Autoimmune disease is not a single condition. It is a category of over 80 distinct diseases that share a common mechanism: the immune system loses the ability to distinguish self from non-self and begins attacking the body’s own tissues.
The scope of the problem is significant. Autoimmune diseases collectively affect approximately 5-8% of the population in Western countries, with prevalence increasing. They are the third leading cause of morbidity in industrialized nations, and their rise over the past 50 years — far too rapid to be explained by genetic change alone — points clearly to environmental drivers.
How Tolerance Works — and Fails
Under normal conditions, the immune system maintains tolerance to self-antigens through several mechanisms:
Central tolerance occurs in the thymus (for T cells) and bone marrow (for B cells). During development, immune cells that react strongly to self-antigens are eliminated through apoptosis. This process is effective but not perfect — some self-reactive cells inevitably escape into the periphery.
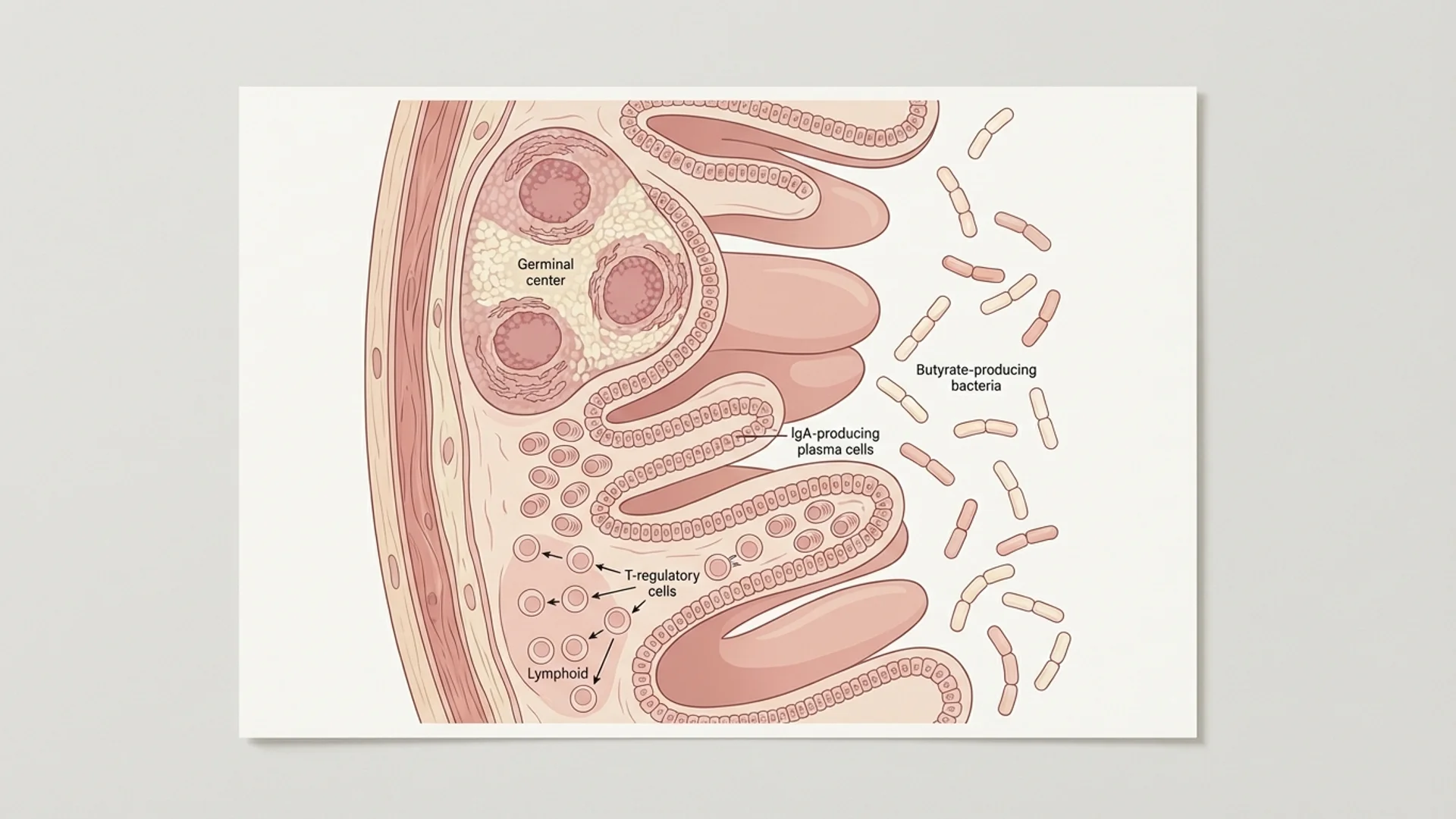
Peripheral tolerance provides a second line of defense. Regulatory T cells (Tregs) actively suppress self-reactive immune cells that escaped central selection. Anergy (functional inactivation) renders other self-reactive cells unresponsive. Immune privilege protects certain tissues (the eye, brain, testes) by limiting immune cell access.
Autoimmune disease develops when these tolerance mechanisms fail. The question is: why do they fail?
Triggers and Mechanisms
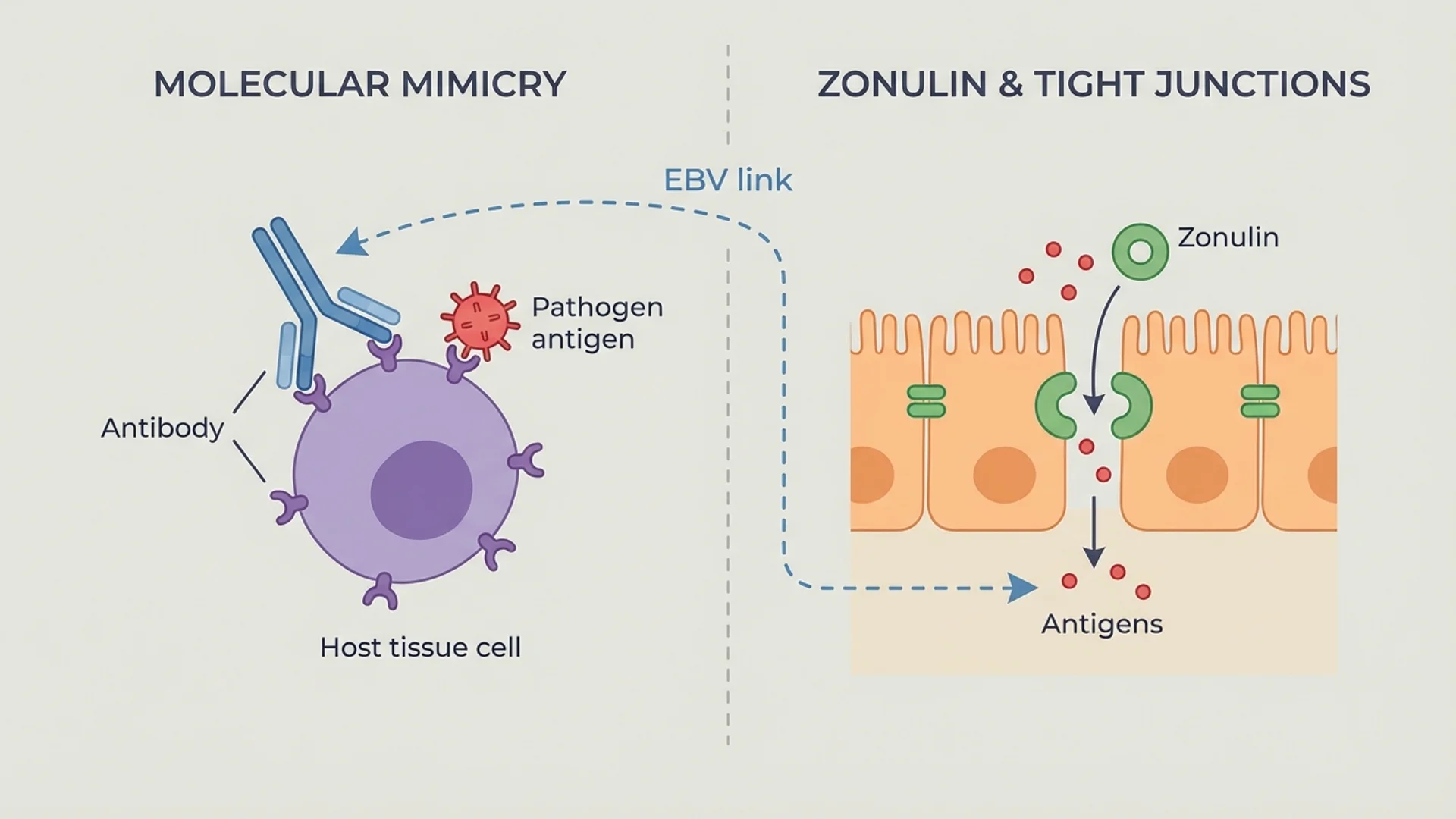
Molecular Mimicry
This is the most studied mechanism. When a pathogen carries proteins that structurally resemble host proteins, the immune response generated against the pathogen can cross-react with the host tissue. The classic example is rheumatic fever, where antibodies against streptococcal M protein cross-react with cardiac myosin.
Molecular mimicry has been implicated in multiple sclerosis (viral triggers), type 1 diabetes (coxsackievirus B), and Guillain-Barre syndrome (Campylobacter jejuni). The evidence varies from strong to suggestive depending on the specific disease.
Intestinal Permeability
The role of gut barrier dysfunction in autoimmune disease has moved from fringe theory to established science, largely through the work of Alessio Fasano and others on zonulin-mediated intestinal permeability. The mechanism is straightforward: when the gut barrier is compromised, bacterial products (particularly LPS) and partially digested food proteins enter the bloodstream, where they trigger immune activation and potentially cross-react with self-antigens.
Increased intestinal permeability has been documented in type 1 diabetes, celiac disease, rheumatoid arthritis, ankylosing spondylitis, and inflammatory bowel disease — often preceding the clinical onset of disease. Evidence level: controlled human studies (multiple).
Chronic Infection
Certain chronic infections are associated with specific autoimmune diseases. Epstein-Barr virus (EBV) has the strongest epidemiological link to multiple sclerosis — the relative risk of MS after EBV infection is approximately 32-fold. The mechanisms are still being characterized but likely involve molecular mimicry, bystander activation, and epitope spreading.
In my clinical experience, I have seen autoimmune conditions improve significantly when underlying chronic infections — including Lyme disease, EBV reactivation, and Chlamydia pneumoniae — are identified and treated. This is not a universal finding, and it does not apply to every patient. But it is a clinical observation I take seriously.
Environmental Toxins
Heavy metals, pesticides, solvents, and other environmental chemicals have documented immunomodulatory effects. Mercury, in particular, has been shown to trigger autoimmune responses in genetically susceptible animal models. The relevance to human autoimmune disease is supported by epidemiological associations but establishing causation remains challenging.
The Genetic Component
Genetics load the gun; environment pulls the trigger. The strongest genetic risk factor for autoimmune disease is the HLA (human leukocyte antigen) complex — the genes that encode the MHC molecules responsible for presenting antigens to T cells. Specific HLA variants are strongly associated with specific autoimmune diseases (HLA-B27 with ankylosing spondylitis, HLA-DR4 with rheumatoid arthritis).
But genetics alone are insufficient. Concordance rates for autoimmune diseases in identical twins are typically 25-50%, meaning that even with identical DNA, more than half the time only one twin develops the disease. The environment is decisive.
The Integrative Approach
Conventional treatment for autoimmune disease relies primarily on immunosuppression — corticosteroids, DMARDs, biologics. These drugs can be life-saving and are often necessary. But they address the downstream immune response without addressing the upstream triggers, and they carry significant long-term side effects.
The integrative approach does not reject conventional treatment. It asks additional questions: What is driving the immune dysregulation? Can we address root causes while managing symptoms? Can we support immune regulation rather than only suppressing immune function?
What This Looks Like in Practice
-
Identify and address gut barrier dysfunction. Comprehensive stool analysis, intestinal permeability assessment, and a systematic gut repair protocol. In my clinical experience, this is the single most impactful intervention for many autoimmune patients.
-
Screen for and treat chronic infections. Comprehensive testing for EBV, Lyme disease, Chlamydia pneumoniae, and other chronic infections using sensitive methodology (ELISpot, PCR, not just standard serology).
-
Assess environmental toxin burden. Heavy metal testing and environmental toxicology screening. Targeted detoxification support when elevated levels are found.
-
Nutritional optimization. Vitamin D optimization (target 40-60 ng/mL), omega-3 fatty acid supplementation (2-4 g EPA/DHA daily), zinc and selenium assessment, and an anti-inflammatory dietary pattern. The evidence for vitamin D in autoimmune risk reduction is supported by randomized controlled trials (VITAL trial, Hahn et al.).
-
Immune modulation therapies. Low-dose naltrexone (LDN) has accumulating clinical trial evidence for several autoimmune conditions. IV vitamin C at moderate doses has anti-inflammatory effects. Mistletoe extract (Viscum album) modulates cytokine balance. These are used as adjuncts, not replacements, for conventional treatment.
-
Regulatory T cell support. Short-chain fatty acids (butyrate), vitamin A, vitamin D, and TGF-beta-supporting nutrients help maintain Treg function. A healthy gut microbiome is the most important source of short-chain fatty acids.
The Honest Perspective
I want to be direct about what integrative medicine can and cannot do for autoimmune disease.
It cannot cure autoimmune disease. No approach — conventional or integrative — reliably cures established autoimmune disease once it has progressed beyond early stages.
What it can do is reduce inflammatory burden, identify and address contributing factors, and in many cases, reduce the level of immunosuppressive medication required. Some patients achieve sustained remission. Others achieve better symptom control with fewer side effects. Neither outcome is guaranteed, and individual variation plays a significant role.
What I tell my patients: autoimmune disease is multifactorial, and the most effective treatment is also multifactorial. Addressing gut health, infection, toxin burden, nutrition, and stress is not a substitute for rheumatology or endocrinology care — it is a complement to it.
Disclaimer: This article is provided for educational purposes and reflects one physician’s clinical perspective. It is not a substitute for individualized medical care. Autoimmune diseases require ongoing medical management. Consult a qualified physician before modifying any treatment regimen.