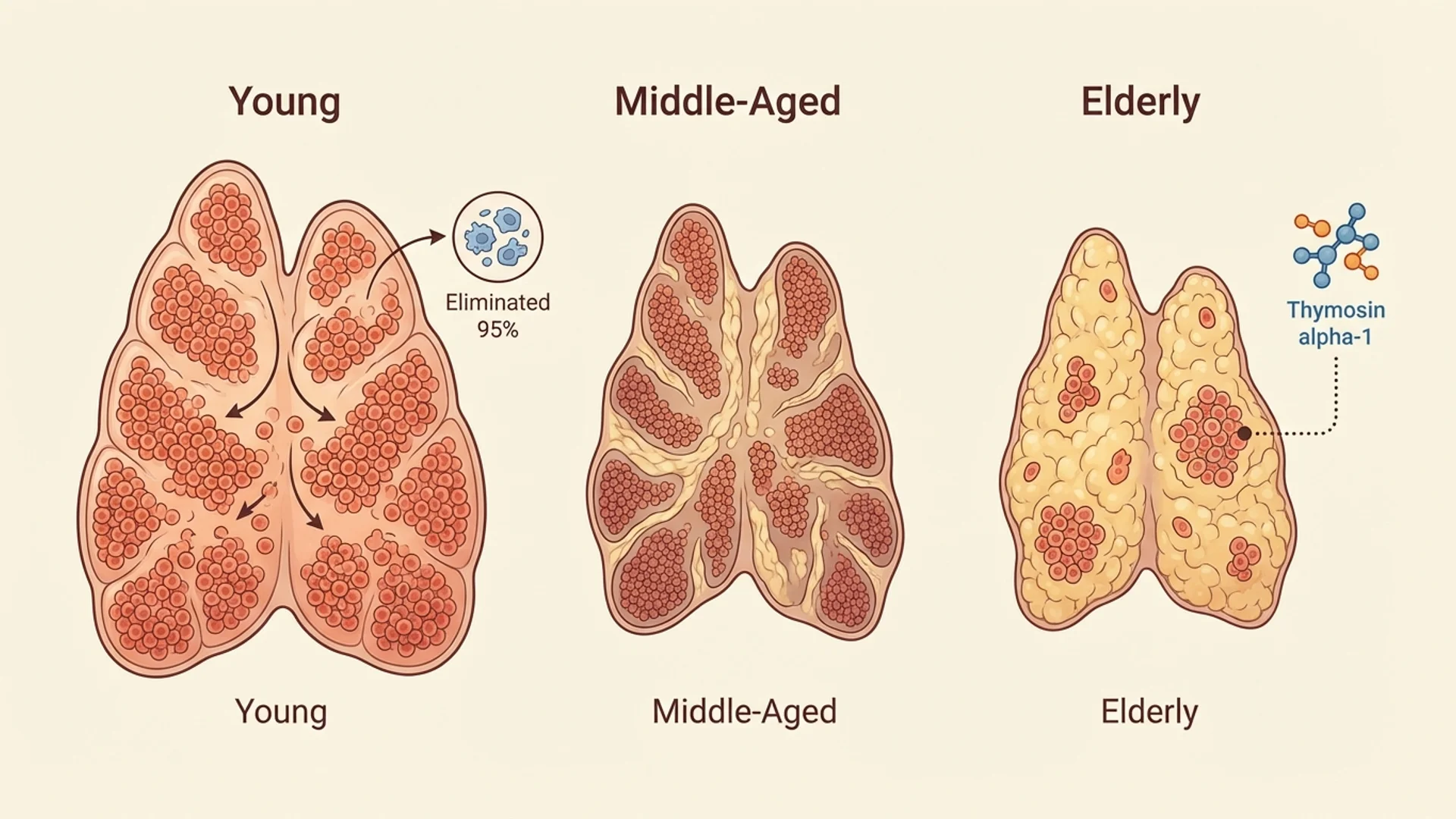
The process is extraordinarily rigorous. Approximately 95% of T cells that enter the thymus are eliminated. Only those that pass both positive and negative selection are released into the periphery as mature, functional, self-tolerant T cells.
The thymus also produces several peptide hormones — thymulin, thymosin alpha-1, thymopoietin, and thymic humoral factor — that influence T cell maturation and function both within the thymus and systemically.
Thymic Involution: The Age Problem
Thymic involution begins after puberty. By age 40, the thymus has lost approximately 80% of its functional tissue. By age 70, less than 5% of functional thymic tissue remains.
The consequences are measurable:
- Declining naive T cell production — the output of new, diverse T cells decreases with each decade. The T cell repertoire becomes increasingly composed of memory T cells from prior encounters, reducing the system’s ability to respond to novel threats.
- Reduced T cell receptor diversity — fewer naive T cells means less combinatorial diversity. The immune system becomes less flexible.
- Accumulation of senescent T cells — older T cells that have undergone multiple rounds of division become senescent, expressing markers like CD28- and KLRG1+. These cells are not dead, but they are functionally impaired — they occupy immunological space without contributing effective responses.
- Impaired immune surveillance — the combination of reduced diversity and accumulated senescence contributes to increased susceptibility to infections, reduced vaccine efficacy, and impaired cancer surveillance in older adults.
This is not merely theoretical. The age-related decline in immune function has documented clinical consequences: older adults have higher rates of pneumonia, influenza complications, herpes zoster reactivation, and cancer — all of which correlate with measurable changes in T cell compartment composition.
Thymic Peptides: Clinical Application
Thymosin Alpha-1 (Ta1)
Thymosin alpha-1 is the most extensively studied thymic peptide. It is a 28-amino acid peptide originally isolated from thymic tissue and now produced synthetically. It is approved as a pharmaceutical agent (under the trade name Zadaxin) in over 30 countries.
Mechanism: Ta1 enhances immune function through multiple pathways — it promotes dendritic cell maturation, increases TLR expression on innate immune cells, enhances NK cell activity, supports Th1 responses, and promotes regulatory T cell function. It acts as an immune modulator rather than a simple immunostimulant.
Clinical evidence:
- Hepatitis B: Multiple RCTs demonstrate improved viral clearance and seroconversion rates when Ta1 is added to standard antiviral therapy.
- Hepatitis C: RCTs show improved sustained virological response rates.
- Vaccine adjuvant: RCTs demonstrate enhanced antibody responses to influenza and hepatitis B vaccines in immunocompromised patients.
- Sepsis: A meta-analysis of RCTs (Wu et al., 2015) found reduced mortality with Ta1 as adjunctive therapy in severe sepsis.
- Cancer: Clinical studies suggest immune-enhancing effects when combined with chemotherapy, though the evidence for direct anti-tumor effects in humans requires further controlled trials.
Evidence level: RCTs for hepatitis and vaccine adjuvant applications; meta-analyses for sepsis; controlled studies and clinical observation for broader use.
Thymulin
Thymulin (formerly known as FTS — facteur thymique serique) is a zinc-dependent peptide produced exclusively by thymic epithelial cells. Its levels decline with age in parallel with thymic involution. Thymulin promotes T cell differentiation and modulates cytokine production.
Evidence level: primarily preclinical and early clinical studies. The research is less extensive than for thymosin alpha-1.
Thymic Peptide Therapy in Practice
In my clinical practice, thymic peptide therapy — primarily thymosin alpha-1 — is used in several contexts:
- Chronic infections (Lyme disease, EBV, hepatitis) where immune exhaustion is documented by functional testing
- Post-COVID patients with persistent immune dysregulation
- Oncology patients as supportive therapy alongside conventional treatment
- Age-related immune decline in patients with documented reduction in naive T cell populations or NK cell function
The therapy is typically administered subcutaneously, two to three times per week for four to eight weeks, then reassessed based on clinical and laboratory response.
I want to be transparent about the limitations: thymic peptide therapy does not reverse thymic involution. It does not regenerate thymic tissue. What it appears to do — based on both clinical trial data and my clinical observation — is partially compensate for reduced thymic function by enhancing the maturation and activation of existing T cell populations and supporting dendritic cell function.
Supporting Thymic Health
Beyond thymic peptides, several factors influence T cell health and thymic function:
Zinc is essential for thymulin activity (thymulin requires zinc for its biological function) and T cell maturation. Zinc deficiency, common in older adults, directly impairs thymic function. Supplementation at 15-30 mg daily is a reasonable baseline.
Vitamin A supports thymic epithelial cell function and T cell differentiation. Deficiency is associated with thymic atrophy in animal models.
Exercise has been shown to promote thymic output of naive T cells, even in older adults (Duggal et al., Aging Cell 2018). Lifelong exercisers maintain higher thymic output than sedentary controls — a significant finding suggesting that physical activity can partially counteract thymic involution.
Caloric restriction preserves thymic function in animal models, likely through reduced chronic inflammation and oxidative stress on thymic epithelial cells. The relevance to humans is supported by observational data but not yet confirmed by controlled trials.
The Broader Context
Thymic involution and the resulting decline in immune competence is one of the core mechanisms of aging. It connects to cancer risk, infection susceptibility, vaccine efficacy, and autoimmune disease. Strategies to maintain or partially restore T cell compartment health — through thymic peptides, lifestyle interventions, nutritional support, and potentially future regenerative approaches — represent one of the more promising areas in longevity medicine.
The nuance matters here. We cannot currently reverse thymic involution. But we can support the immune system’s ability to function effectively despite it, and the evidence base for doing so is growing.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical care. Thymic peptide therapy requires physician supervision and is prescribed based on individual clinical assessment.