Natural killer cells occupy a unique position in the immune system. They are innate immune cells — they do not require prior sensitization to recognize and destroy their targets. Yet they perform a function that seems almost adaptive in its sophistication: they patrol the body identifying cells that have lost normal surface markers, a hallmark of viral infection and malignant transformation.
NK cells are your immune system’s surveillance force. When they function well, virus-infected cells and early cancer cells are eliminated before they can establish themselves. When NK cell function is impaired, these threats gain a foothold.
How NK Cells Work
NK cells identify their targets through a balance of activating and inhibitory signals. Healthy cells display MHC class I molecules on their surface, which engage inhibitory receptors on NK cells and prevent attack. When a cell loses MHC class I expression — as commonly occurs during viral infection or malignant transformation — the inhibitory signal is absent, and the NK cell is activated to kill.
This “missing self” recognition is elegant. Viruses often downregulate MHC class I to avoid detection by cytotoxic T cells, but in doing so, they expose the infected cell to NK cell killing. Cancer cells similarly may lose MHC expression as they accumulate mutations, rendering them visible to NK cells.
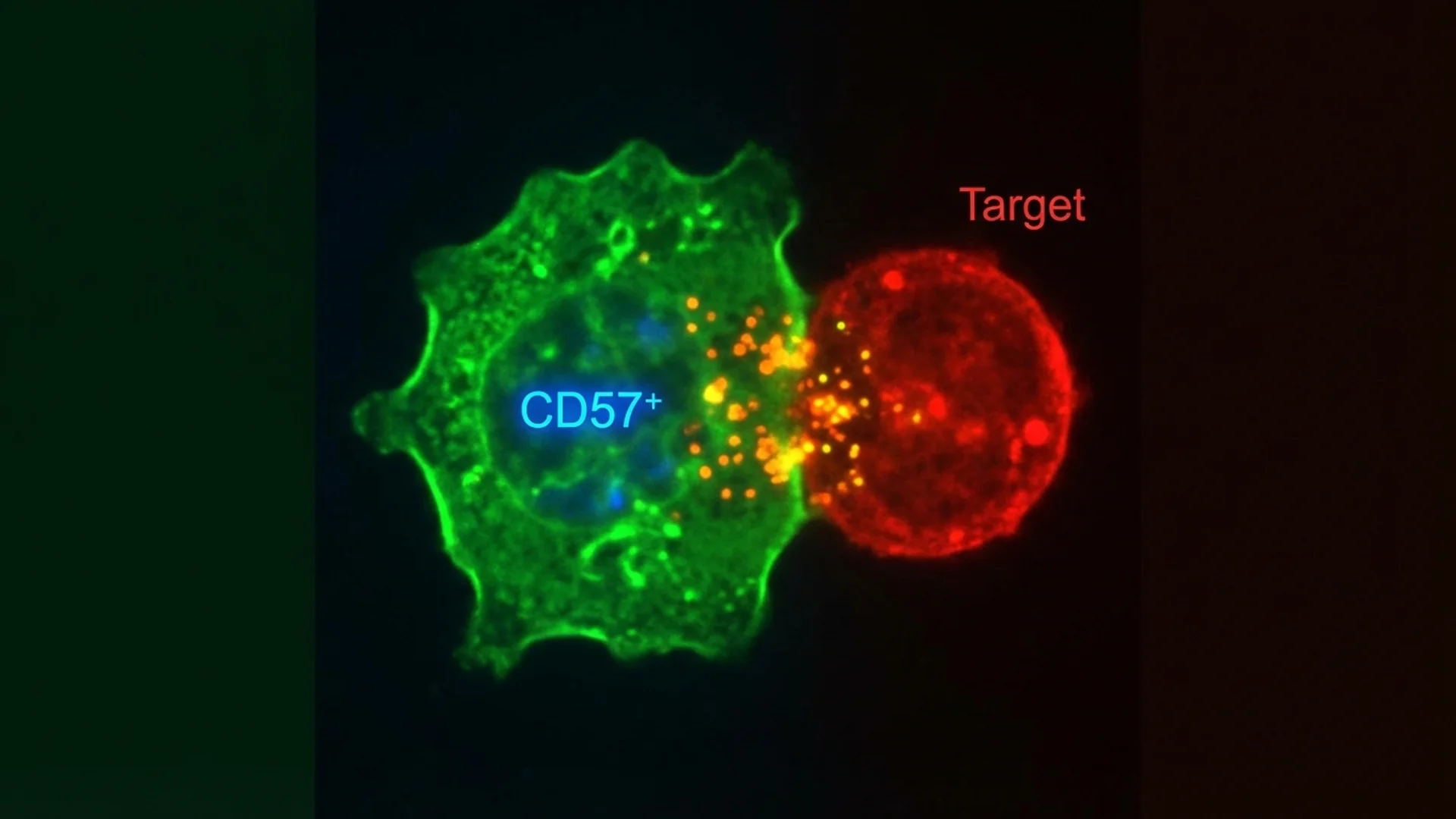
NK cells kill through two primary mechanisms:
- Perforin/granzyme pathway — NK cells release perforin (which forms pores in the target cell membrane) and granzymes (serine proteases that enter through the pores and trigger apoptosis)
- Death receptor ligation — NK cells express FasL and TRAIL, which engage death receptors on target cells and trigger programmed cell death
Beyond direct killing, NK cells produce cytokines — particularly interferon-gamma (IFN-gamma) and TNF-alpha — that activate macrophages and shape the adaptive immune response. NK cells are not just killers; they are coordinators.
CD57: A Marker with Clinical Significance
CD57 is a surface glycoprotein expressed on a subset of NK cells and T cells. In immunology, CD57 marks cells that have undergone extensive replication and are approaching replicative senescence — they are highly differentiated but have limited proliferative capacity.
CD57 in Lyme Disease
Dr. Raphael Stricker proposed the use of CD57+ NK cell counts as a marker for chronic Lyme disease activity. The observation is that patients with active chronic Lyme disease tend to have low CD57+ NK cell counts (below 60 cells/microL), and that successful treatment is often accompanied by normalization of CD57 levels.
Here is what I want to be transparent about: the CD57-Lyme connection is clinically observed and used by Lyme-literate physicians worldwide, but it remains debated in mainstream infectious disease circles. The original observations have not been replicated in large, multicenter controlled trials. The marker is also not specific to Lyme disease — low CD57 can be seen in other chronic infections and immunodeficiency states.
In my clinical experience, CD57 is a useful but imperfect marker. I use it as one data point among many — alongside clinical symptoms, ELISpot testing, inflammatory markers, and treatment response. I do not diagnose or rule out Lyme disease based on CD57 alone, and any physician who does is overinterpreting a single marker.
Evidence level: clinical observation, case series, and retrospective studies. Further controlled research is warranted.
CD57 in Aging and Cancer
High CD57 expression on T cells is a well-established marker of immunosenescence. Accumulation of CD57+CD28- T cells is associated with reduced immune competence, poor vaccine responses, and increased infection risk in older adults. This is supported by controlled human studies.
In oncology, NK cell activity — including the CD57+ subset — correlates with prognosis in several cancers. Patients with higher NK cell activity tend to have better outcomes. This observation has driven research into NK cell-based immunotherapies, some of which have reached clinical trials.
Assessing NK Cell Function
There is an important distinction between NK cell counts and NK cell function. A patient can have normal NK cell numbers but impaired NK cell activity — the cells are present but not performing optimally.
NK cell count (from a standard lymphocyte subset panel) tells you how many NK cells are circulating. It is a starting point.
NK cell cytotoxicity assay measures the actual killing capacity of NK cells against standardized target cells (typically K562 leukemia cells). This functional assessment provides more clinically relevant information than counts alone.
CD57+ NK cell count provides information about the differentiation state of the NK cell population and, in the context of suspected Lyme disease, may offer additional diagnostic data.
In my practice, I order NK cell functional assessments for patients with:
- Chronic infections (Lyme disease, EBV, recurrent viral infections)
- Cancer (baseline assessment and treatment monitoring)
- Immunosenescence (patients with recurrent infections or poor vaccine responses)
- Chronic fatigue (particularly when infection-related causes are suspected)
Supporting NK Cell Function
Several interventions have documented effects on NK cell activity:
Sleep is the single most impactful lifestyle factor. A single night of sleep deprivation reduces NK cell activity by approximately 70% (Irwin et al., Psychosomatic Medicine). This is not a subtle effect. It is profound and rapid. Evidence level: controlled human studies.
Exercise at moderate intensity increases circulating NK cell numbers and activity. The effect is acute (measurable within hours of exercise) and chronic (regular exercisers have higher baseline NK cell activity). Evidence level: RCTs and controlled studies.
Stress reduction — chronic psychological stress suppresses NK cell activity through cortisol-mediated mechanisms. Interventions that reduce stress hormones (meditation, mindfulness, adequate social connection) improve NK cell function. Evidence level: controlled human studies.
Medicinal mushrooms — beta-glucans from mushrooms such as Lentinula edodes (shiitake), Grifola frondosa (maitake), and Trametes versicolor (turkey tail) enhance NK cell activity through innate immune activation. The evidence base includes multiple controlled studies and some RCTs, particularly for lentinan (shiitake-derived beta-glucan) as an oncology adjunct in Japan.
Selenium supplementation (200 mcg daily) has been shown to enhance NK cell activity in multiple controlled studies. Selenium is essential for selenoprotein production, which supports both innate and adaptive immune function.
Vitamin C at moderate to high doses supports NK cell function. The IV vitamin C literature in oncology partially relies on NK cell activation as one mechanism of action.
Cold exposure — cold water immersion increases circulating NK cell numbers, likely through norepinephrine-mediated mobilization. The evidence is from controlled studies; the long-term clinical significance of acute NK cell mobilization through cold exposure is still being characterized.
Clinical Perspective
NK cells are one of the immune system’s most important first-line defenses, and yet they are rarely assessed in standard medical practice. A complete blood count tells you nothing about NK cell function. Even a basic lymphocyte subset panel provides only counts, not functional data.
For patients with chronic infections, recurrent cancers, or unexplained immune dysfunction, NK cell assessment should be a routine part of the diagnostic workup. The information it provides often changes the treatment approach.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical care. Consult a qualified physician for immune assessment and treatment decisions.