If I could convey a single concept to every patient I see, it would be this: the gut is not just a digestive organ. It is the largest immune organ in the body, and its health — or dysfunction — ripples into virtually every other system.
This is not a fringe idea. It is established immunology. Approximately 70% of the body’s immune tissue resides in the gut-associated lymphoid tissue (GALT), which includes Peyer’s patches, isolated lymphoid follicles, mesenteric lymph nodes, and the vast network of immune cells distributed throughout the intestinal mucosa.
The question for clinicians is no longer whether the gut influences immunity. The question is how to use this understanding to improve patient outcomes.
Architecture of Gut Immunity
The gastrointestinal tract presents a unique immunological challenge. It must simultaneously absorb nutrients (requiring permeability), tolerate commensal bacteria and food antigens (requiring restraint), and defend against pathogens (requiring aggression). Getting this balance right is the central task of gut immunity.
The Epithelial Barrier
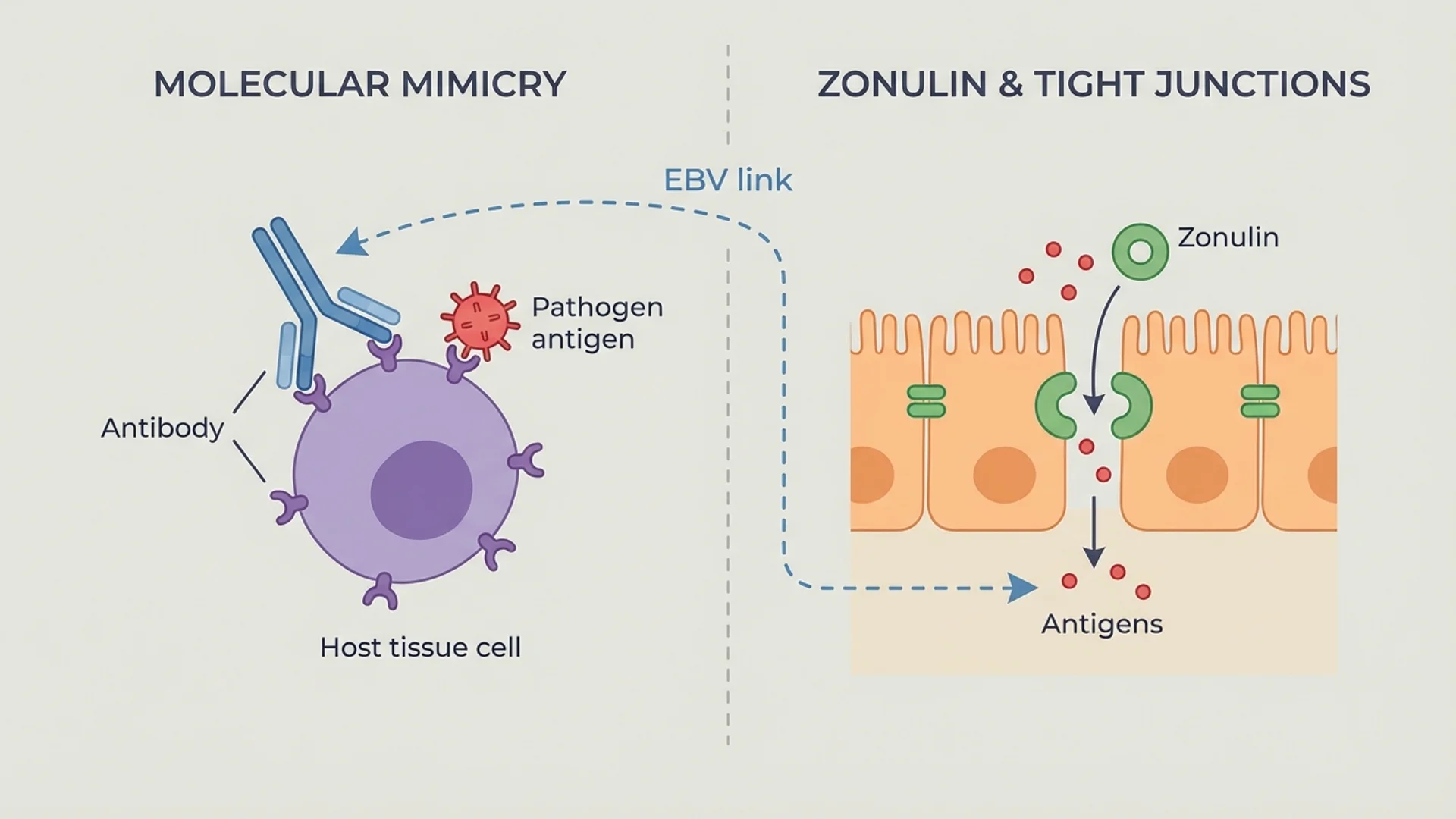
The intestinal epithelium is a single-cell-thick layer that separates the body’s internal environment from the contents of the gut lumen. Tight junction proteins (claudins, occludin, zonula occludens) seal the spaces between epithelial cells, creating a selective barrier that allows nutrient absorption while preventing the passage of bacteria and large molecules.
When tight junction integrity is compromised — through infection, inflammation, dietary triggers (gluten-mediated zonulin release), or toxic exposures — the result is increased intestinal permeability. This allows bacterial products, particularly lipopolysaccharide (LPS), to enter the bloodstream, where they activate immune cells through toll-like receptor 4 (TLR4) signaling.
This process — endotoxemia from gut barrier dysfunction — is now recognized as a contributing factor in conditions ranging from metabolic syndrome to autoimmune disease to neuroinflammation. The evidence is Level 1 for the mechanism; the clinical application is still being refined.
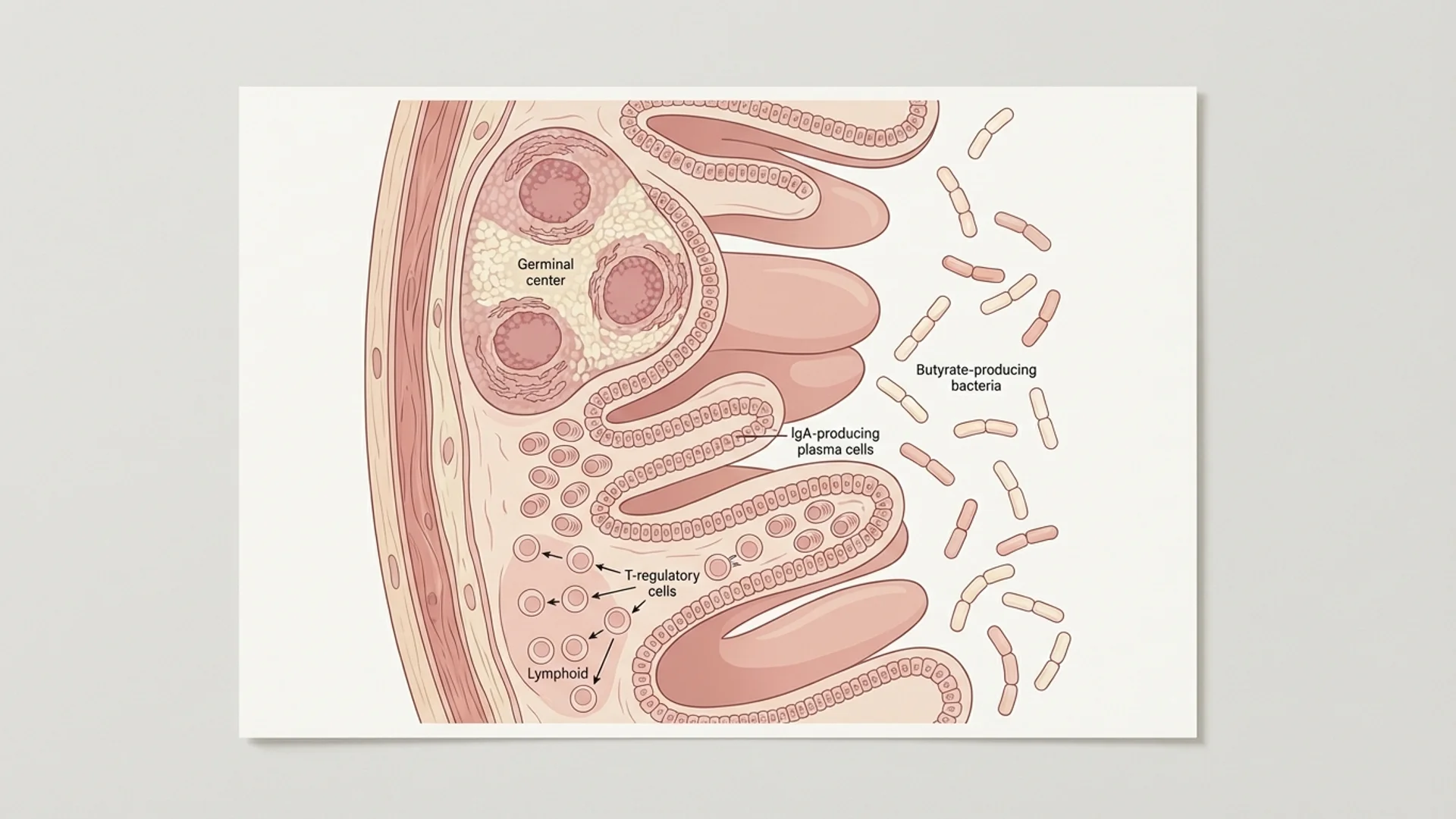
The GALT
The gut-associated lymphoid tissue includes:
- Peyer’s patches — organized lymphoid structures in the small intestine that sample luminal antigens through specialized M cells and initiate adaptive immune responses
- Isolated lymphoid follicles — smaller lymphoid aggregates distributed throughout the intestinal wall
- Mesenteric lymph nodes — where dendritic cells that have sampled gut antigens present them to T and B cells
- Lamina propria lymphocytes — immune cells distributed throughout the connective tissue beneath the epithelium, including T cells, B cells, macrophages, and dendritic cells
- Intraepithelial lymphocytes — specialized T cells that reside within the epithelial layer itself, serving as a first line of adaptive defense
Secretory IgA
The gut produces more antibody than any other organ — primarily secretory IgA (sIgA), which is released into the gut lumen where it binds to pathogens and toxins, preventing their adherence to the epithelium. Adequate sIgA production is essential for mucosal defense, and low sIgA levels are associated with increased susceptibility to gut infections and food sensitivities.
sIgA can be measured through stool testing and is one of the markers I assess routinely in patients with chronic gut or immune issues.
The Microbiome-Immune Dialogue
The relationship between gut bacteria and the immune system is not one of tolerance despite the bacteria’s presence. It is active, bidirectional, and essential for normal immune development.
The microbiome educates the immune system. Germ-free animals (raised without any bacteria) have profoundly abnormal immune systems — underdeveloped GALT, deficient IgA production, and impaired T cell function. Colonization with normal gut bacteria restores these functions. In humans, early microbial exposure — particularly through vaginal delivery, breastfeeding, and diverse environmental contact — is critical for proper immune maturation.
The microbiome produces immune-active metabolites. Short-chain fatty acids (butyrate, propionate, acetate), produced by bacterial fermentation of dietary fiber, directly modulate immune function. Butyrate, in particular, promotes regulatory T cell differentiation and suppresses pro-inflammatory NF-kB signaling. This is one of the strongest mechanistic links between diet, gut bacteria, and immune regulation.
The immune system shapes the microbiome. sIgA selectively promotes beneficial species and suppresses pathogenic ones. Antimicrobial peptides (defensins) produced by epithelial cells maintain microbial balance. When immune function is impaired, dysbiosis often follows.
Clinical Implications
Autoimmune Disease
The connection between gut barrier dysfunction and autoimmune disease is supported by a substantial body of evidence. Increased intestinal permeability has been documented before the clinical onset of type 1 diabetes, celiac disease, and rheumatoid arthritis.
In my clinical experience, addressing gut health is one of the most impactful interventions for autoimmune patients. Not every patient responds, and gut repair is not a substitute for appropriate medical management. But for many patients, restoring gut barrier integrity and microbial balance produces measurable improvements in inflammatory markers and symptoms.
Chronic Infection
Patients with chronic infections — Lyme disease, EBV reactivation, recurrent Candida — frequently have concurrent gut dysfunction. The relationship is likely bidirectional: chronic infection impairs gut integrity, and impaired gut integrity weakens the immune response to infection. Addressing both simultaneously produces better outcomes than addressing either alone.
Mental Health
The gut-brain axis — mediated through the vagus nerve, microbial metabolites, and immune signaling — is one of the most active areas of current research. Gut dysbiosis has been associated with depression, anxiety, and cognitive dysfunction through mechanisms that include altered serotonin metabolism (approximately 90% of serotonin is produced in the gut), inflammatory cytokine signaling, and direct vagal afferent stimulation.
This is an area where our understanding is still developing. The associations are strong, and the mechanistic pathways are plausible and increasingly well-characterized. But we are not yet at the point of reliably treating psychiatric conditions through gut interventions alone.
Practical Recommendations
For patients who want to support gut-immune health, here is what the evidence supports:
- Dietary diversity — aim for 30+ different plant foods per week. The American Gut Project data shows that microbial diversity correlates directly with plant food diversity.
- Adequate fiber — 25-35 g daily from whole food sources. Fiber is the substrate for short-chain fatty acid production, which is the primary metabolic link between diet and immune regulation.
- Fermented foods — regular consumption of sauerkraut, kimchi, kefir, and other fermented foods increases microbial diversity (Stanford study, Sonnenburg et al., Cell 2021).
- Minimize gut barrier disruptors — excessive alcohol, NSAIDs, glyphosate-exposed foods, emulsifiers (polysorbate 80, carboxymethylcellulose), and chronic stress all impair tight junction integrity.
- Consider targeted supplementation — L-glutamine (5 g daily), zinc carnosine, and butyrate support gut barrier function with moderate to strong evidence.
The gut-immune axis is not a trend. It is a fundamental biological reality that should inform how we think about immune health, chronic disease, and treatment strategy.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical care. Consult a qualified physician before making decisions about your health.